CheckMate 067: 6.5-year outcomes in patients (pts) with advanced melanoma
Authors: Jedd D. Wolchok, Vanna Chiarion-Sileni, Rene Gonzalez, Jean-Jacques Grob, Piotr Rutkowski, Christopher D. Lao, Charles Lance Cowey, Dirk Schadendorf, John Wagstaff, Reinhard Dummer, Pier Francesco Ferrucci, Michael Smylie, Marcus O. Butler, Andrew Graham Hill, Ivan Marquez-Rodas, John B. A. G. Haanen, Tuba Bas, Wim van Dijck, James Larkin, F. Stephen Hodi; Medical Oncology, Memorial Sloan Kettering Cancer Center, and Weill Cornell Medical College, New York, NY; Oncology Institute of Veneto IRCCS, Padua, Italy; University of Colorado Cancer Center, Denver, CO; Aix-Marseille University, APHM, Hôpital Timone, Marseille, France; Maria Sklodowska-Curie Institute-Oncology Center, Warsaw, Poland; Michigan Medicine, Rogel Cancer Center, University of Michigan, Ann Arbor, MI; Texas Oncology-Baylor Charles A. Sammons Cancer Center, Dallas, TX; Department of Dermatology, University of Essen, Essen, and German Cancer Consortium, Heidelberg, Germany; The College of Medicine, Swansea University, Swansea, United Kingdom; Skin Cancer Center, University Hospital of Zürich, Zürich, Switzerland; European Institute of Oncology–IRCCS, Milan, Italy; Cross Cancer Institute, Edmonton, AB, Canada; Princess Margaret Cancer Centre, Toronto, ON, Canada; Tasman Oncology Research, Southport, QLD, Australia; Medical Oncology, General University Hospital Gregorio Marañón & CIBERONC, Madrid, Spain; Netherlands Cancer Institute, Amsterdam, Netherlands; Bristol Myers