OlympiA: A phase III, multicenter, randomized, placebo-controlled trial of adjuvant olaparib after (neo)adjuvant chemotherapy in patients with germline BRCA1/2 mutations and high-risk HER2-negative early breast cancer
Authors: Andrew Tutt, Judy Ellen Garber, Bella Kaufman, Giuseppe Viale, Debora Fumagalli, Priya Rastogi, Richard D. Gelber, Evandro de Azambuja, Anitra Fielding, Judith Balmana Gelpi, Karen A. Gelmon, Nigel Baker, Amal Arahmani, Elzbieta Senkus-Konefka, Eleanor Mc Fadden, Vassiliki Karantza, Sunil R. Lakhani, Greg Yothers, Christine Campbell, Charles E. Geyer; Breast Cancer Now, Toby Robins Research Centre, The Institute of Cancer Research, and The Breast Cancer Now Unit, Guy’s Hospital Cancer Centre, King’s College London, London, United Kingdom; Dana-Farber Cancer Institute, Harvard Medical School, Boston, MA; Breast Oncology Institute, Sheba Medical Center, Tel Aviv University, Tel Hashomer, Israel; University of Milan, Milan, Italy; Breast International Group, Brussels, Belgium; University of Pittsburgh, Pittsburgh, PA; Dana-Farber Cancer Institute, Harvard Medical School, and Harvard TH Chan School of Public Health, Frontier Science Foundation, Boston, MA; Institut Jules Bordet and Université Libre de Bruxelles (U.L.B), Brussels, Belgium; AstraZeneca, Gaithersburg, MD; Hospital Vall d’Hebron and Vall d’Hebron Institute of Oncology, Barcelona, Spain; Department of Medical Oncology, BC Cancer, Vancouver, BC, Canada; AstraZeneca, Cambridge, United Kingdom; Medical University of Gdańsk, Department of Oncology and