177 – CARTITUDE-1: Phase 1b/2 Study of Ciltacabtagene Autoleucel, a B-Cell Maturation Antigen–Directed Chimeric Antigen Receptor T Cell Therapy, in Relapsed/Refractory Multiple Myeloma
Author(s): Deepu Madduri, MD1*, Jesus G. Berdeja, MD2, Saad Z. Usmani, MD, MBBS, MBA3, Andrzej Jakubowiak, MD, PhD4, Mounzer Agha, MD5, Adam D. Cohen, MD6, A. Keith Stewart, MBChB7*, Parameswaran Hari, MBBS, MD8, Myo Htut, MD9, Elizabeth O’Donnell, MD10*, Nikhil C. Munshi, MD11, David E. Avigan, MD12, Abhinav Deol, MD13, Alexander M. Lesokhin, MD14, Indrajeet Singh, PhD15, Enrique Zudaire, PhD15*, Tzu-Min Yeh16*, Alicia J. Allred, PhD15*, Yunsi Olyslager, MSc17*, Arnob Banerjee, MD15*, Jenna D. Goldberg, MD16*, Jordan M. Schecter, MD16, Carolyn C. Jackson, MD, MPH16*, William Deraedt, MSc17*, Sen Hong Zhuang, MD, PhD16*, Jeffrey R. Infante, MD15*, Dong Geng, PhD18*, Xiaoling Wu, PhD18*, Marlene J. Carrasco, MD, PhD, MPH18*, Muhammad Akram, MD18*, Farah Hossain, PharmD18*, Syed Rizvi, MD18, Frank Fan, MD, PhD19*, Sundar Jagannath, MD20*, Yi Lin, MD, PhD21 and Thomas Martin III, MD22
1Mount Sinai Medical Center, New York, NY
2Sarah Cannon Research Institute and Tennessee Oncology, Nashville, TN
3Levine Cancer Institute, Charlotte, NC
4University of Chicago, Chicago, IL
5UPMC Hillman Cancer Center, Pittsburgh, PA
6University of Pennsylvania, Abramson Cancer Center, Philadelphia, PA
7UHN and the Princess Margaret Cancer Centre, Toronto, ON, Canada
8Division of Hematology and Oncology, Department of Medicine, Medical College of Wisconsin, Brookfield, WI
9City of Hope Comprehensive Cancer Center, Duarte, CA
10Department of Hematology/Oncology, Massachusetts General Hospital, Boston, MA
11Department of Medical Oncology, Dana-Farber Cancer Institute, Harvard Medical School, Boston, MA
12Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, MA
13Department of Oncology, Blood and Marrow Stem Cell Transplant Program, Karmanos Cancer Institute/Wayne State University, Detroit, MI
14Memorial Sloan Kettering Cancer Center, New York, NY
15Janssen R&D, Spring House, PA
16Janssen R&D, Raritan, NJ
17Janssen R&D, Beerse, Belgium
18Legend Biotech USA Inc., Piscataway, NJ
19Nanjing Legend Biotechnology Co., Ltd., Nanjing, China
20Department of Hematology and Medical Oncology, Mount Sinai Medical Center, New York, NY
21Division of Hematology, Mayo Clinic, Rochester, MN
22University of California, San Francisco, San Francisco, CA
*signifies non-member of ASH
Disclosures: Madduri: Celgene: Consultancy, Honoraria; AbbVie: Consultancy, Honoraria; Foundation Medicine: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Legend: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Speaking Engagement, Speakers Bureau; Kinevant: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Speaking Engagement, Speakers Bureau; GSK: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Speaking Engagement, Speakers Bureau. Berdeja: Novartis: Research Funding; Lilly: Research Funding; Kite Pharma: Consultancy; CRISPR Therapeutics: Consultancy, Research Funding; Servier: Consultancy; Takeda: Consultancy, Research Funding; Amgen: Consultancy, Research Funding; Cellularity: Research Funding; Kesios: Research Funding; Acetylon: Research Funding; Janssen: Consultancy, Research Funding; Bluebird: Research Funding; Poseida: Research Funding; Glenmark: Research Funding; BMS: Consultancy, Research Funding; CURIS: Research Funding; Genentech, Inc.: Research Funding; EMD Sorono: Research Funding; Abbvie: Research Funding; Vivolux: Research Funding; Celgene: Consultancy, Research Funding; Legend: Consultancy; Karyopharm: Consultancy; Teva: Research Funding; Prothena: Consultancy; Constellation: Research Funding; Bioclinica: Consultancy. Usmani: Amgen: Consultancy, Honoraria, Other: Speaking Fees, Research Funding; BMS, Celgene: Consultancy, Honoraria, Other: Speaking Fees, Research Funding; Takeda: Consultancy, Honoraria, Other: Speaking Fees, Research Funding; SkylineDX: Consultancy, Research Funding; Seattle Genetics: Consultancy, Research Funding; Merck: Consultancy, Research