Squibb, Princeton, NJ; Bristol-Myers Squibb, Princeton, NJ; The Royal Marsden Hospital NHS Foundation Trust, London, United Kingdom; Dana-Farber Cancer Institute, Boston, MA
Research Funding: Bristol Myers Squibb
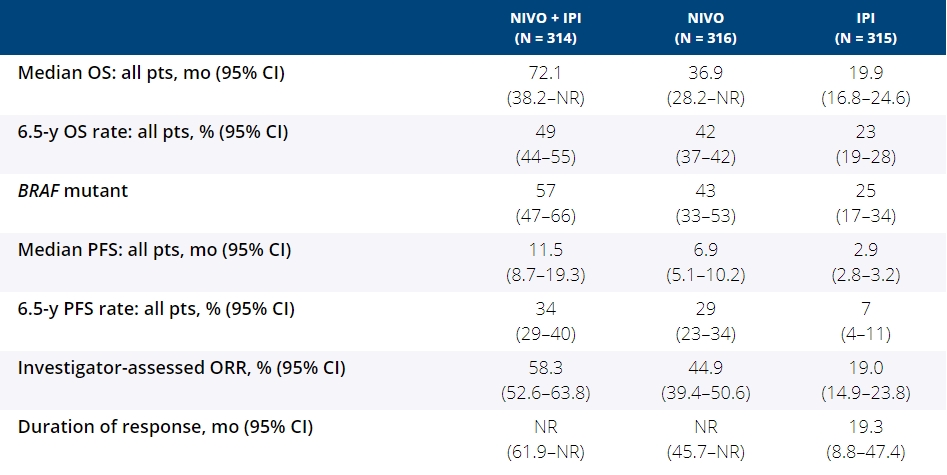
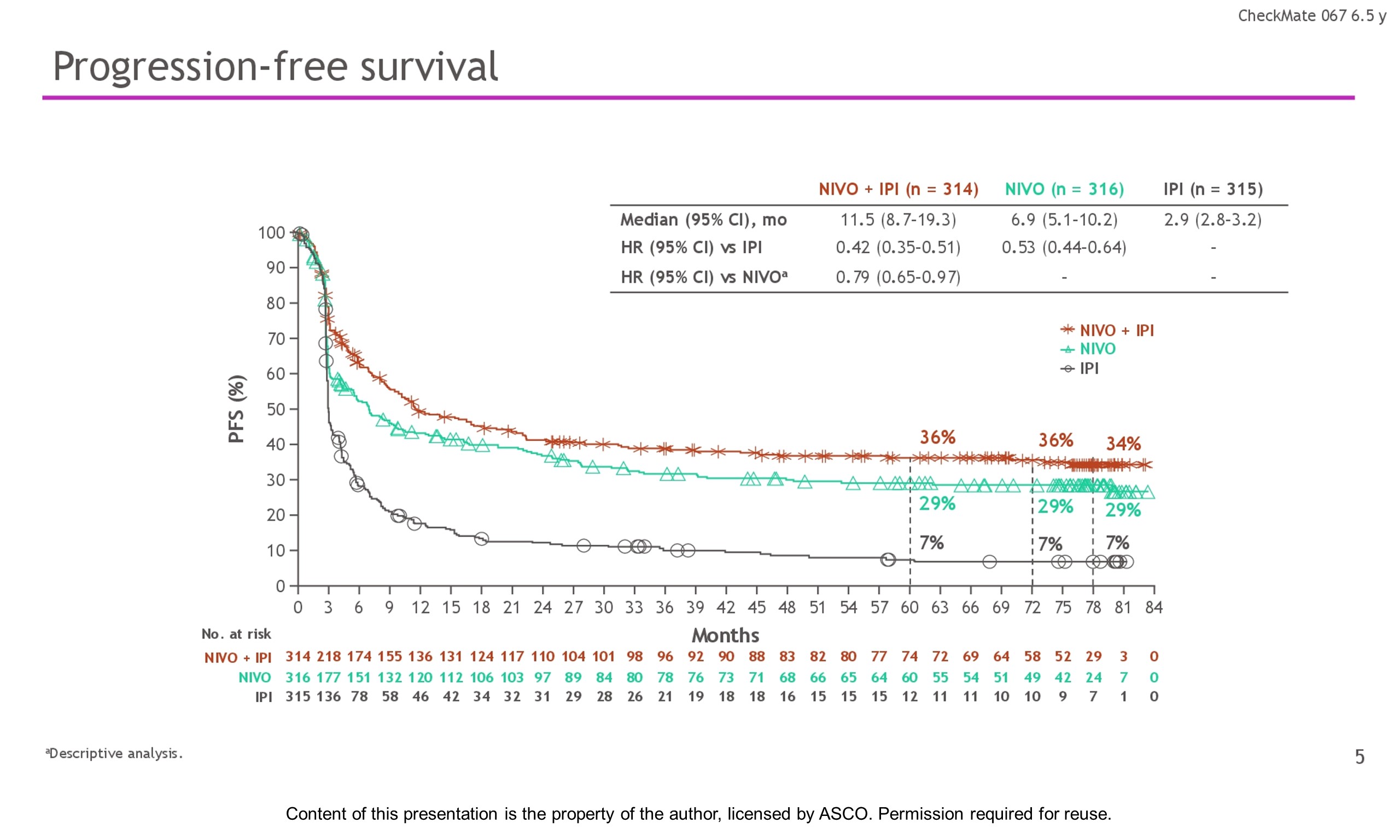
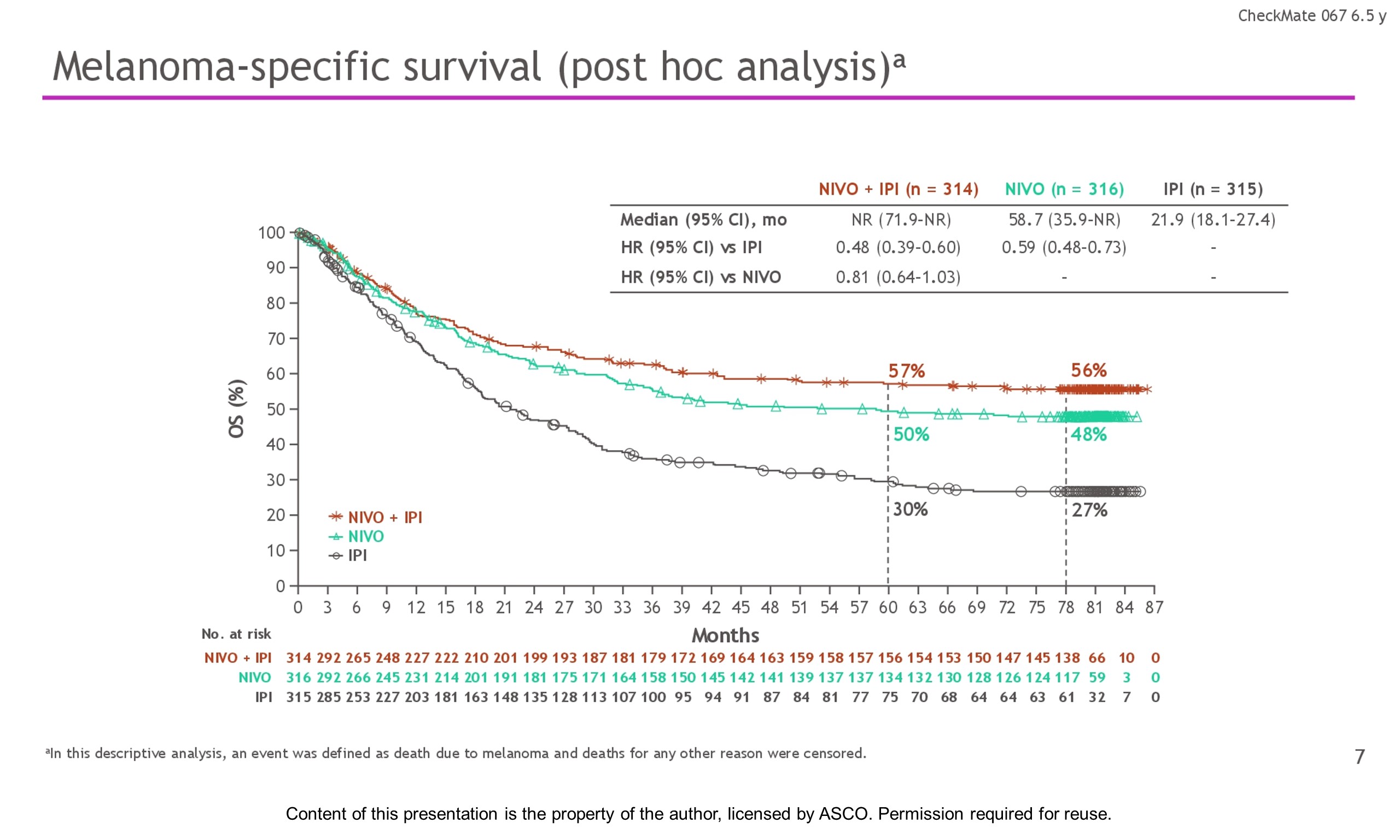
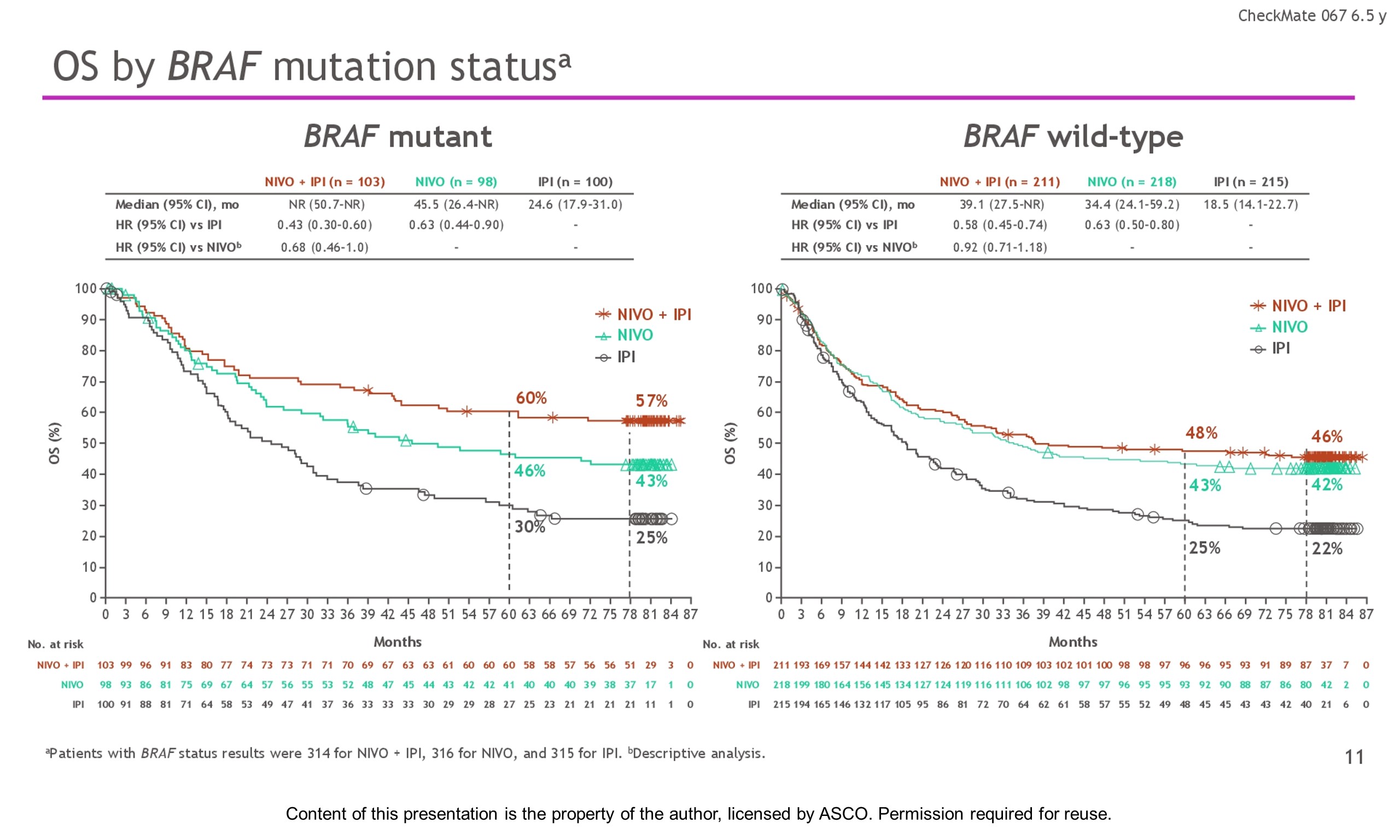
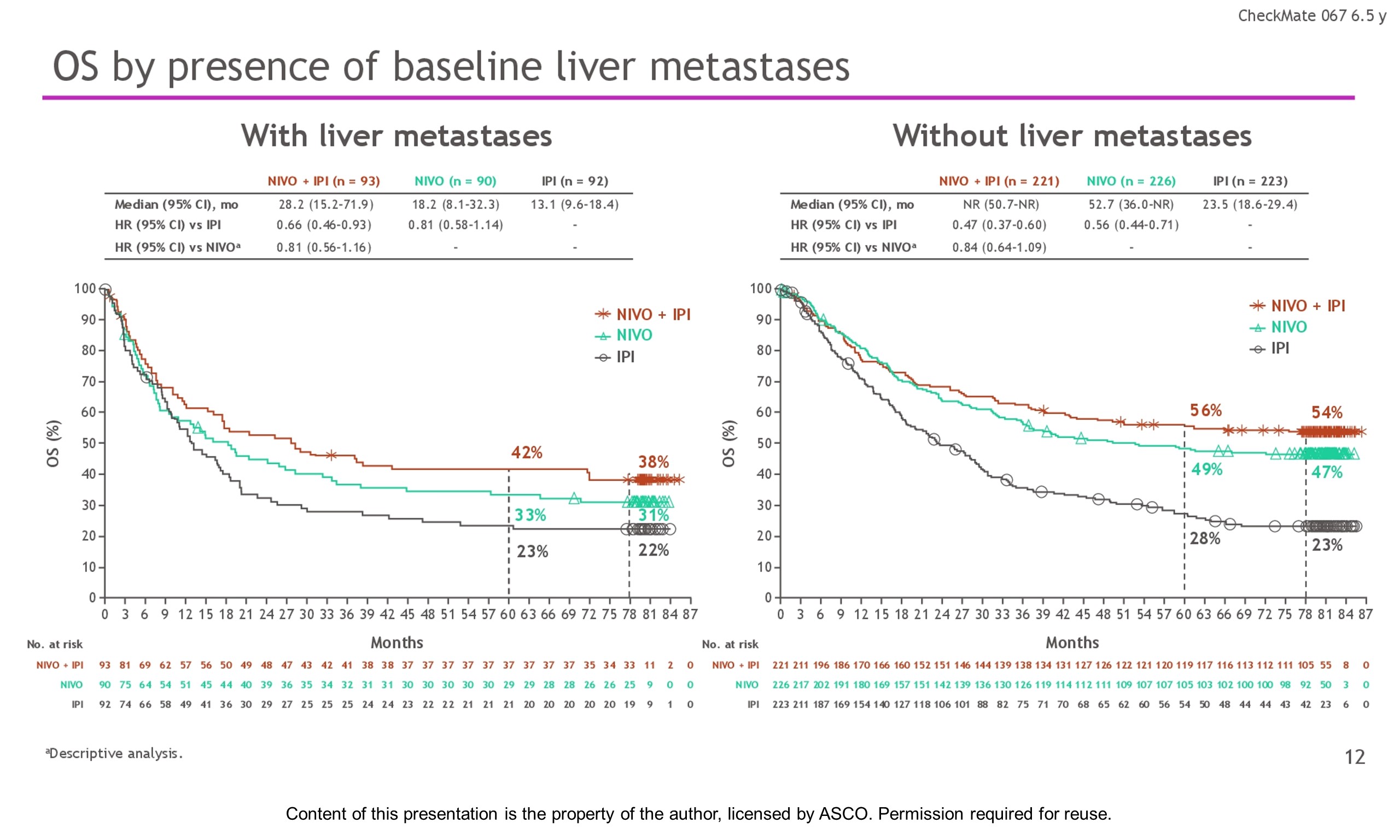
Background: In the phase 3 CheckMate 067 trial, a durable and sustained clinical benefit was achieved with nivolumab (NIVO) + ipilimumab (IPI) and NIVO alone vs IPI at 5-y of follow-up (overall survival [OS] and progression-free survival [PFS] rates: 52%, 44%, 26% and 36%, 29%, 8%, respectively). Here we report 6.5-y efficacy and safety outcomes.