Radiotherapy, Gdańsk, Poland; Frontier Science (Scotland) Ltd, Kincraig, United Kingdom; Merck & Co., Inc., Kenilworth, NJ; The University of Queensland Centre for Clinical Research & Pathology Queensland, Brisbane, Australia; University of Pittsburgh Department of Biostatistics, and NRG Oncology Statistics and Data Management Center, Pittsburgh, PA; Frontier Science (Scotland) Ltd., Kincraig, United Kingdom; Houston Methodist Cancer Center, Houston, TX
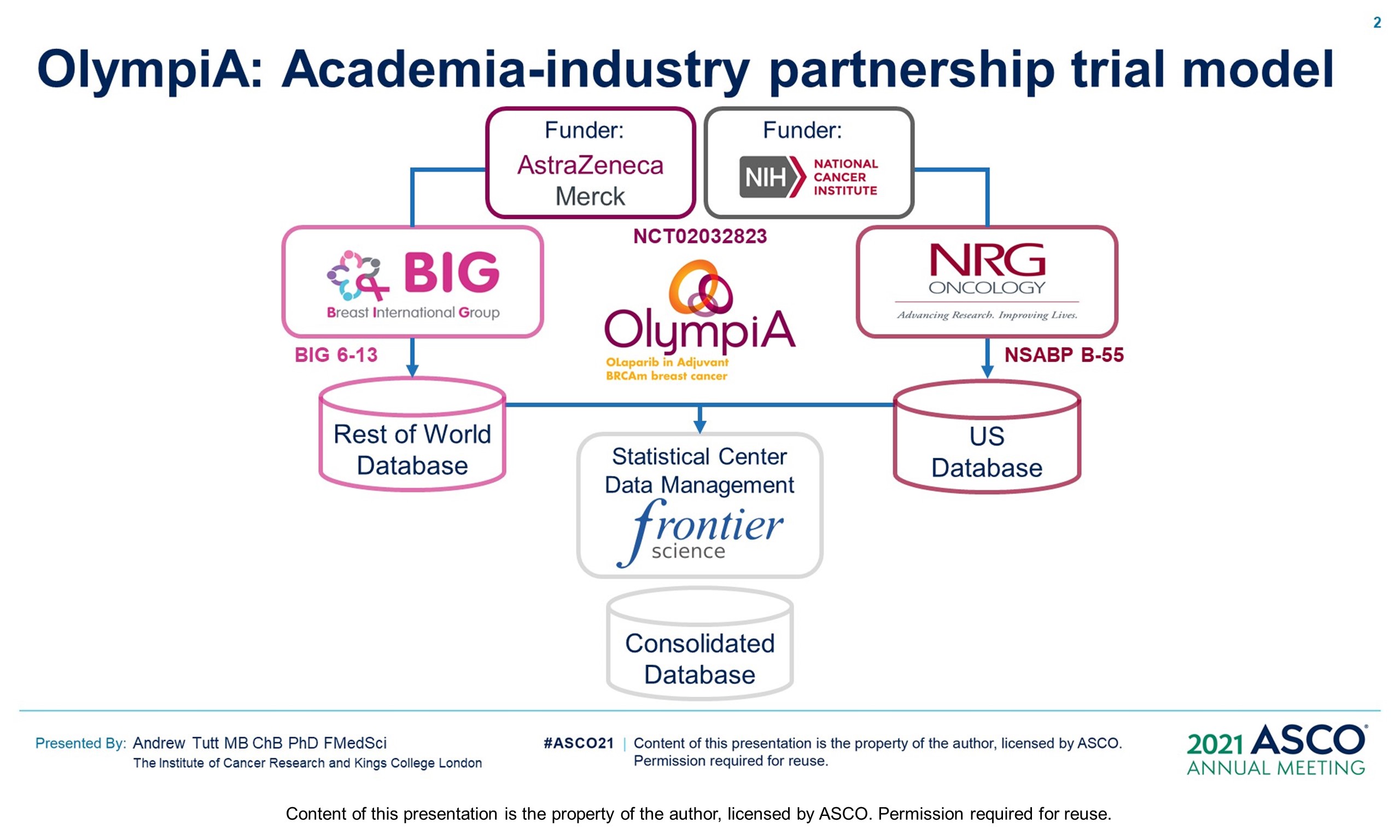
Research Funding: The study was funded by the National Cancer Institute (grants U10CA180868 UG1CA189867 U10CA180822) and, Pharmaceutical/Biotech Company, OlympiA is a partnership between Breast International Group (BIG), NRG Oncology, the US National Cancer Institute (NCI), Frontier Science & Technology Research Foundation (FSTRF), AstraZeneca and MSD. The trial is sponsored by NRG Oncology in the US and by AstraZeneca outside the US
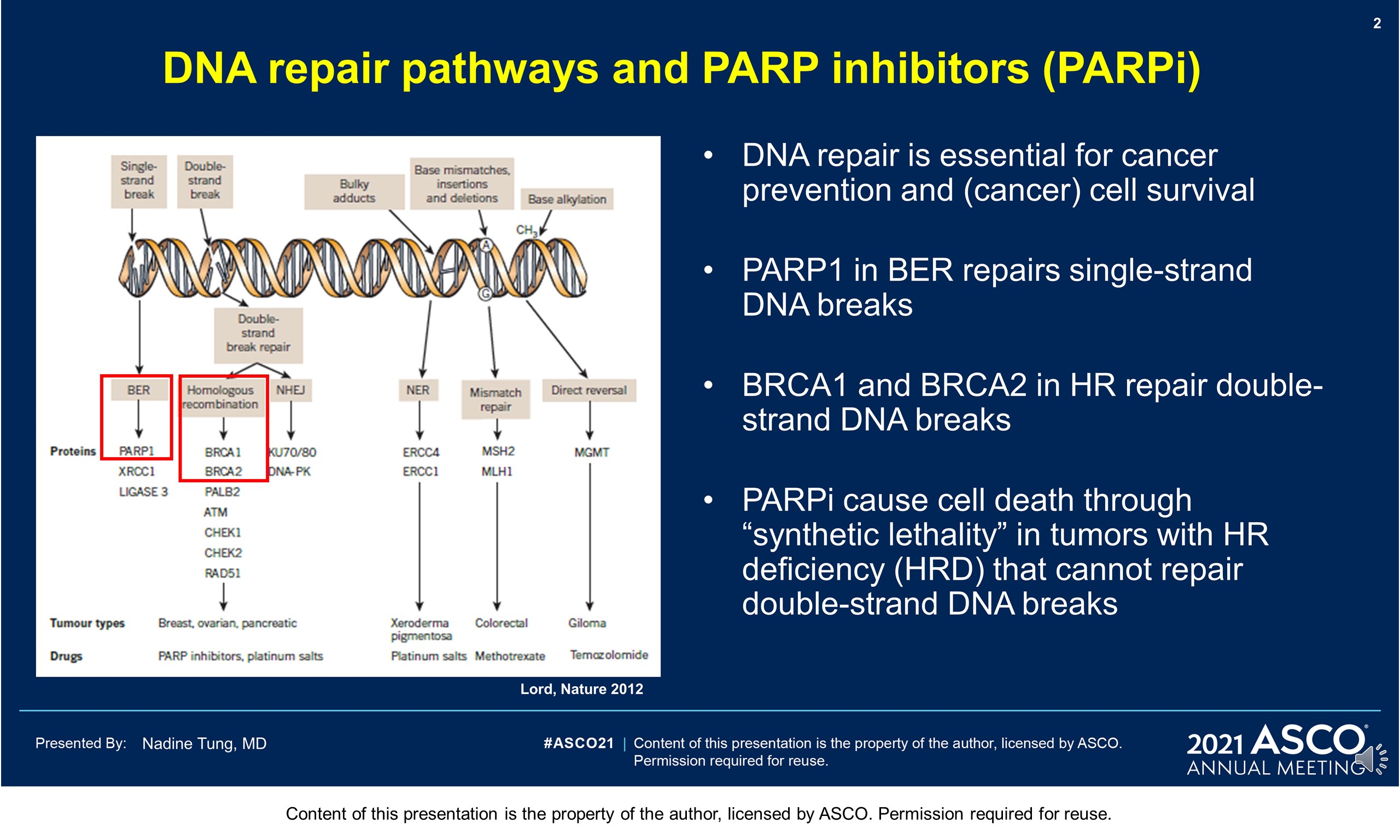
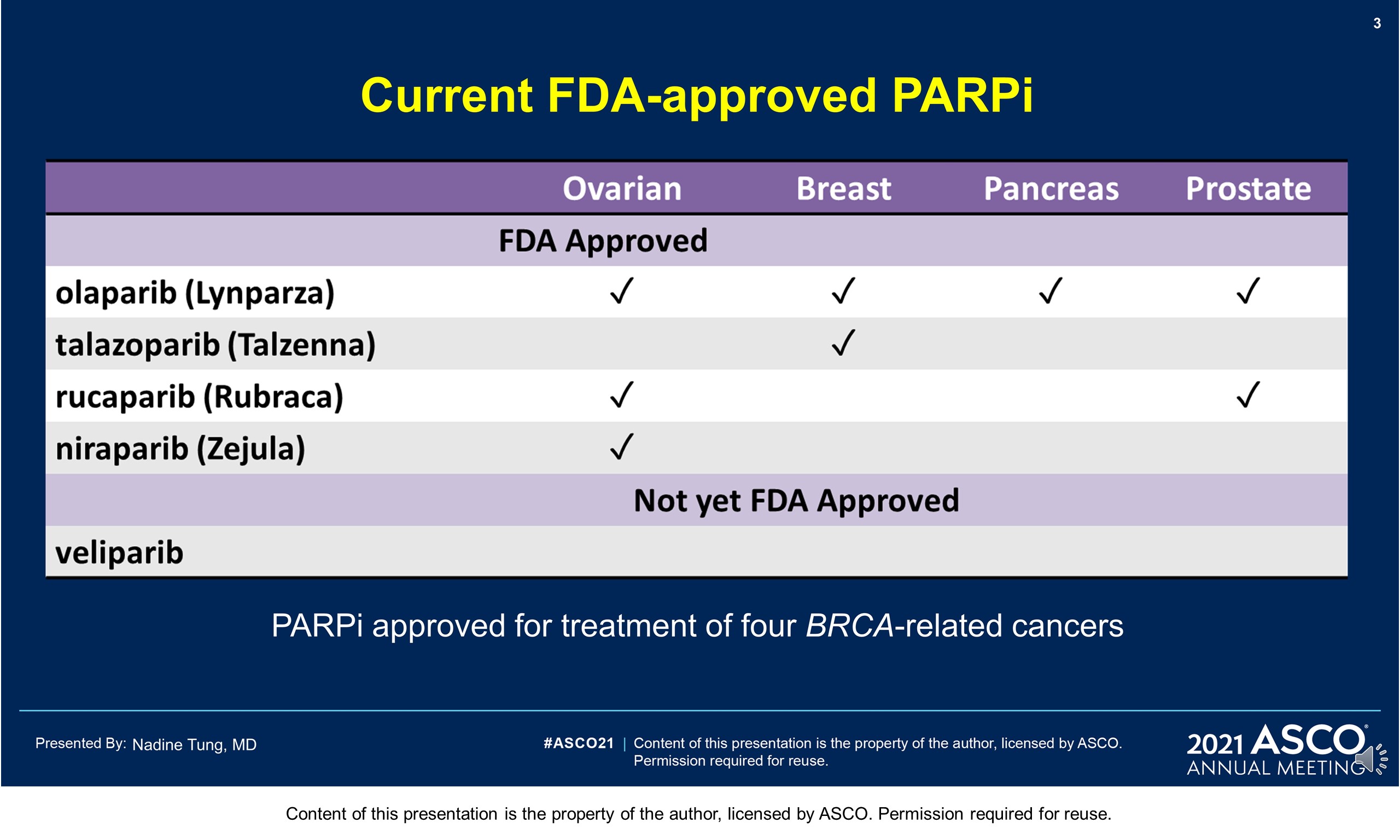
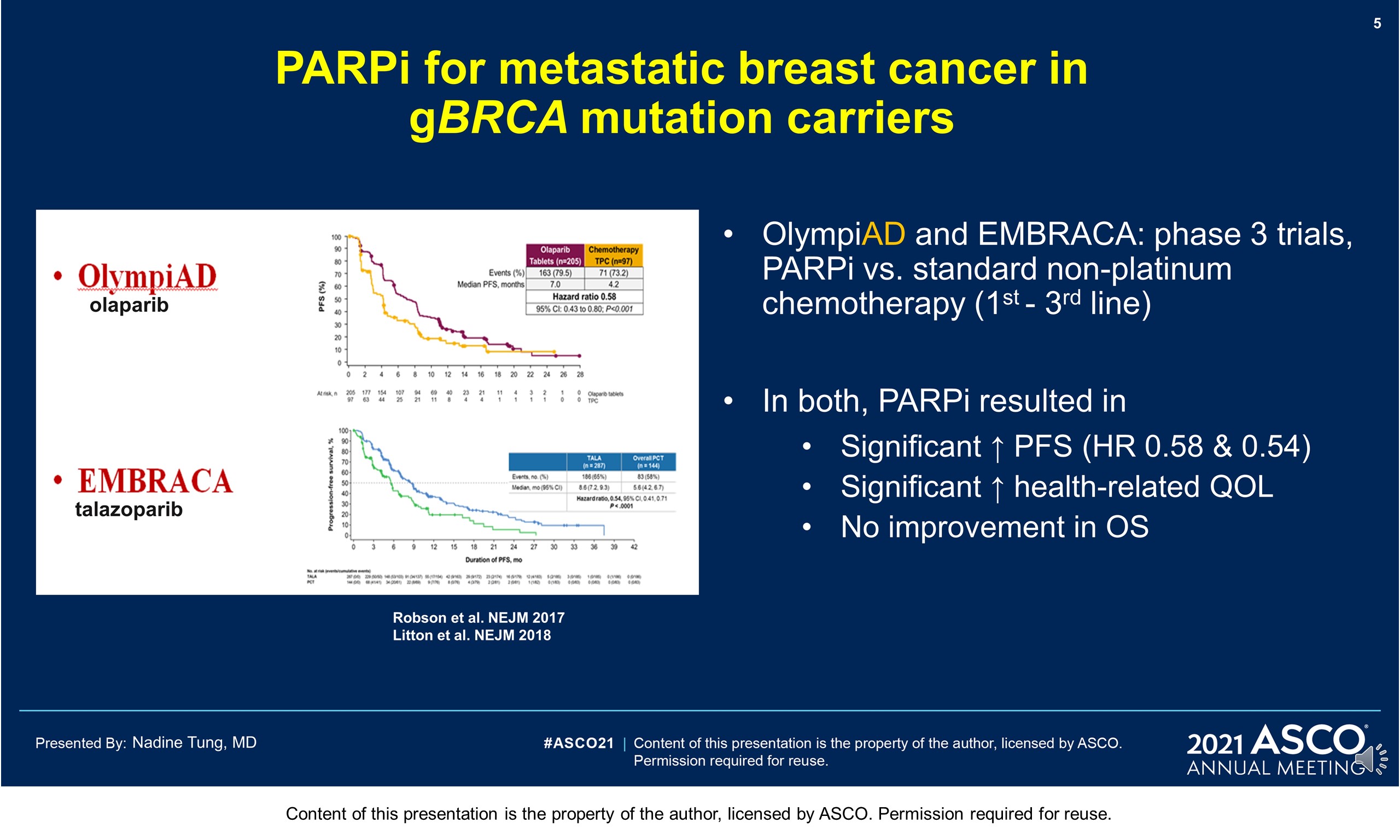
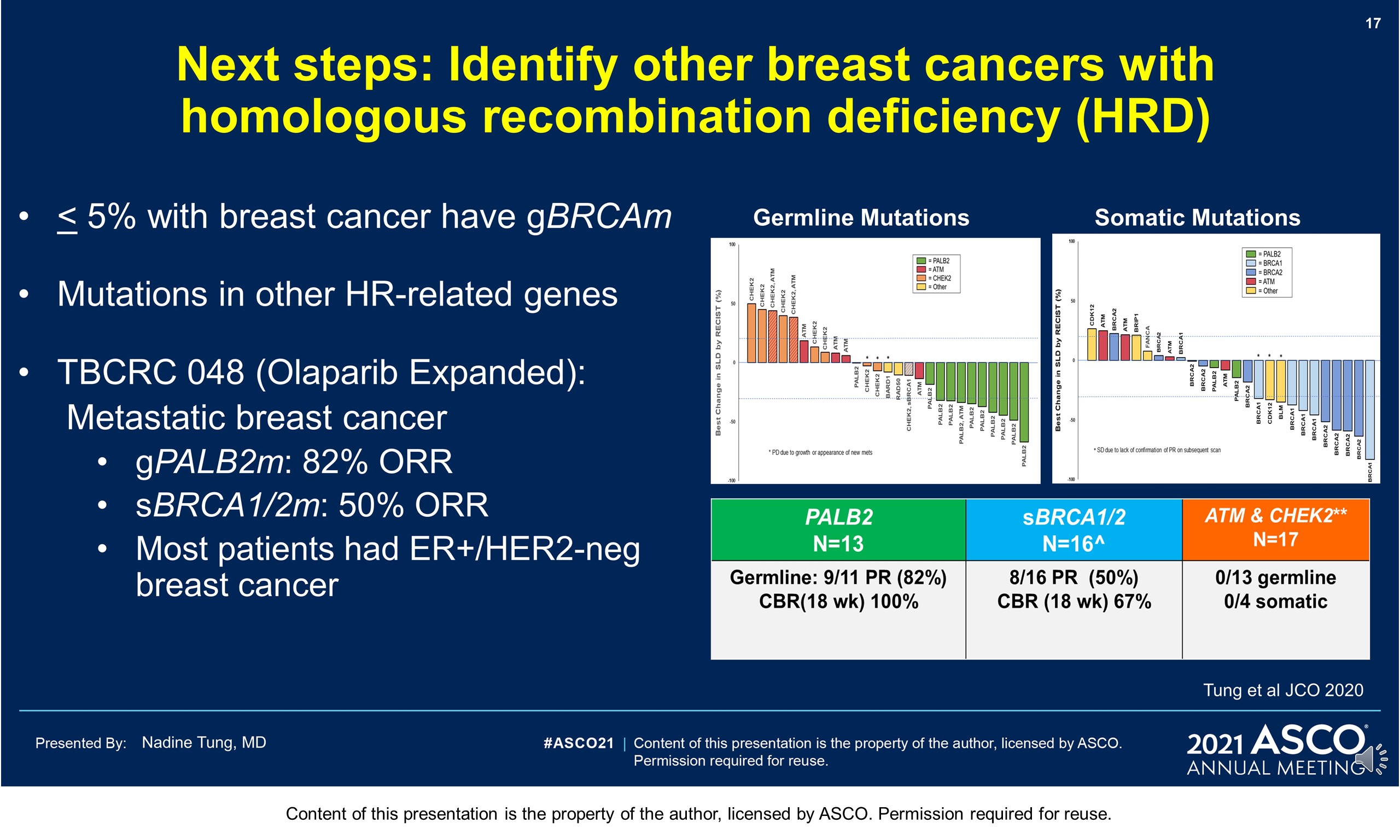
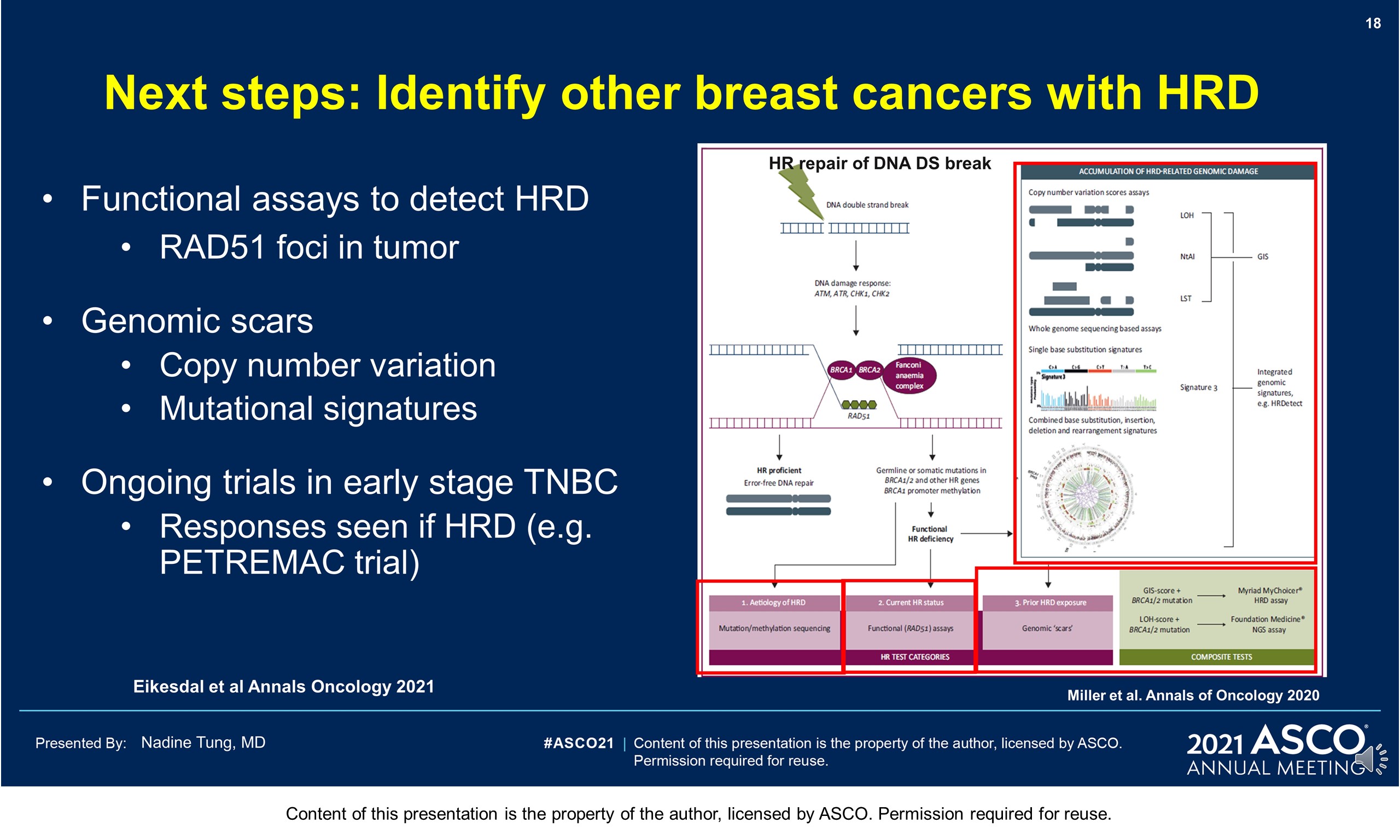
Background: PARP inhibitors (PARPi) target cancers with homologous recombination repair defects by synthetic lethality. The PARPi olaparib (OL) is licensed for metastatic HER2-negative breast cancer with BRCA1/2 germline mutation (gBRCAm). Despite (neo)adjuvant chemotherapy ([N]ACT), recurrence rates in patients (pts) with gBRCAm early breast cancer (EBC) can be high. Novel adjuvant treatments are needed.