Funding; Incyte: Research Funding; Pharmacyclics: Research Funding; Array Biopharma: Research Funding; GSK: Consultancy, Research Funding; Celgene: Other; Janssen: Consultancy, Honoraria, Other: Speaking Fees, Research Funding; Sanofi: Consultancy, Honoraria, Research Funding; Abbvie: Consultancy. Jakubowiak: Adaptive, Juno: Consultancy, Honoraria; AbbVie, Amgen, BMS/Celgene, GSK, Janssen, Karyopharm: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees. Cohen: Celgene: Membership on an entity’s Board of Directors or advisory committees; Takeda,: Membership on an entity’s Board of Directors or advisory committees; Janssen: Membership on an entity’s Board of Directors or advisory committees; GlaxoSmithKline: Membership on an entity’s Board of Directors or advisory committees; Kite Pharma: Membership on an entity’s Board of Directors or advisory committees; Oncopeptides: Membership on an entity’s Board of Directors or advisory committees; Seattle Genetics: Membership on an entity’s Board of Directors or advisory committees; AstraZeneca: Membership on an entity’s Board of Directors or advisory committees; Genentech/Roche: Membership on an entity’s Board of Directors or advisory committees; Bristol-Myers Squibb: Membership on an entity’s Board of Directors or advisory committees, Research Funding; Novartis: Other: Patents/Intellectual property licensed, Research Funding. Stewart: Janssen, BMS, Sanofi-Aventis, GSK: Honoraria; Tempus, Inc., Genomics England LLC: Membership on an entity’s Board of Directors or advisory committees. Hari: Amgen: Consultancy; BMS: Consultancy; GSK: Consultancy; Janssen: Consultancy; Takeda: Consultancy; Incyte Corporation: Consultancy. Htut: City of Hope Medical Center: Current Employment. Munshi: OncoPep: Consultancy, Current equity holder in private company, Membership on an entity’s Board of Directors or advisory committees, Patents & Royalties; BMS: Consultancy; Janssen: Consultancy; Adaptive: Consultancy; Legend: Consultancy; Amgen: Consultancy; Karyopharm: Consultancy; Takeda: Consultancy; AbbVie: Consultancy; C4: Current equity holder in private company. Deol: Novartis: Consultancy; Kite, a Gilead Company: Consultancy. Lesokhin: BMS: Consultancy, Honoraria, Research Funding; Genentech: Research Funding; Janssen: Research Funding; Juno: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Serametrix Inc.: Patents & Royalties; GenMab: Consultancy, Honoraria. Singh: Janssen: Current Employment. Zudaire: Janssen: Current Employment. Yeh: Janssen: Current Employment. Allred: Janssen: Current Employment. Olyslager: Janssen: Current Employment. Banerjee: Janssen: Current Employment. Goldberg: Johnson & Johnson: Current Employment, Current equity holder in publicly-traded company. Schecter: Janssen: Current Employment. Jackson: Janssen: Current Employment; Memorial Sloan Kettering Cancer Center: Consultancy. Deraedt: Janssen: Current Employment, Current equity holder in publicly-traded company. Zhuang: Janssen: Current Employment. Infante: Janssen: Current Employment. Geng: Legend Biotech USA Inc.: Current Employment. Wu: Legend Biotech USA Inc.: Current Employment. Carrasco: Legend Biotech USA Inc.: Current Employment. Akram: Legend Biotech USA Inc.: Current Employment. Hossain: Legend Biotech USA Inc.: Current Employment. Rizvi: Legend Biotech USA Inc.: Current Employment. Fan: Legend Biotech USA Inc.: Current Employment. Jagannath: BMS, Janssen, Karyopharm, Legend Biotech, Sanofi, Takeda: Consultancy. Lin: Kite, a Gilead Company: Consultancy, Research Funding; Janssen: Consultancy, Research Funding; Merck: Research Funding; Legend BioTech: Consultancy; Juno: Consultancy; Bluebird Bio: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; Novartis: Consultancy; Vineti: Consultancy; Takeda: Research Funding; Gamida Cells: Consultancy; Sorrento: Consultancy, Membership on an entity’s Board of Directors or advisory committees. Martin: AMGEN: Research Funding; Seattle Genetics: Research Funding; Janssen: Research Funding; GSK: Consultancy; Sanofi: Research Funding.