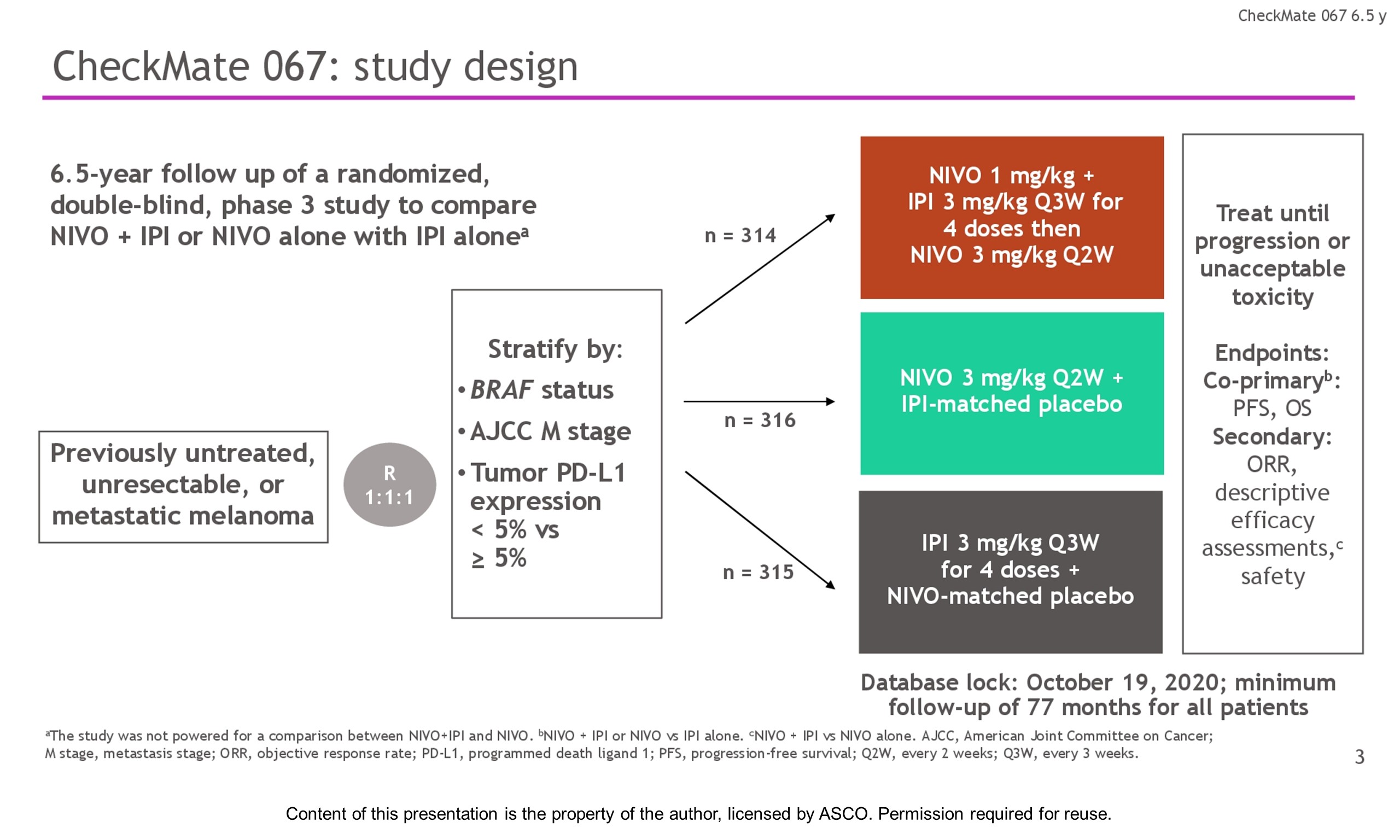
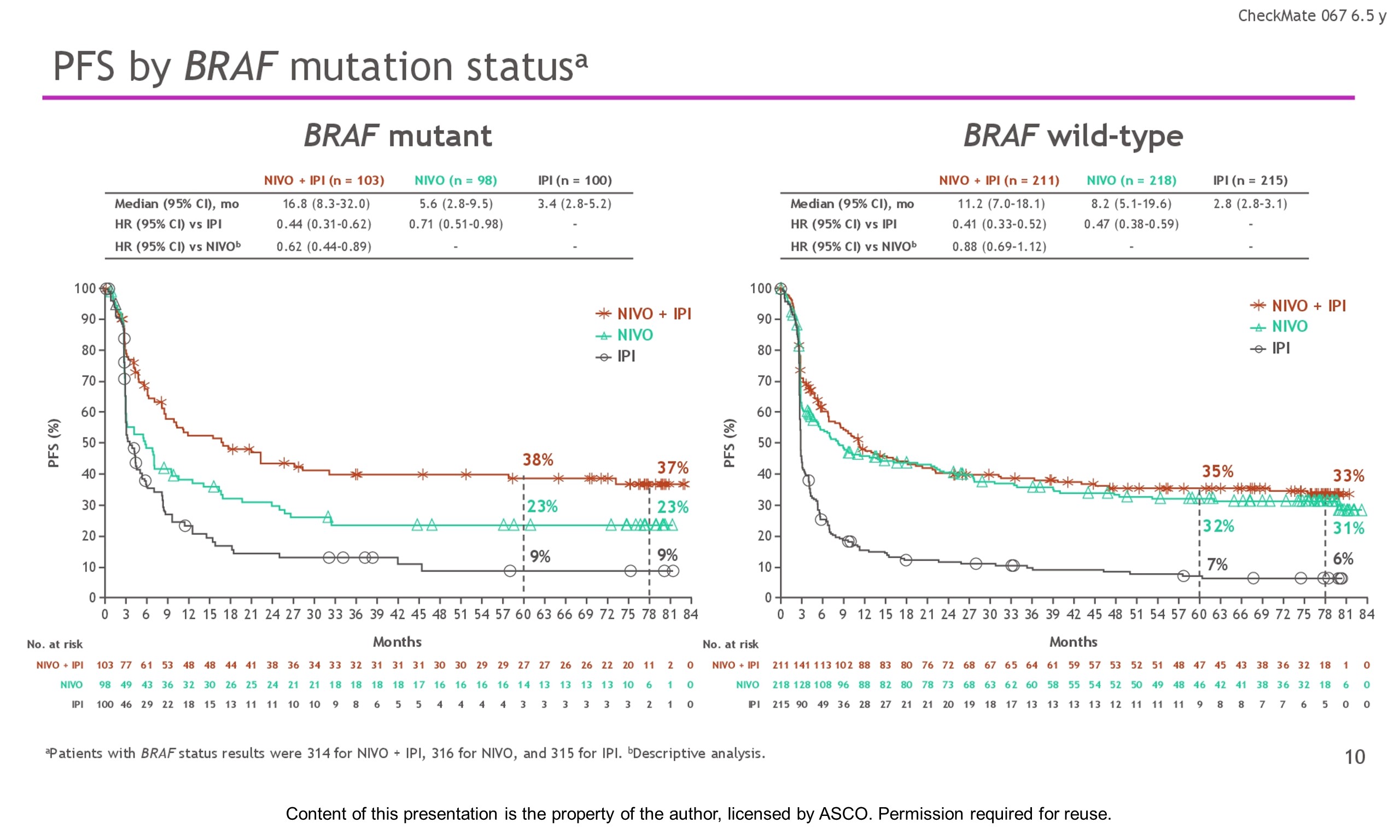
Methods: Eligible pts with previously untreated unresectable stage III or IV melanoma were randomly assigned in a 1:1:1 ratio and stratified by PD-L1 status, BRAF mutation status, and metastasis stage. Pts received NIVO 1 mg/kg + IPI 3 mg/kg for 4 doses Q3W followed by NIVO 3 mg/kg Q2W (n = 314), NIVO 3 mg/kg Q2W + placebo (n = 316), or IPI 3 mg/kg Q3W for 4 doses + placebo (n = 315) until progression or unacceptable toxicity. Co-primary endpoints were PFS and OS with NIVO + IPI or NIVO vs IPI. Secondary endpoints included objective response rate (ORR), descriptive efficacy assessments of NIVO + IPI vs NIVO alone, and safety.
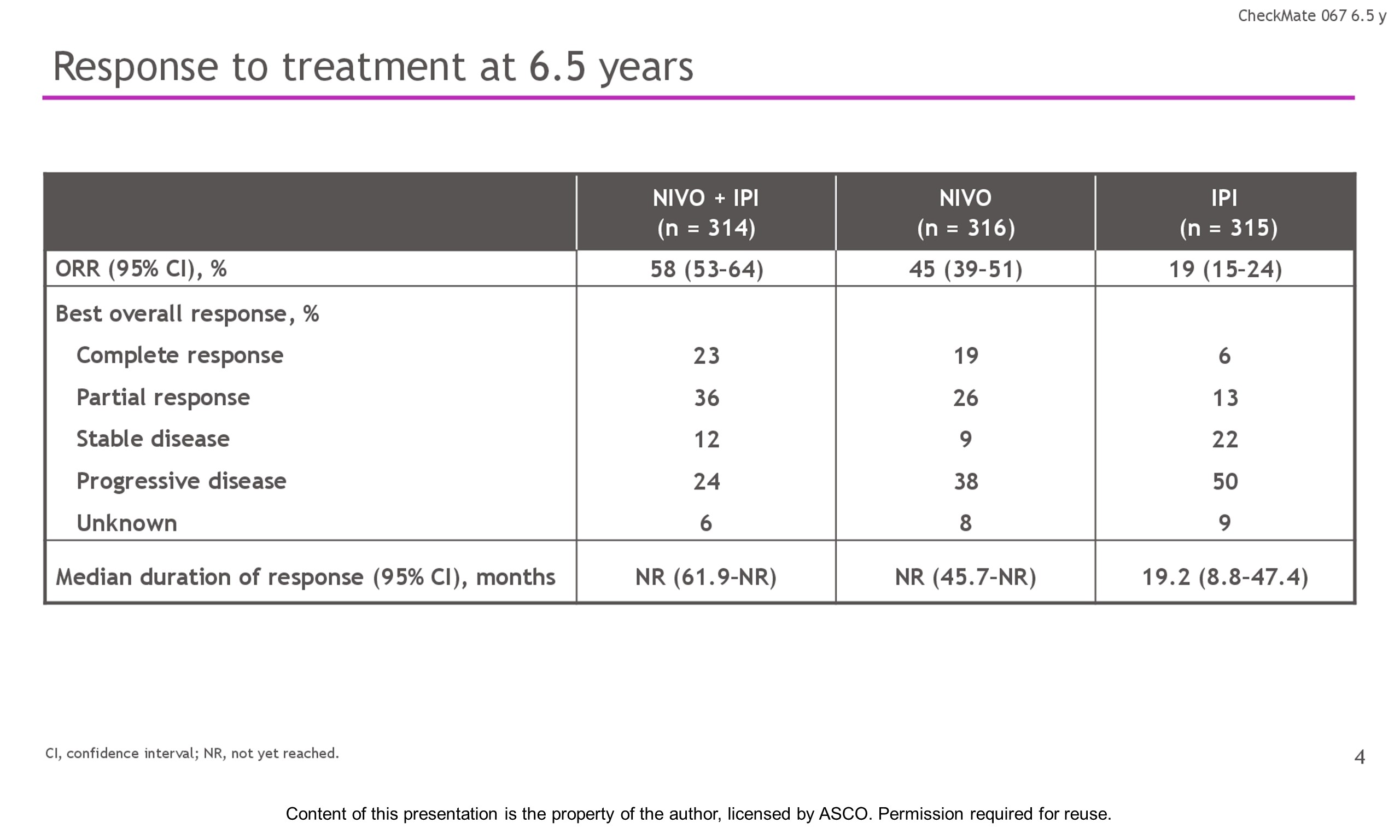
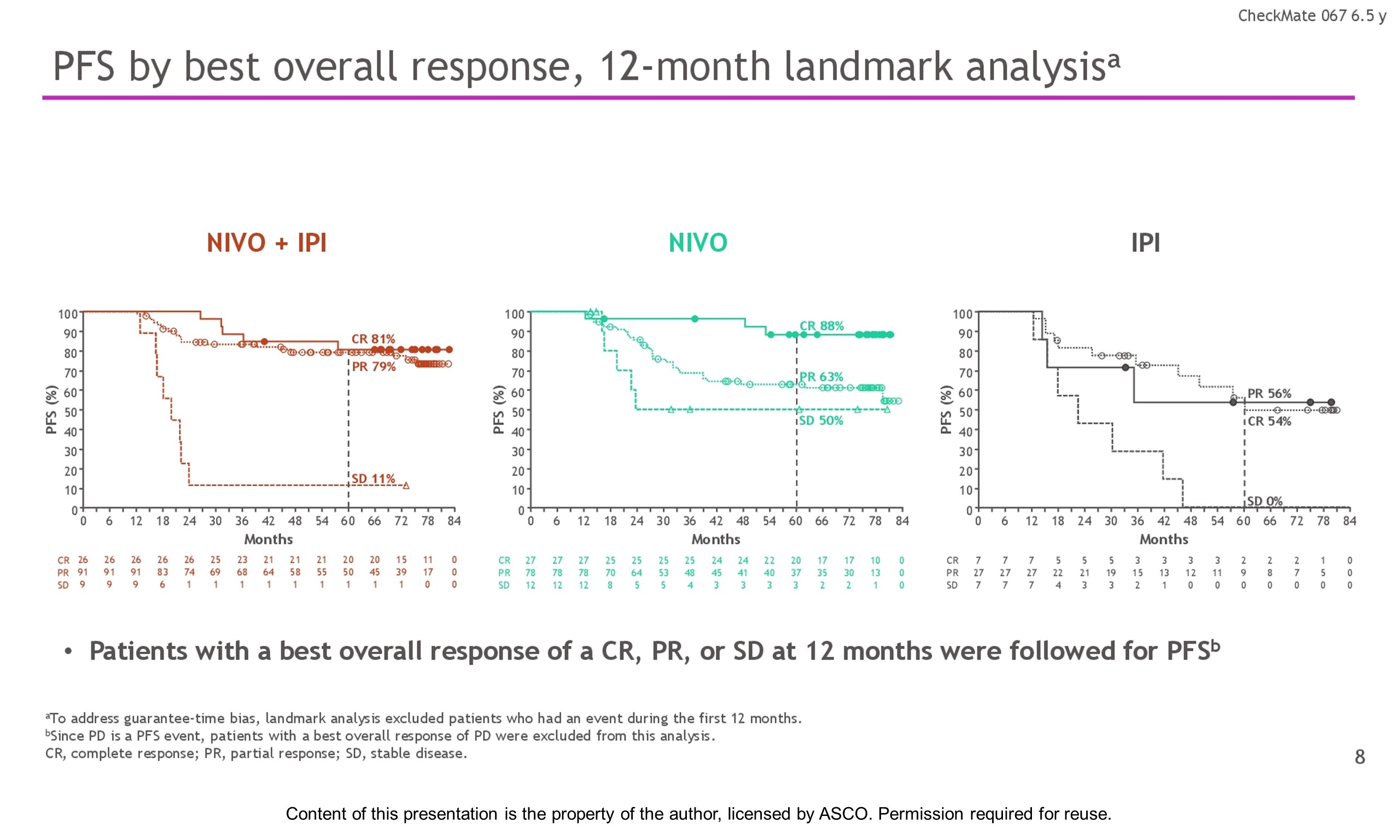
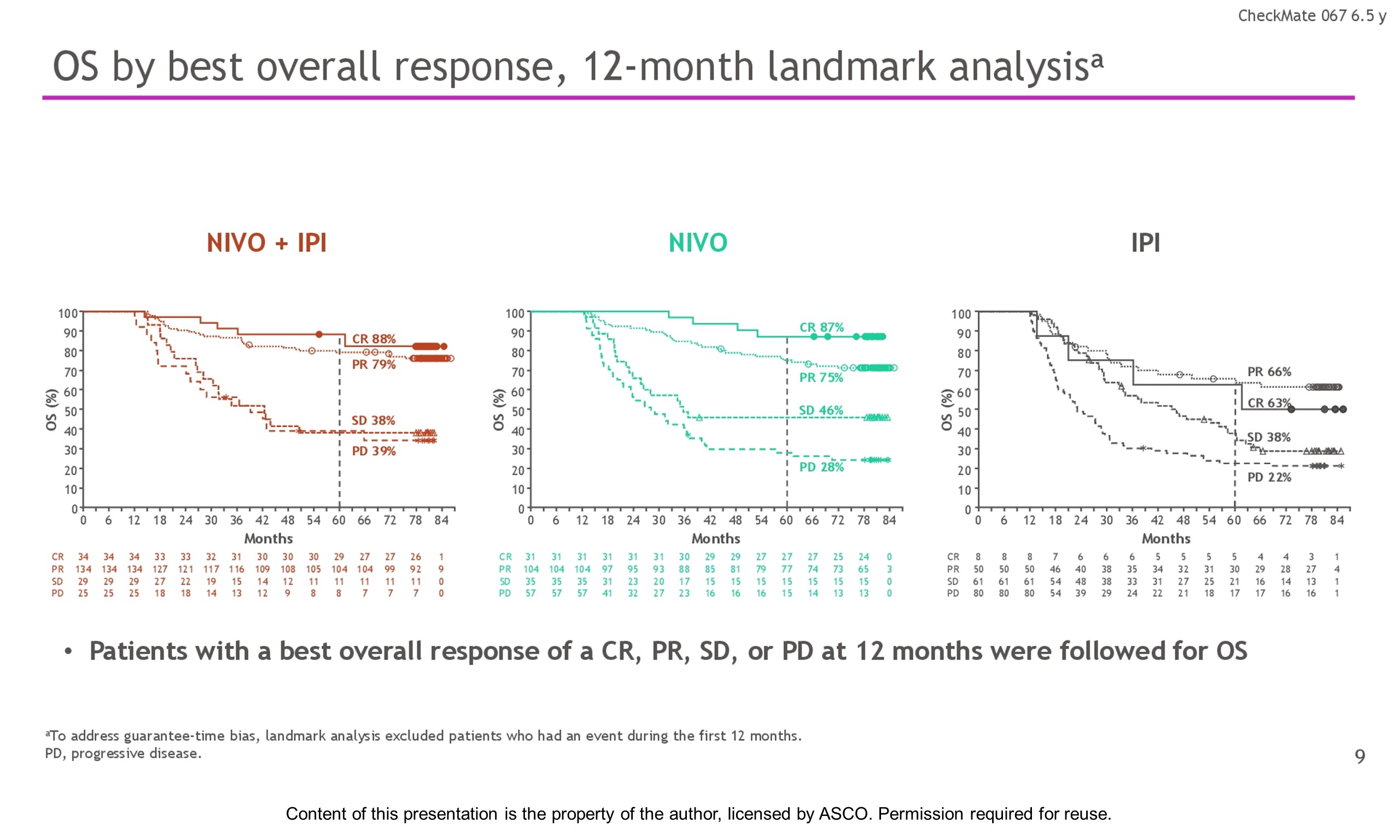