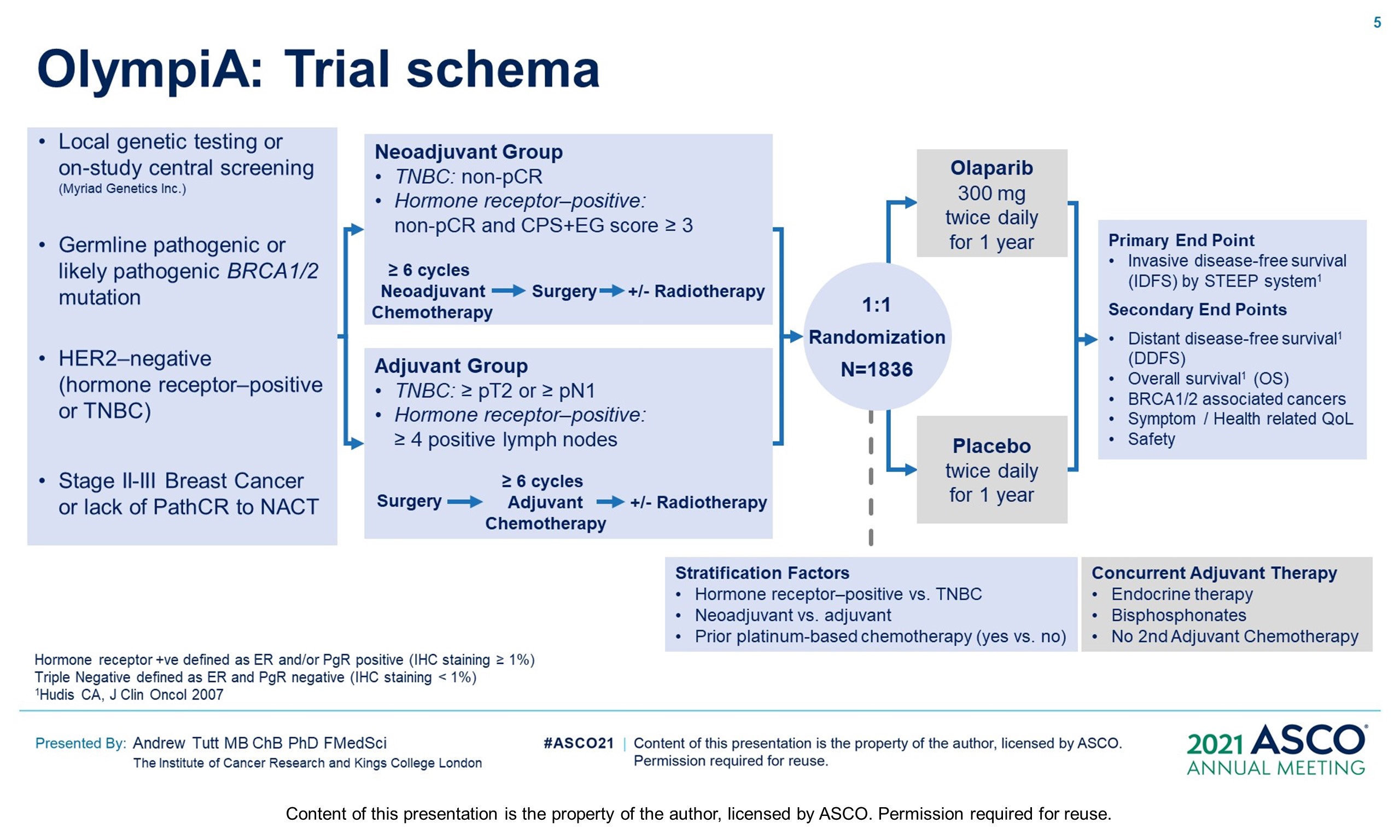
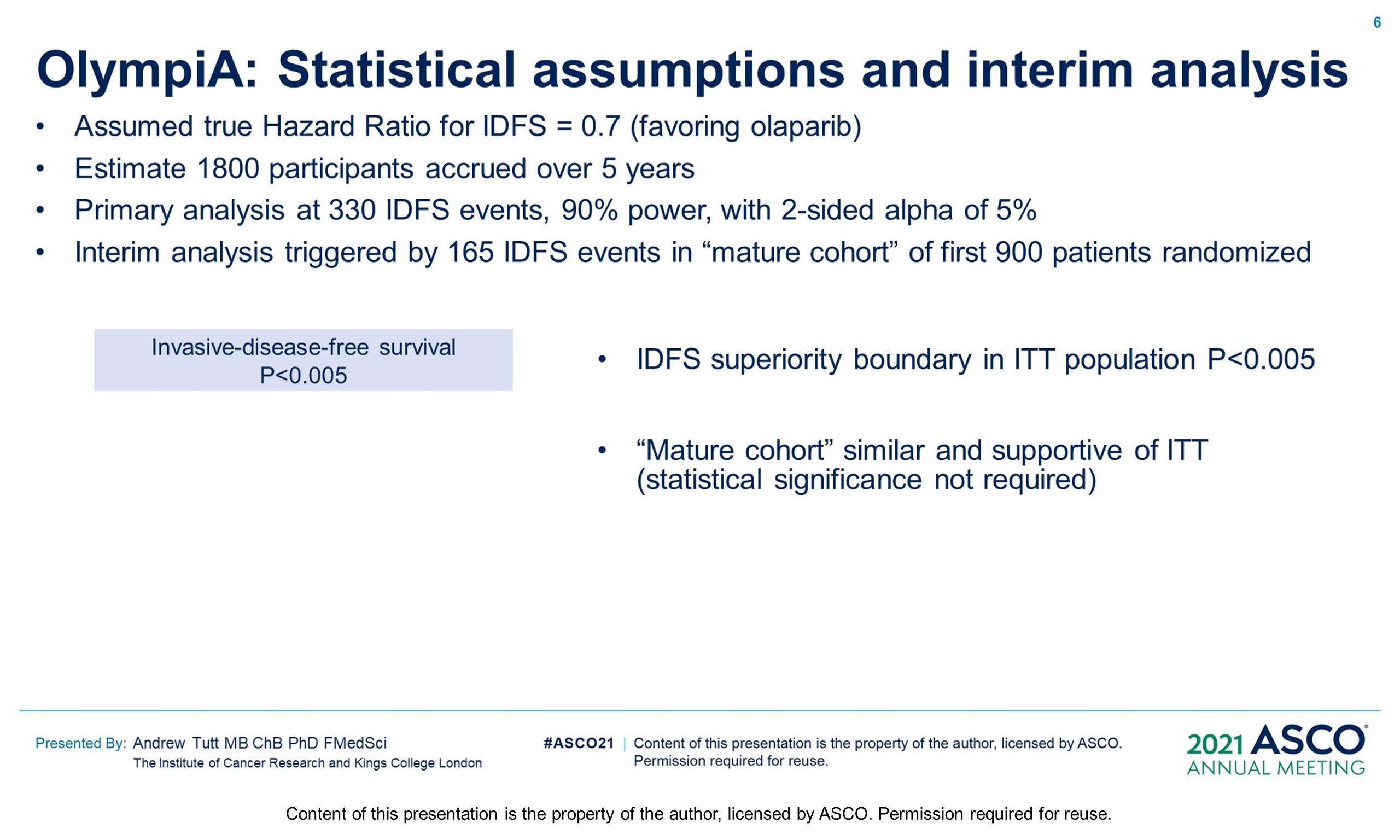
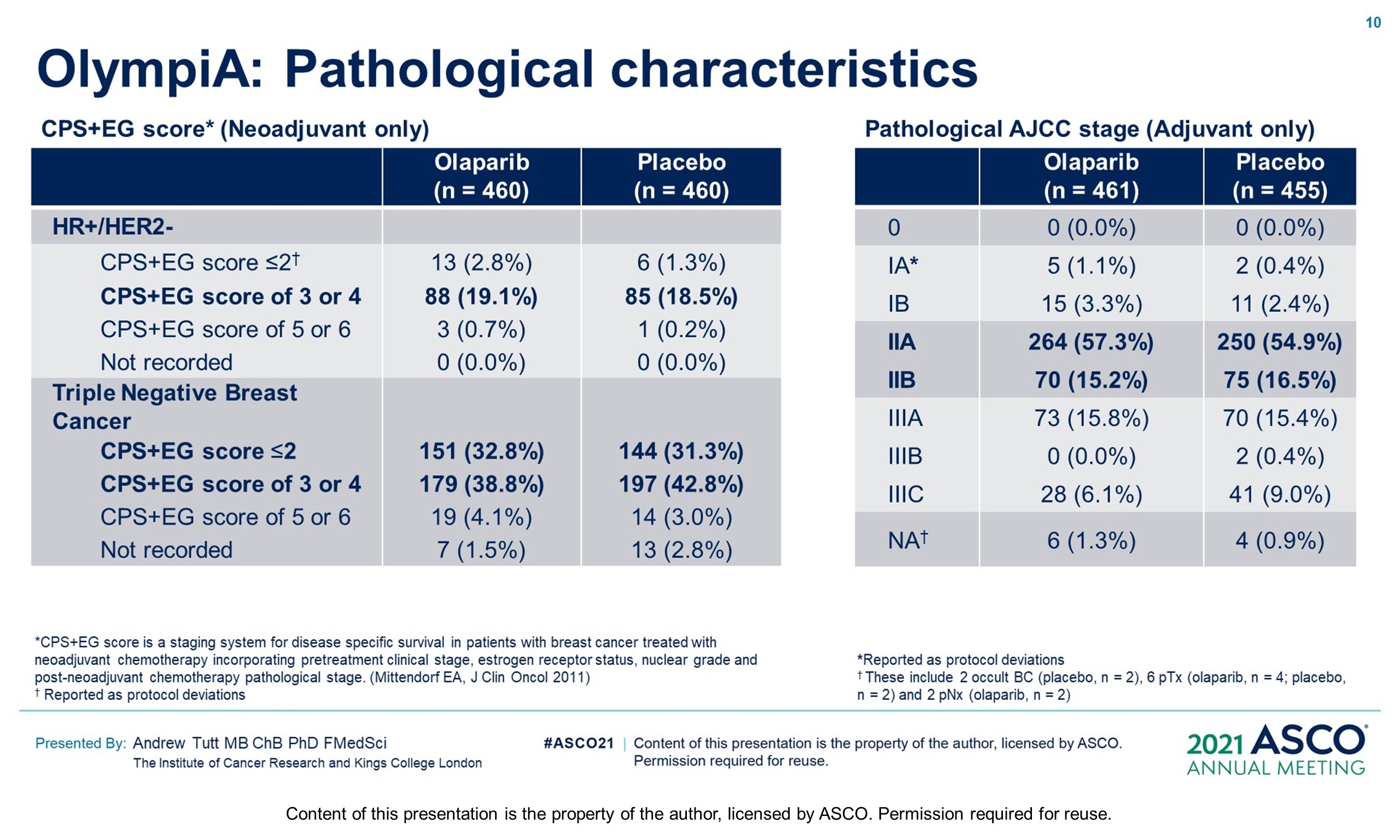
Methods: OlympiA (NCT02032823), a randomized, double-blind, phase III study, enrolled pts with gBRCAm and HER2-negative (TNBC or hormone-receptor+ [HR+]) high-risk EBC after primary local treatment and ACT/NACT. Eligible pts with TNBC had ≥pT2 or ≥pN1 disease prior to ACT or non-pCR after NACT; those with HR+ BC had ≥4 positive nodes prior to ACT or non-pCR and CPS&EG score ≥3 after NACT. Pts were randomized 1:1 to 1 year of continuous oral OL (300 mg BID) or placebo (PL). Endocrine therapy and bisphosphonates were allowed. The primary endpoint was invasive disease-free survival (IDFS) in the ITT population. Secondary endpoints included distant DFS (DDFS), overall survival (OS) and safety. Safety analysis included adverse events of special interest (AESI) (myelodysplastic syndrome/ acute myeloid leukemia, new primary malignancy, pneumonitis). Per protocol IDMC interim analysis (IA) review was triggered at 165 IDFS events in the first 900 pts, with superiority boundaries based on a hierarchical multiple testing procedure: P < 0.005 for IDFS, followed by P < 0.005 for DDFS and p<0.01 for OS.