Background: Ciltacabtagene autoleucel (cilta-cel; JNJ-68284528; LCAR-B38M CAR-T cells) is a chimeric antigen receptor T (CAR-T) cell therapy with 2 B-cell maturation antigen–targeting single-domain antibodies designed to confer avidity. In the phase 1 LEGEND-2 study in China, LCAR-B38M yielded deep, durable responses with a manageable safety profile in patients (pts) with relapsed/refractory multiple myeloma (R/R MM). The phase 1b/2 CARTITUDE-1 study (NCT03548207) is further evaluating cilta-cel in this pt population in the US. We present updated data from the phase 1b portion along with initial phase 2 data.
Methods: Eligible pts (aged ≥18 y) were diagnosed with MM per International Myeloma Working Group (IMWG) criteria and had measurable disease, Eastern Cooperative Oncology Group performance status ≤1, received ≥3 prior regimens or were double-refractory to a proteasome inhibitor and immunomodulatory drug, and received an anti-CD38 antibody. After apheresis, bridging therapy was permitted. Cyclophosphamide 300 mg/m2 and fludarabine 30 mg/m2 daily for 3 d were used for lymphodepletion. A single infusion of cilta-cel at a target dose of 0.75×106 (range 0.5–1.0×106) CAR+ viable T cells/kg was administered 5–7 d after start of lymphodepletion. The primary objective of the phase 1b portion was to characterize cilta-cel safety and establish the recommended phase 2 dose; the primary objective of the phase 2 portion was to evaluate cilta-cel efficacy. Response was assessed per IMWG criteria and minimal residual disease (MRD) by next-generation sequencing. Adverse events (AEs) were graded using CTCAE v5.0. In the phase 1b portion, cytokine release syndrome (CRS) was graded by Lee et al (Blood 2014) and neurotoxicity by CTCAE v5.0; in the phase 2 portion, CRS and neurotoxicity were graded by American Society for Transplantation and Cellular Therapy (ASTCT) criteria. In this combined analysis, Lee et al and CTCAE v5.0 were mapped to ASTCT criteria for CRS and immune effector cell‐associated neurotoxicity syndrome (ICANS), respectively.
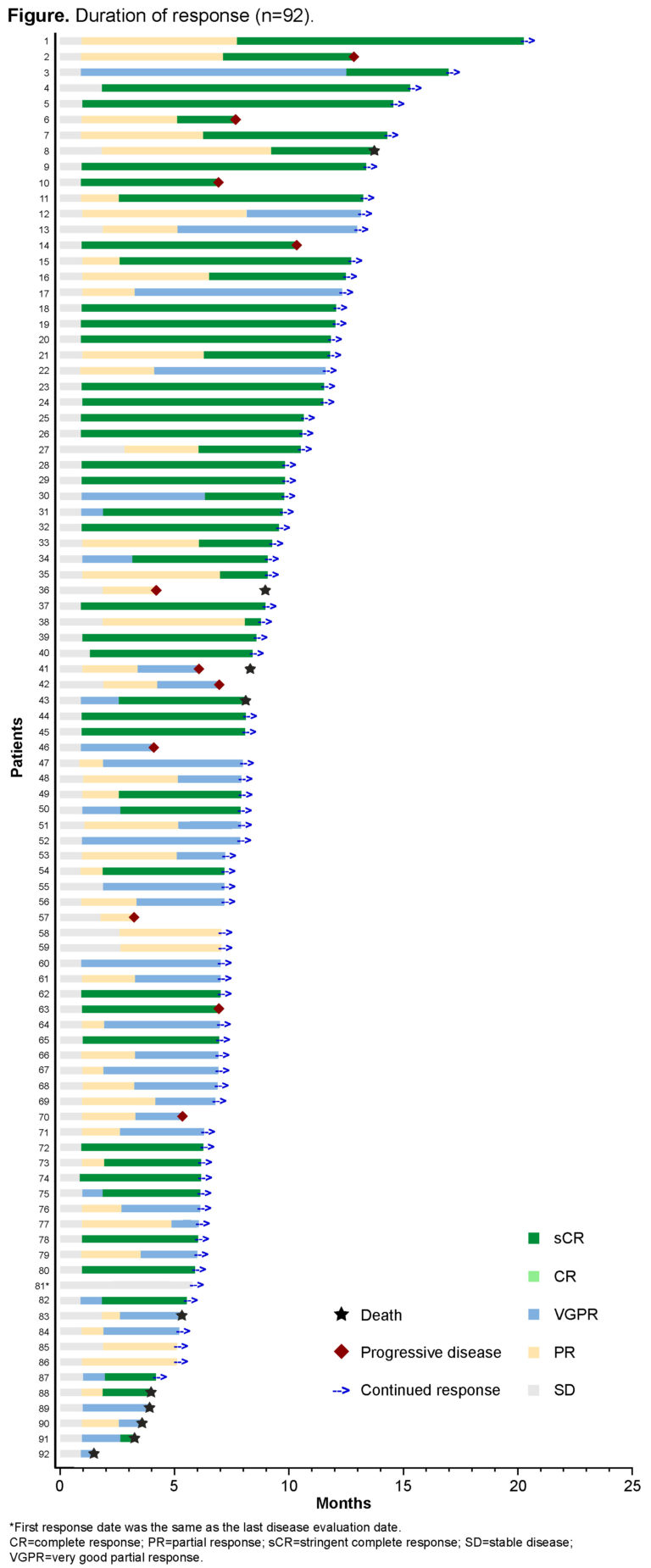
Results: As of the May 20, 2020 clinical cutoff, 97 pts (58.8% male; median age 61.0 y [range 43–78]) with R/R MM received cilta-cel (29 in phase 1b; 68 in phase 2). Median follow-up duration was 8.8 mo (range 1.5–20.4). Pts had received a median of 6 prior lines of therapy (range 3–18); 83.5% were penta-exposed, 87.6% were triple-refractory, 41.2% were penta-refractory, and 97.9% were refractory to last line of therapy. Overall response rate per independent review committee (primary endpoint) was 