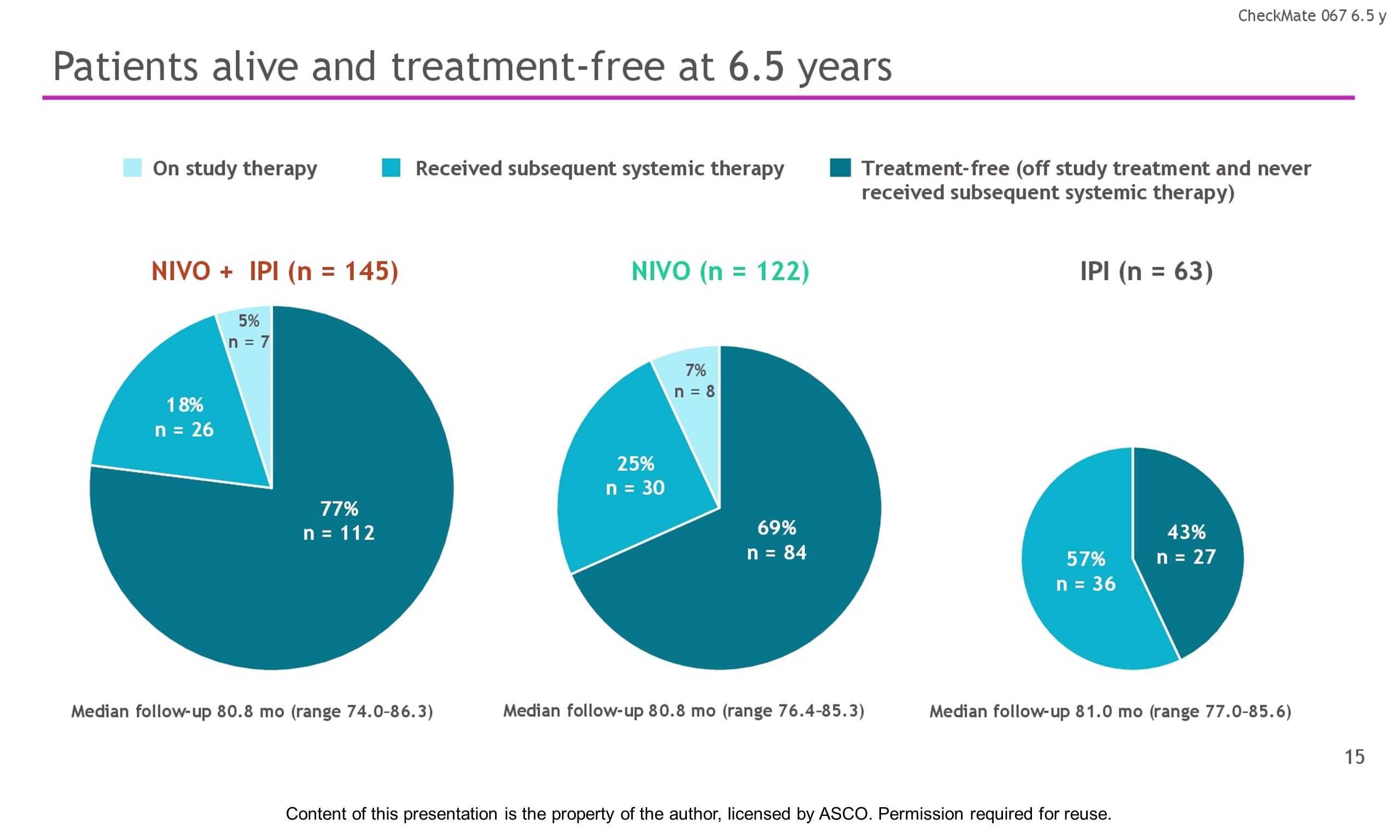
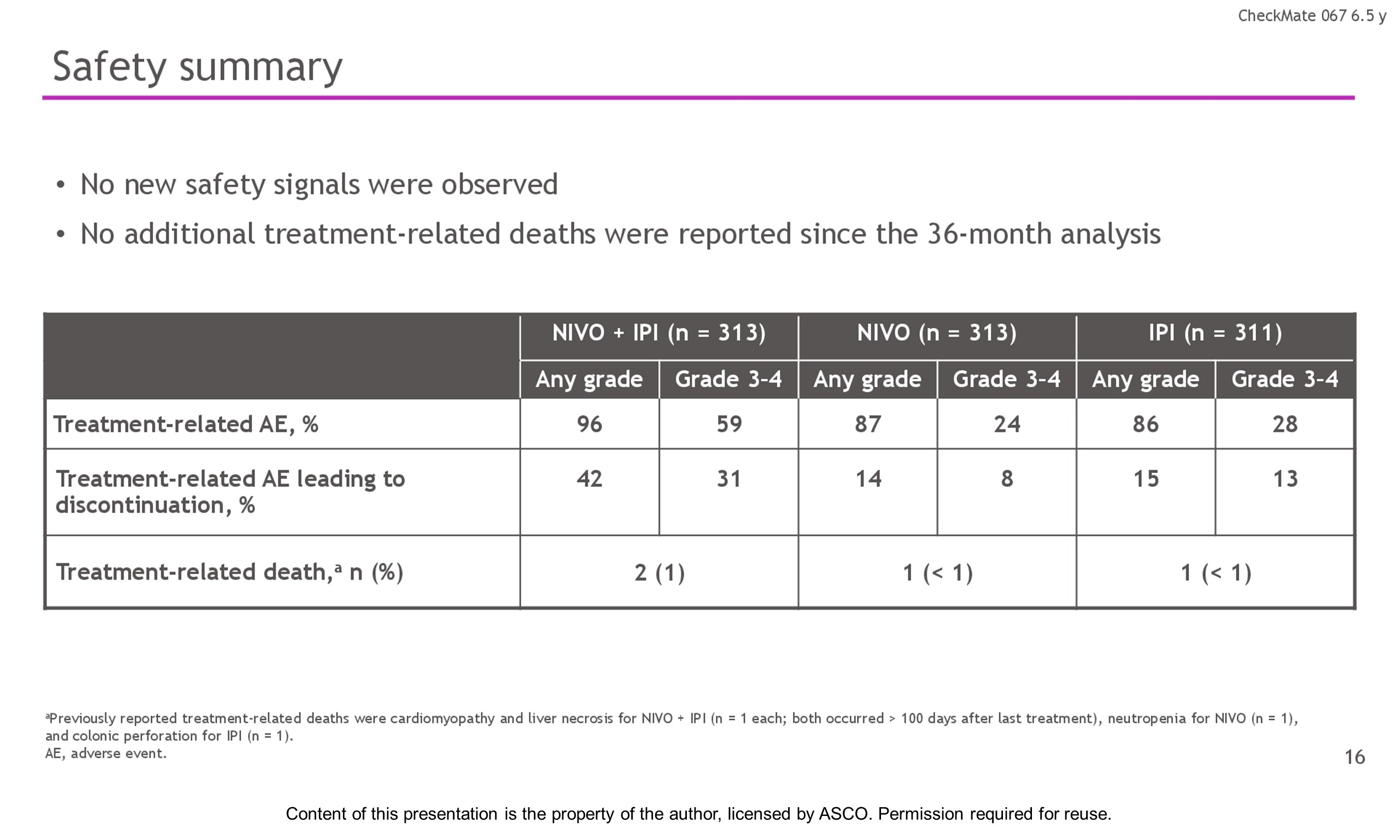
Results: With a minimum follow-up of 6.5 y, median OS was 72.1 mo with NIVO + IPI, 36.9 mo with NIVO, and 19.9 mo with IPI (table). Median time from randomization to subsequent systemic therapy was not reached (NR; 95% CI, 59.6–NR) with NIVO + IPI, 25.2 mo (95% CI, 16.0–43.2) with NIVO, and 8.0 mo (95% CI, 6.5–8.7) with IPI; 36%, 49%, and 66% of pts, respectively, received any subsequent systemic therapy. Median treatment-free interval (which excluded pts who discontinued follow-up prior to initiation of subsequent systemic therapy) was 27.6 mo (range, 0–83.0), 2.3 mo (range, 0.2–81.6), and 1.9 mo (range, 0.1–81.9) with NIVO + IPI, NIVO, and IPI, respectively. Of the pts alive and in follow-up, 112/138 (81%; NIVO + IPI), 84/114 (74%; NIVO), and 