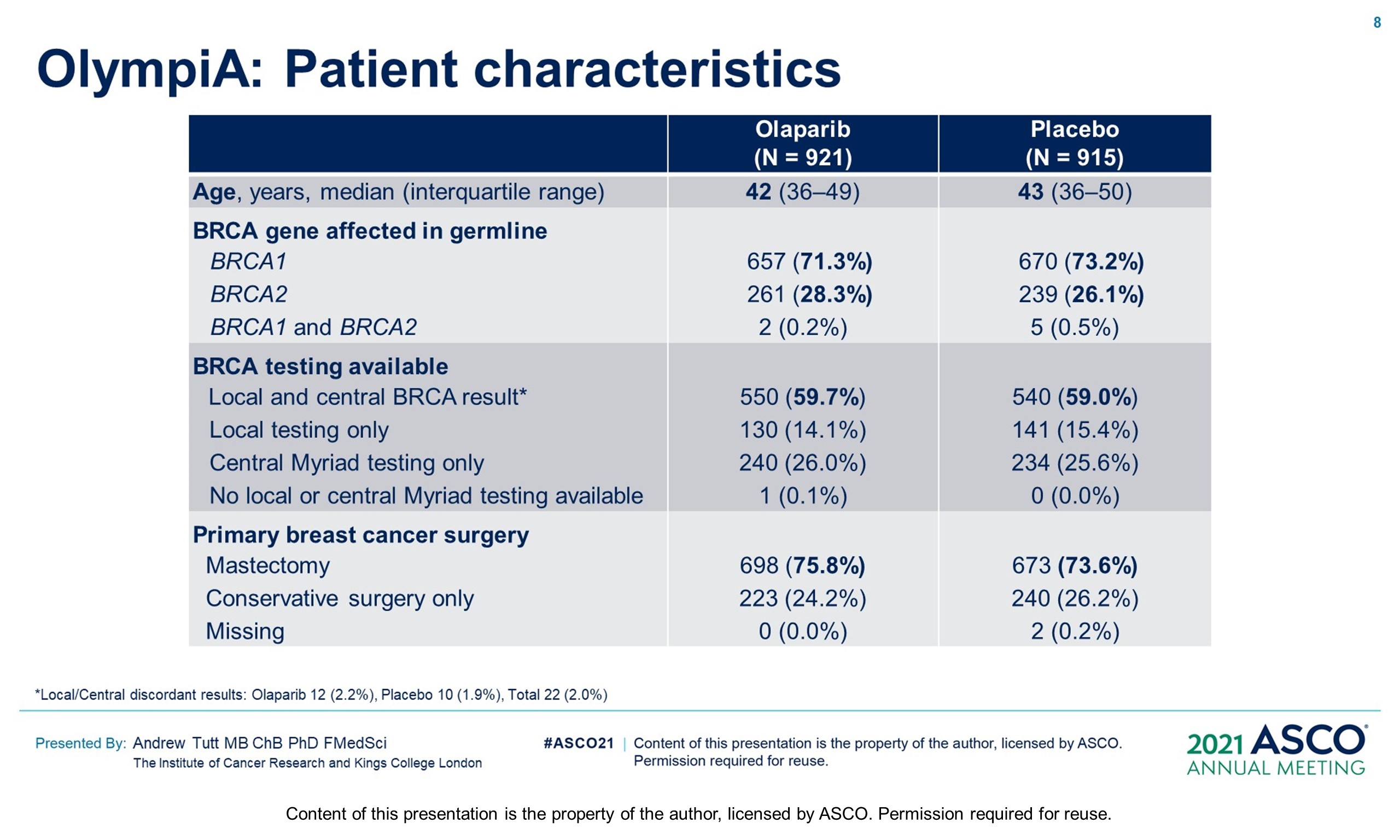
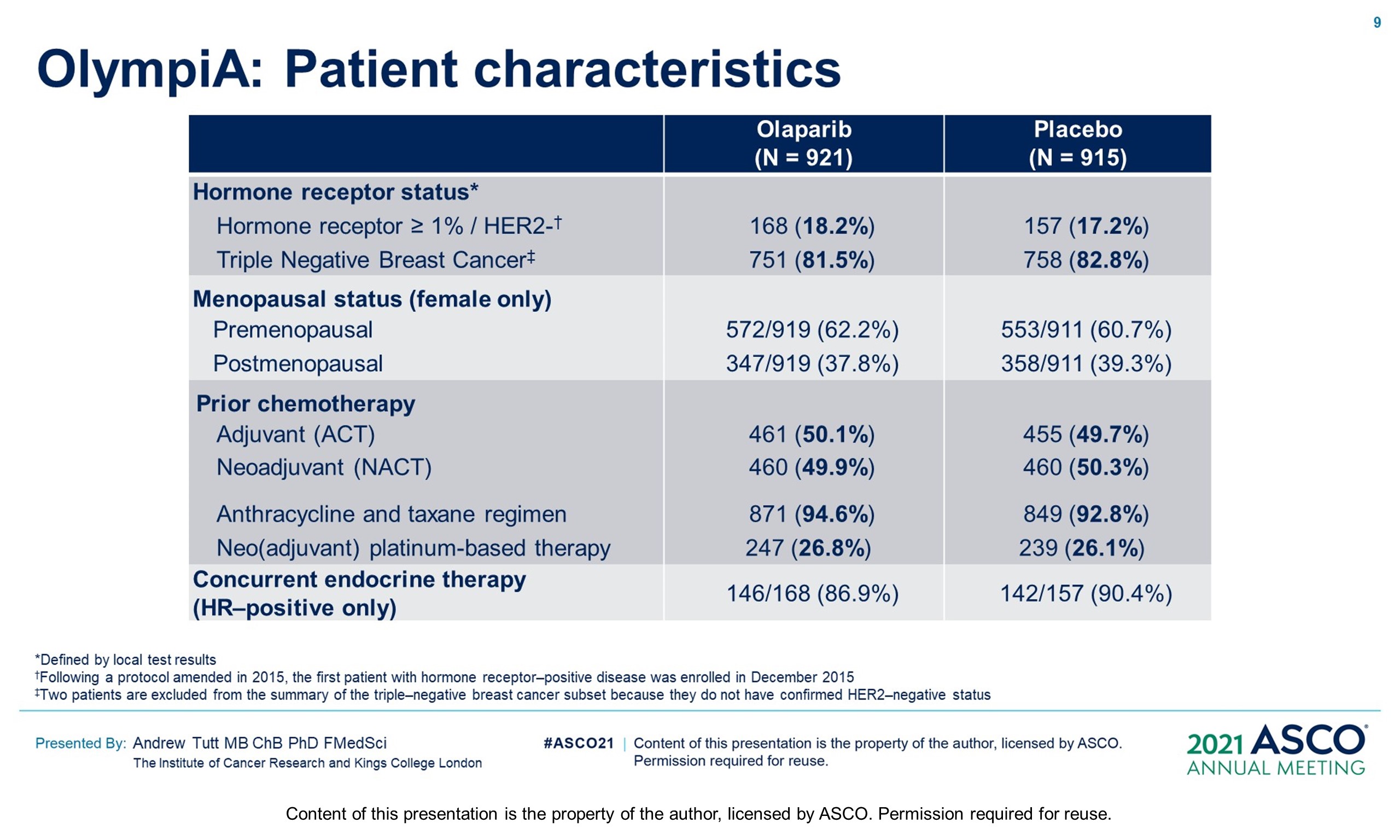
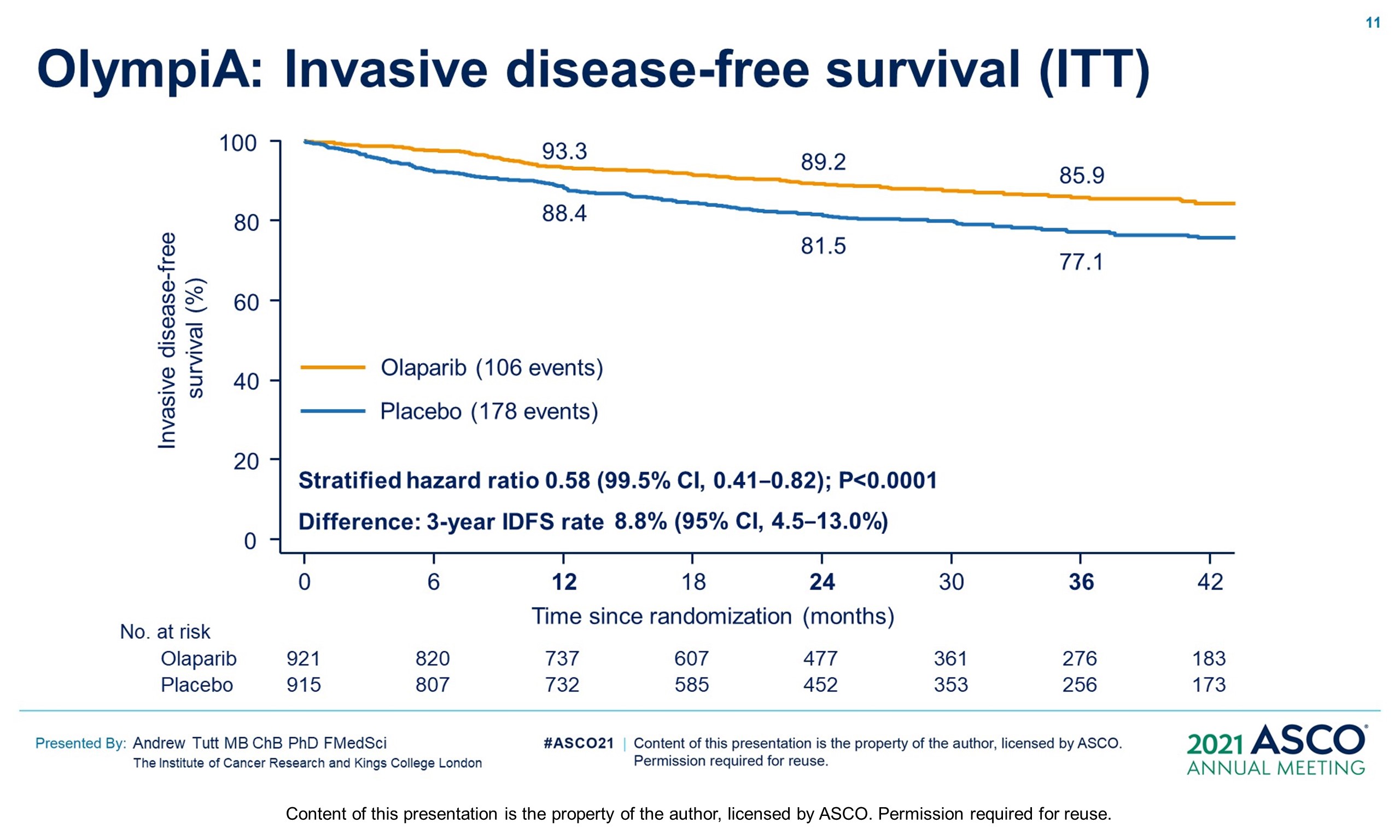
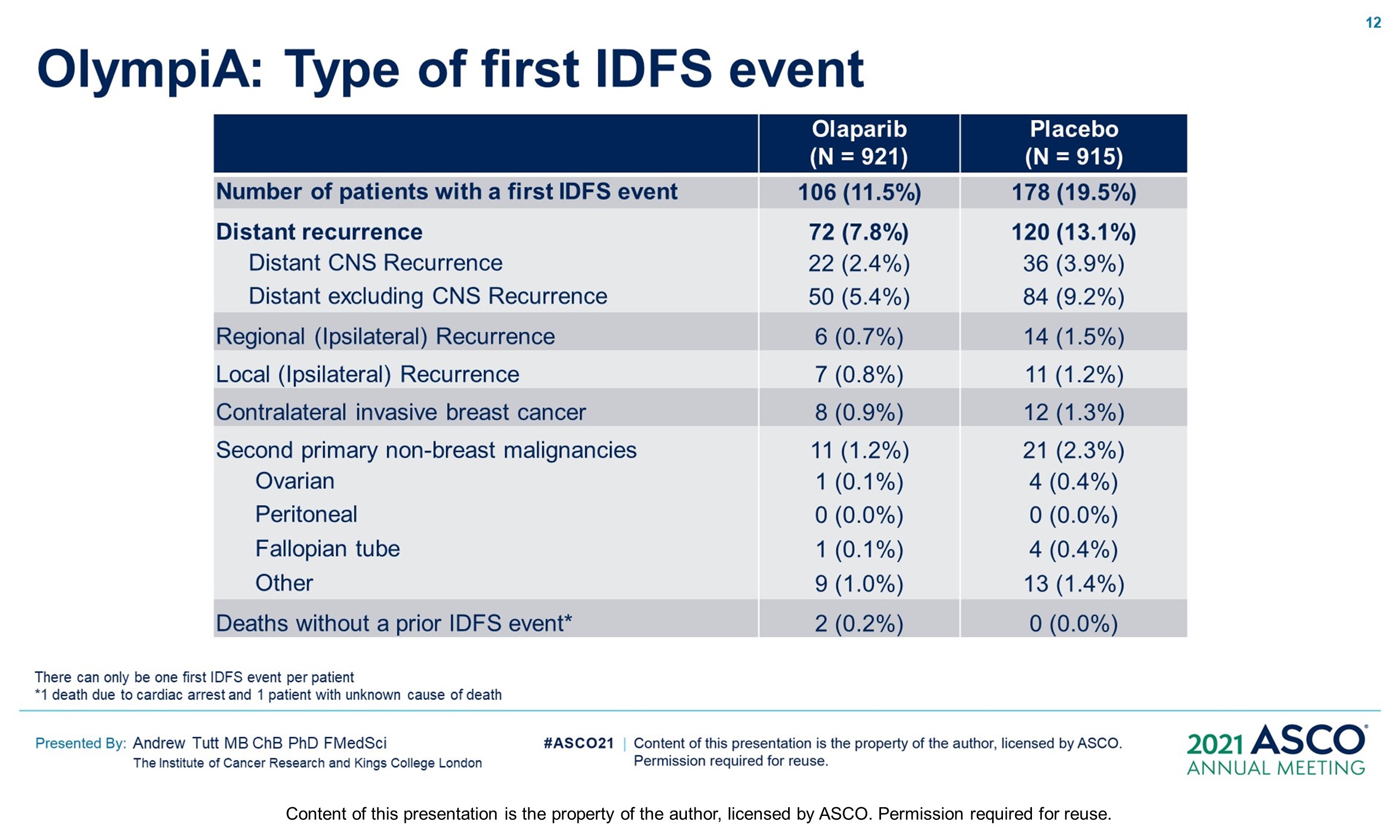
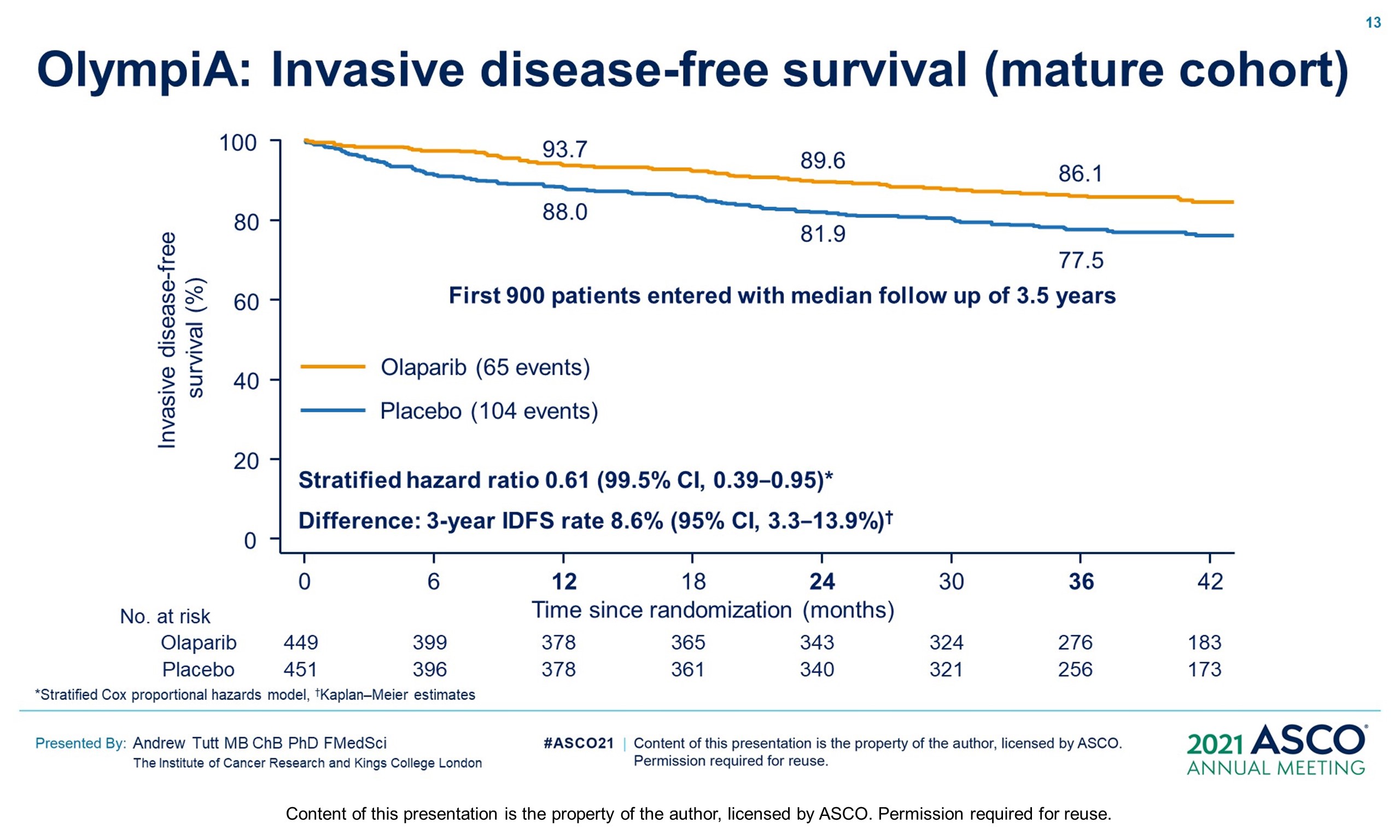
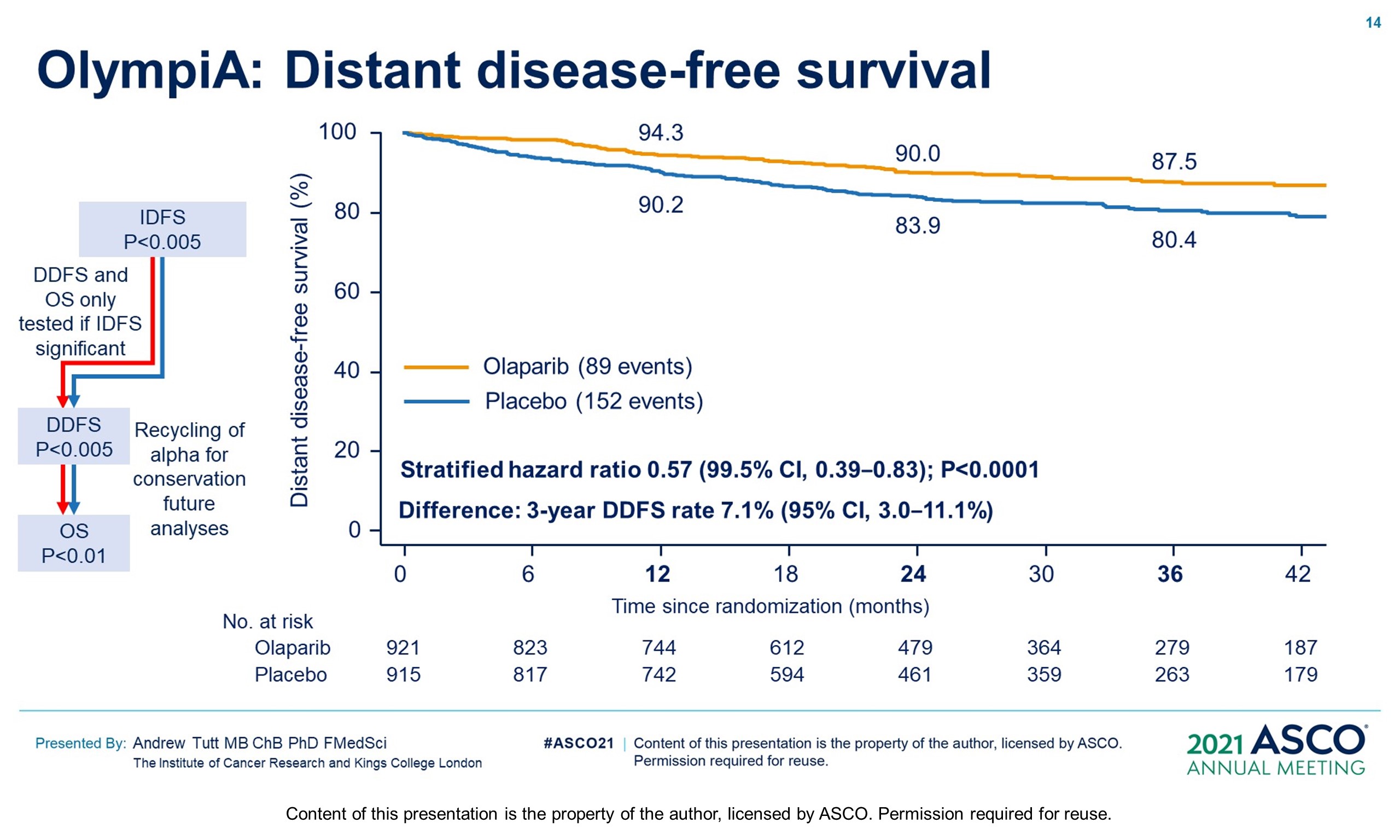
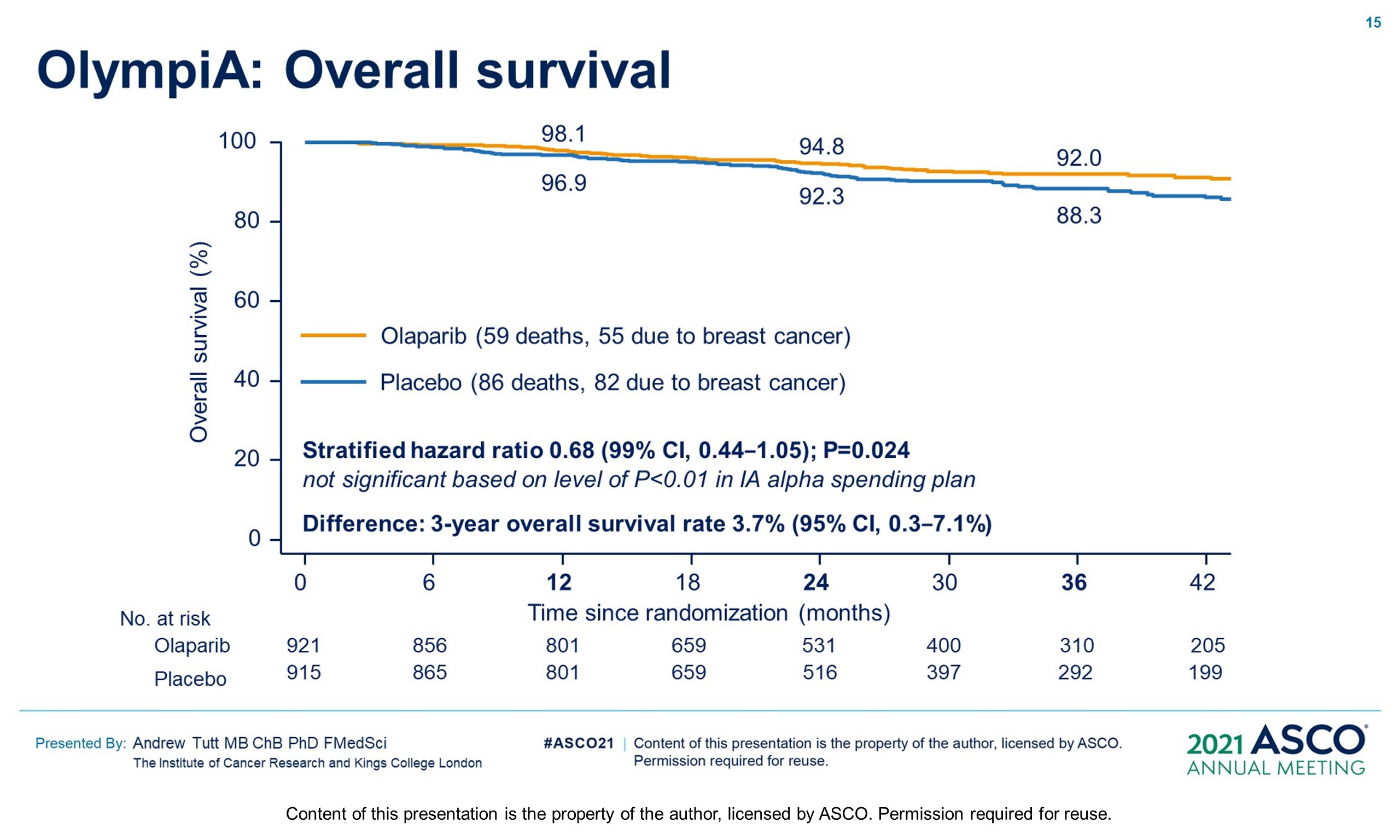
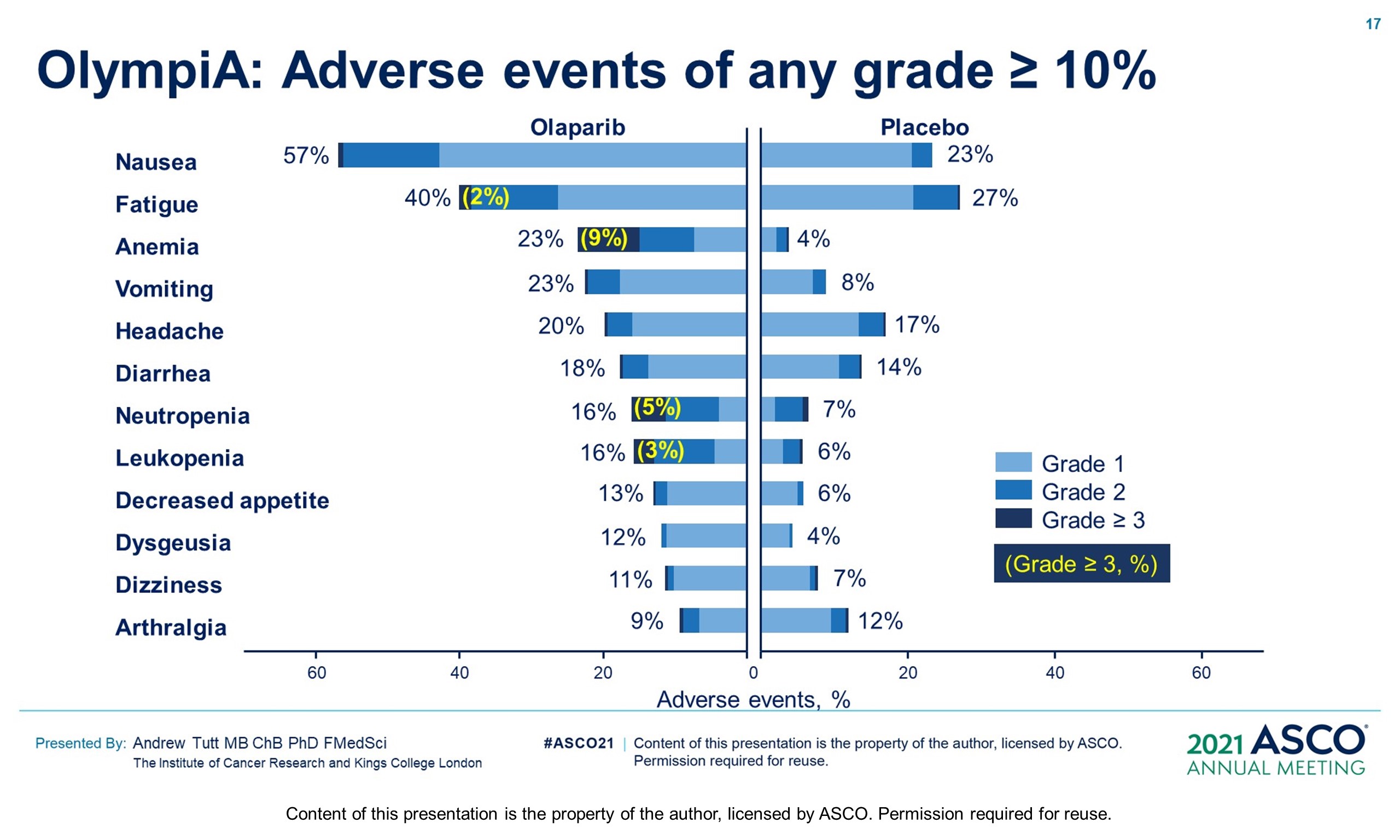
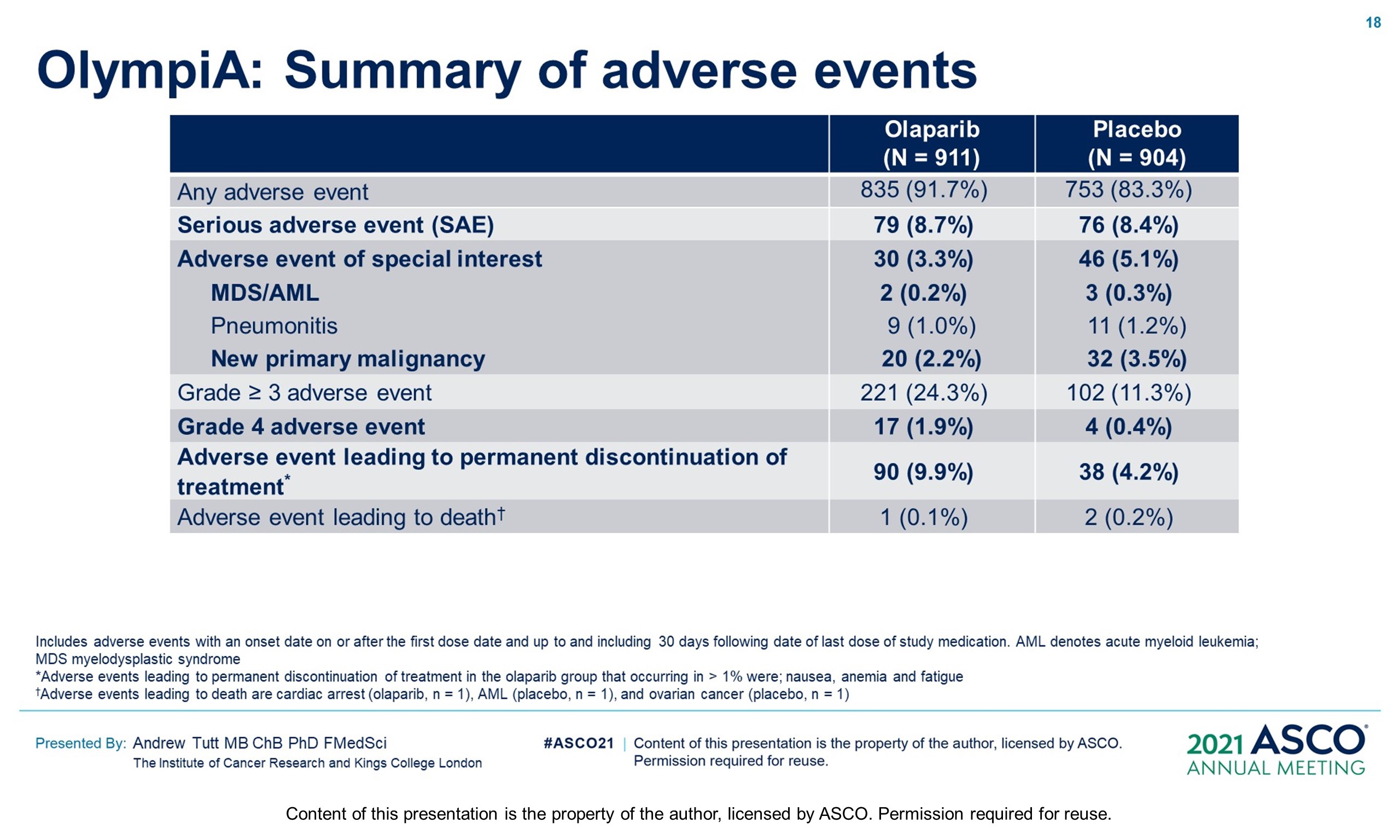
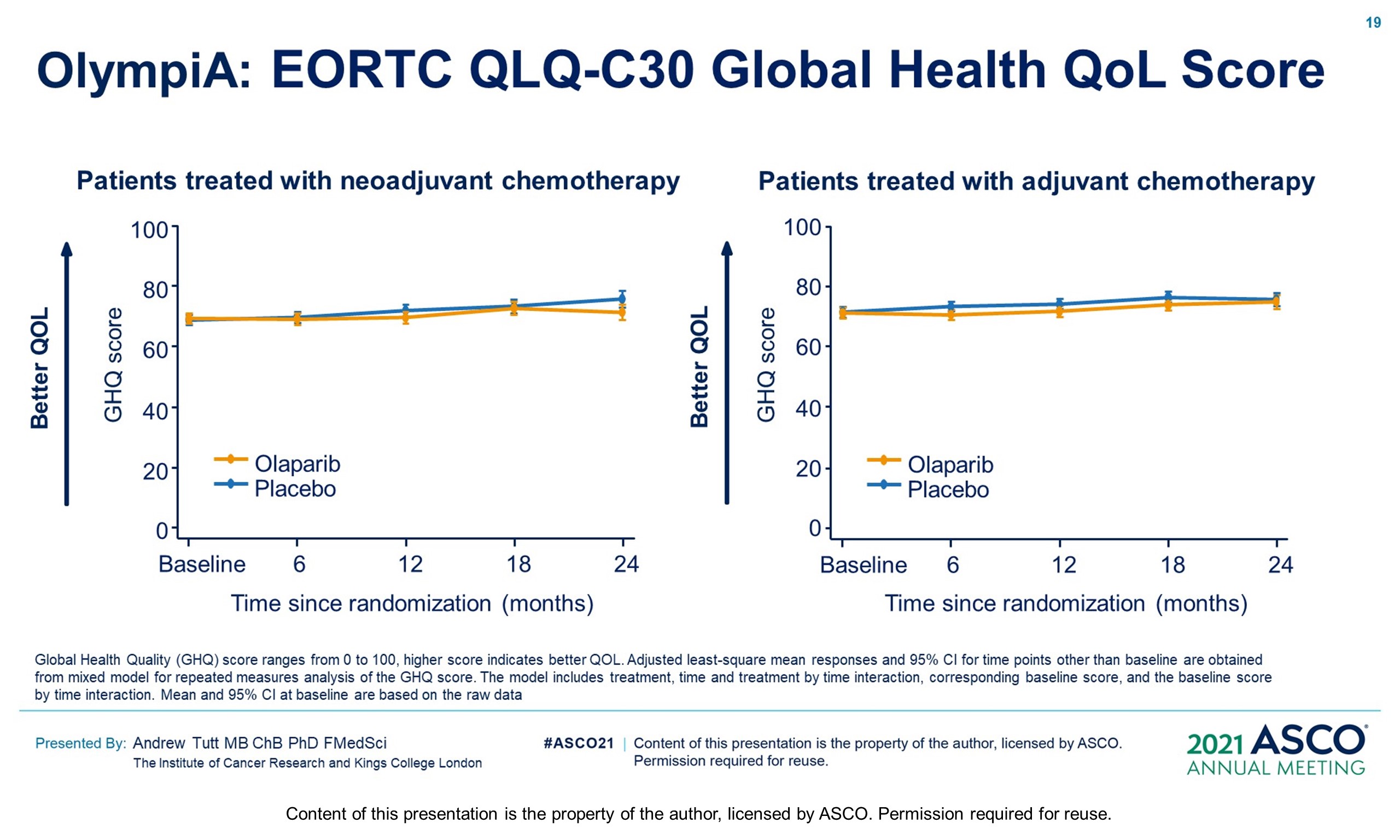
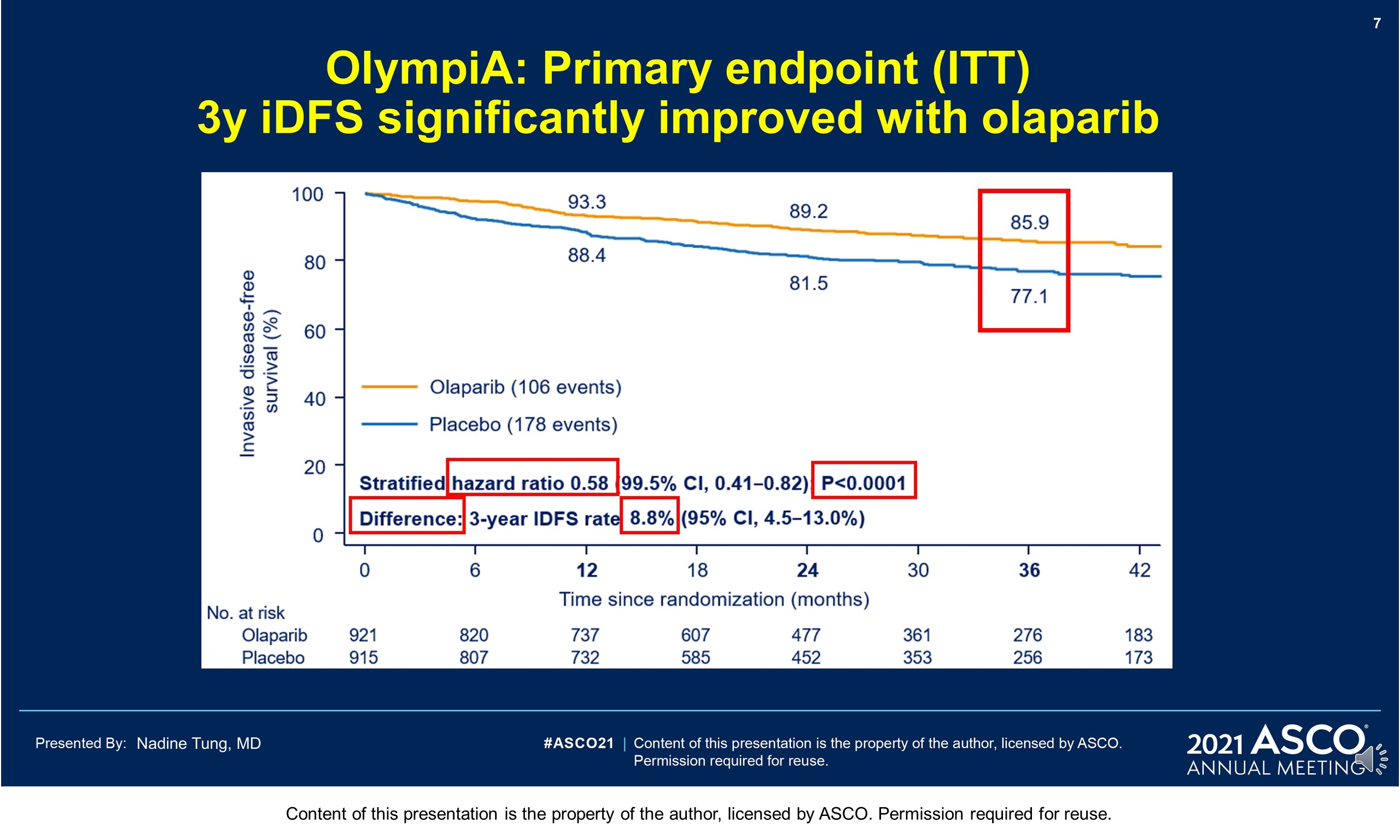
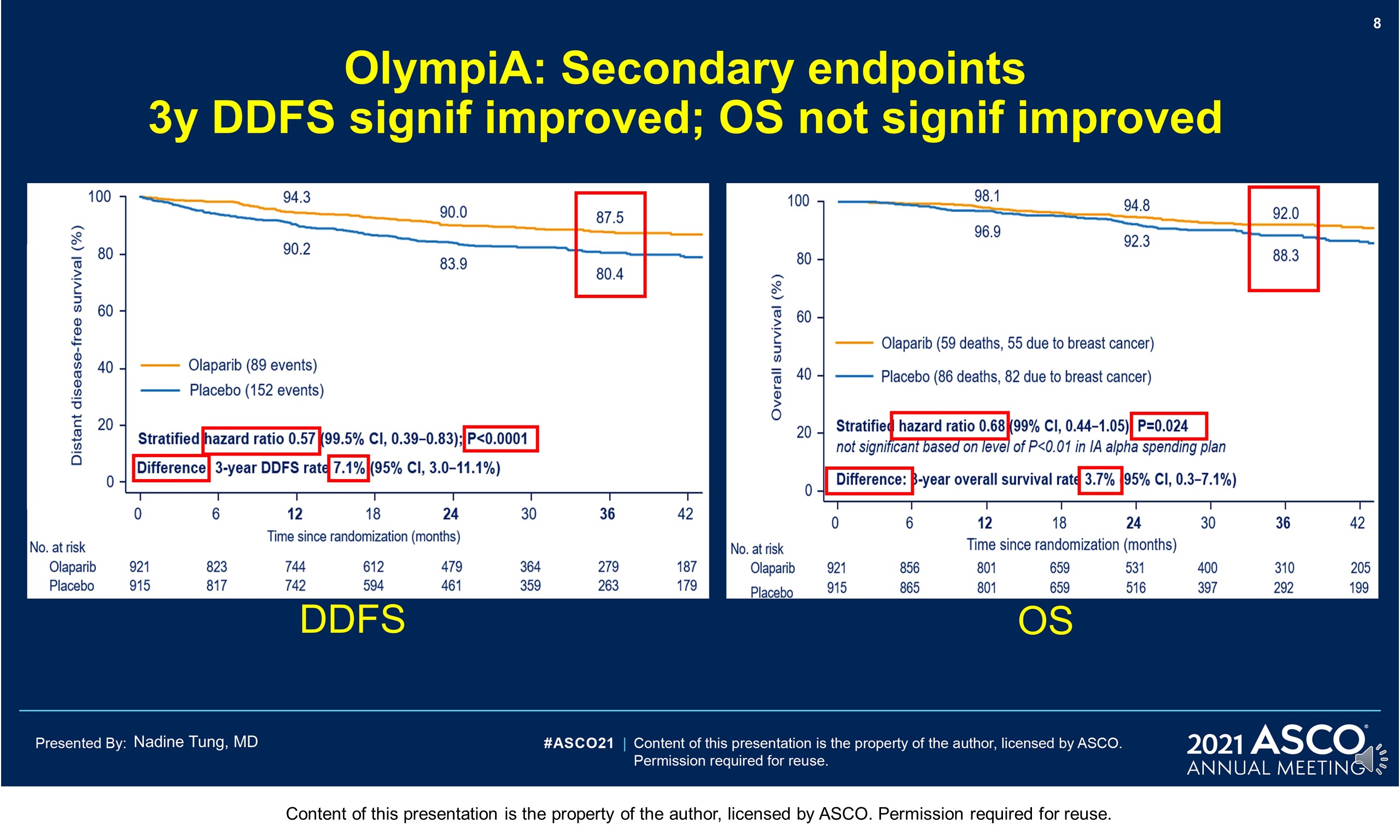
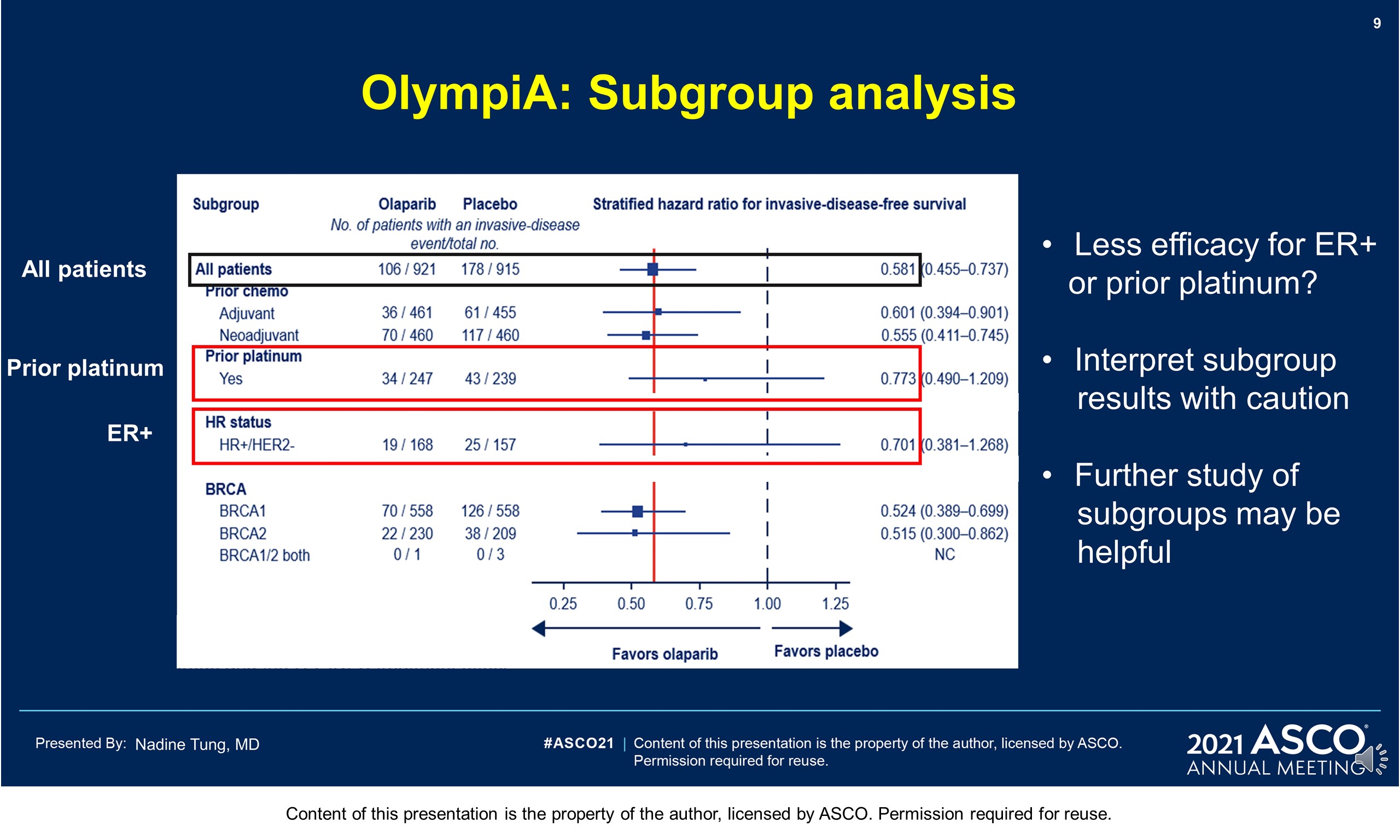
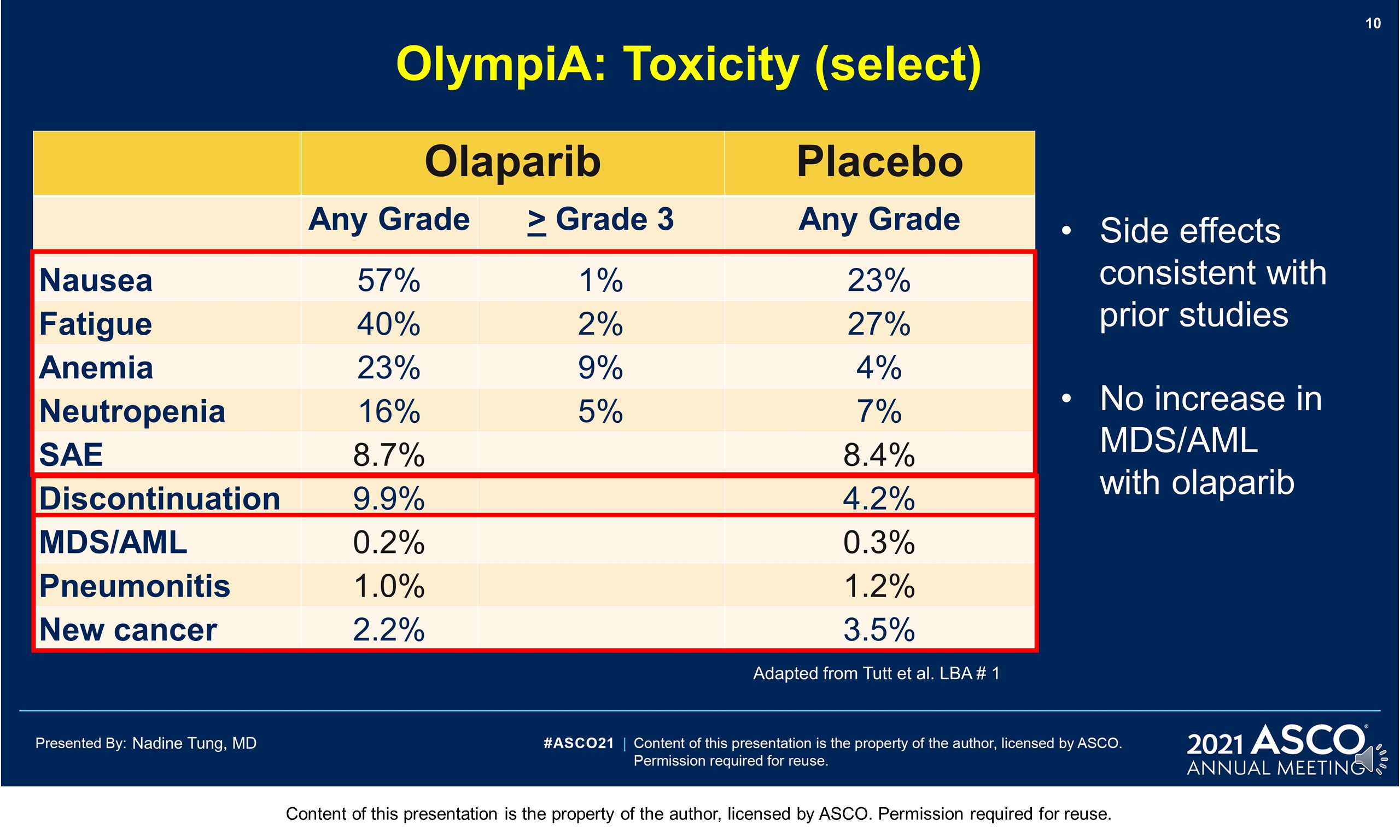
Results: 1836 pts were enrolled between 06/14–05/19; 49.9% had ACT, 50.1% NACT. Baseline demographics and