94.8% (95% CI 88.4–98.3), with a stringent complete response rate of 55.7% (95% CI 45.2–65.8), very good partial response rate of 32.0% (95% CI 22.9–42.2), and partial response rate of 7.2% (95% CI 3.0–14.3). All pts achieved a reduction in M-protein. Median time to first response was 1.0 mo (range 0.9–5.8; 80.4% ≤1.0 mo), and median time to complete response or better was 1.8 mo (range 0.9–12.5; 74.1% ≤3.0 mo); responses deepened over time (Figure). Median duration of response was not reached (NR). Of 52 MRD-evaluable pts, 94.2% were MRD-negative at 10-5. The 6-mo progression-free survival (PFS) and overall survival (OS) rates (95% CI) were 87.4% (78.9–92.7) and 93.8% (86.7–97.2), respectively; median PFS and OS were NR. Ten deaths occurred during the study; 8 were due to AEs (both related and unrelated; CRS/hemophagocytic lymphohistiocytosis, neurotoxicity, respiratory failure, sepsis, septic shock, pneumonia, lung abscess, and acute myelogenous leukemia [n=1 each]), and 2 due to progressive disease. AEs reported in >70% of pts were CRS (94.8%; grade [gr] 3/4 4.1%), neutropenia (90.7%; gr 3/4 90.7%), anemia (81.4%; gr 3/4 68.0%), and thrombocytopenia (79.4%; gr 3/4 59.8%). Median time to CRS onset was 7.0 d (range 1–12) and median duration 4.0 d (range 1–27, excluding n=1 with 97 d). CAR-T cell–related neurotoxicity was reported in 20.6% of pts (gr 3/4 10.3%). Cilta-cel CAR+ T cells showed maximum peripheral expansion at 14 d (range 9–43). Among pts with 6 mo’ individual follow-up, 67% had cilta-cel CAR+ T cells below the level of quantification (2 cells/µL) in peripheral blood.
Conclusions: Preliminary phase 1b/2 data from CARTITUDE-1 indicate a single low-dose infusion of cilta-cel leads to early, deep, and durable responses in heavily pretreated pts with MM with a safety profile consistent with LEGEND-2. Further investigation of cilta-cel in other MM populations is underway.
ASH Disclaimer
© 2020 American Society of Hematology. All rights reserved. ASH® and the ASH logo are registered trademarks of the American Society of Hematology. Used with permission.
This content is provided for informational purposes and personal use only, and is not intended to provide medical advice, diagnosis, or treatment or for commercial use. The ideas and opinions expressed herein do not necessarily reflect those of the American Society of Hematology (ASH). The mention of any product, service or therapy in this collection of materials should not be construed as an endorsement of the products mentioned.
Site provided by Springer Healthcare. Part of the Springer Nature Group.