27/63 (43%; IPI) were off treatment and never received subsequent systemic therapy; 7, 8, and 0 pts, respectively, were still on treatment. Grade 3/4 treatment-related adverse events were reported in 59% of NIVO + IPI-treated pts, 24% of NIVO-treated pts, and 28% of IPI-treated pts. Since the 5-y analysis, no new safety signals were observed and no additional treatment-related deaths occurred.
Conclusions: This 6.5-y analysis represents the longest follow-up from a phase 3 melanoma trial in the modern checkpoint inhibitor combination therapy and targeted therapy era. The results show durable improved outcomes with NIVO + IPI and NIVO vs IPI in pts with advanced melanoma. We observed improvement in OS, PFS, and ORR with NIVO + IPI over NIVO alone. Clinical trial information: NCT01844505
7356-EP-2100002 Junio 2021
Disclaimer
The information contained in this website is for healthcare professionals.
La información contenida en esta web está dirigida a profesionales sanitarios.
As informações contidas neste site são destinadas aos profissionais de saúde.
ASCO® and American Society of Clinical Oncology® are registered trademarks of the American Society of Clinical Oncology, Inc. Used with permission.
This presentation does not necessarily represent a balanced view or full discussion of any given subject. The ideas and opinions expressed herein do not necessarily reflect those of ASCO. The authors, editors, and ASCO are not responsible for errors or omissions in translations. The mention of any company, product, service, or therapy in this collection of materials does not constitute an endorsement of any kind by ASCO. It is the responsibility of the treating physician or other health care provider, relying on independent experience and knowledge of the patient, to determine drug dosages and the best treatment for the patient. Viewers are advised to check the appropriate medical literature and the product information currently provided by the manufacturer of each drug to be administered to verify, among other matters, the dosage, method, and duration of administration, or contraindications. Viewers are also encouraged to contact the manufacturer with questions about the features or limitations of any products. ASCO assumes no responsibility for any injury or damage to persons or property arising out of or related to any use of the material contained in this publication or to any errors or omissions.
Site provided by Springer Healthcare Limited. Part of the Springer Nature Group.