tumor characteristics were balanced between arms. 82.2% had TNBC; 26.5% received a platinum agent. The IDMC recommended data unblinding as IA showed a significant benefit of OL vs PL for IDFS (hazard ratio [HR] 0.58; 99.5% CI 0.41, 0.82; P < 0.0001) at 2.5 yrs median follow-up. IDFS events occurred in 106/921 and 178/915 pts assigned to OL and PL, respectively. 3-yr IDFS was 85.9% vs 77.1% (diff. 8.8%; 95% CI 4.5%, 13.0%). DDFS was significantly improved with OL (HR 0.57; 99.5% CI 0.39, 0.83; P< 0.0001); 3-yr DDFS was 87.5% vs 80.4% (diff. 7.1%; 95% CI 3.0%, 11.1%). OS was greater for OL than PL but was not statistically significant at IA (HR 0.68; 99.0% CI 0.44, 1.05; P = 0.024); 3-yr OS% 92.0% vs 88.3% (diff. 3.7%; 95% CI 0.3%, 7.1%). Median intended OL exposure was 94.8%. AEs were consistent with the label. G3+ AEs in >1% of OL pts were; anemia (8.7%), neutropenia (4.8%), leukopenia (3.0%), fatigue (1.8%), and lymphocytopenia (1.2%). SAEs and AESI were not increased by OL, SAE 8.7% vs 8.4% and AESI 3.3% vs 5.1%, OL vs PL respectively.
Conclusions: Adjuvant OL following ACT or NACT significantly improved IDFS and DDFS with acceptable toxicity in pts with gBRCAm and high-risk HER2-negative EBC. Clinical trial information: NCT02032823
Discussion - LBA1
Disclaimer
ASCO® and American Society of Clinical Oncology® are registered trademarks of the American Society of Clinical Oncology, Inc. Used with permission.
This presentation does not necessarily represent a balanced view or full discussion of any given subject. The ideas and opinions expressed herein do not necessarily reflect those of ASCO. The authors, editors, and ASCO are not responsible for errors or omissions in translations. The mention of any company, product, service, or therapy in this collection of materials does not constitute an endorsement of any kind by ASCO. It is the responsibility of the treating physician or other health care provider, relying on independent experience and knowledge of the patient, to determine drug dosages and the best treatment for the patient. Viewers are advised to check the appropriate medical literature and the product information currently provided by the manufacturer of each drug to be administered to verify, among other matters, the dosage, method, and duration of administration, or contraindications. Viewers are also encouraged to contact the manufacturer with questions about the features or limitations of any products. ASCO assumes no responsibility for any injury or damage to persons or property arising out of or related to any use of the material contained in this publication or to any errors or omissions.
Site provided by Springer Healthcare Limited. Part of the Springer Nature Group.