LBA2 – JUPITER-02: Randomized, double-blind, phase III study of toripalimab or placebo plus gemcitabine and cisplatin as first-line treatment for recurrent or metastatic nasopharyngeal carcinoma (NPC)
Authors: Rui-hua Xu, Hai-Qiang Mai, Qiu-Yan Chen, Dongping Chen, Chaosu Hu, Kunyu Yang, Jiyu Wen, Jin-Gao Li, Yingrui Shi, Feng Jin, Ruilian Xu, Jianji Pan, Shenhong Qu, Ping Li, Chunhong Hu, Yi-Chun Liu, Yi Jiang, Xia He, Hung-Ming Wang, Wan-Teck Lim, Coherus Biosciences and Shanghai Junshi Biosciences; Sun Yat-sen University Cancer Center, State Key Laboratory of Oncology in South China, Collaborative Innovation Center for Cancer Medicine, Guangzhou, China; Department of Nasopharyngeal Carcinoma, Sun Yat-sen University Cancer Center, Guangzhou, China; The Affiliated Cancer Hospital of Guangzhou Medical University, Guangzhou, China; Fudan University Shanghai Cancer Center, Shanghai, China; Cancer Center, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China; Affiliated Hospital of Guangdong Medical University, Zhanjiang, China; Jiangxi Cancer Hospital, Nanchang, China; Hunan Cancer Hospital and the Affiliated Cancer Hospital of Xiangya School of Medicine, Changsha, China; Guizhou Cancer Hospital, Guiyang, China; Shenzhen People’s Hospital, Shenzhen, China; Fujian Cancer Hospital, Fuzhou, China; Guangxi Zhuang Autonomous Region People’s Hospital, Nanning, China; West China Hospital, Sichuan University, Chengdu, China; The Second Xiangya Hospital of Central South University, Changsha, China; Department of Radiation Oncology, Taichung Veterans General Hospital, Taichung, Taiwan; Cancer Hospital of Shantou University Medical College, Shantou, China; Jiang-Su Cancer Hospital, Nanjing, Jiangsu, China; Chang Gung Memorial Hospital, Taipei, Taiwan; National Cancer Centre Singapore, Singapore, Singapore
Research Funding: Shanghai Junshi Biosciences
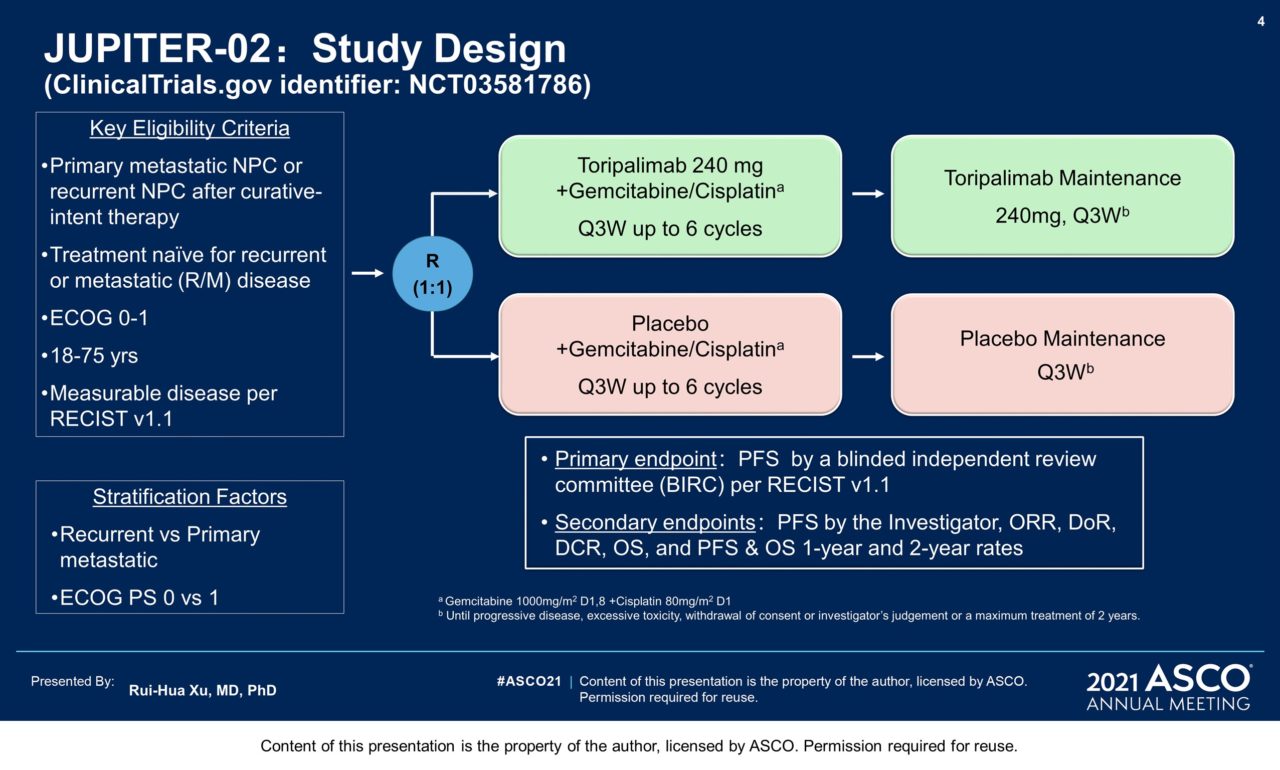
Background: Gemcitabine-cisplatin (GP) chemotherapy is the standard 1st line treatment for locally advanced, recurrent or metastatic (r/m) NPC. Toripalimab, a humanized IgG4K monoclonal antibody specific for PD-1, provided durable responses in patients (pts) with r/m NPC as monotherapy in the ≥2nd line setting (POLARIS-02 study). The results of JUPITER-02, a randomized, placebo-controlled, double-blinded Phase III trial of toripalimab in combination with GP chemotherapy as first-line treatment for r/m NPC are summarized.
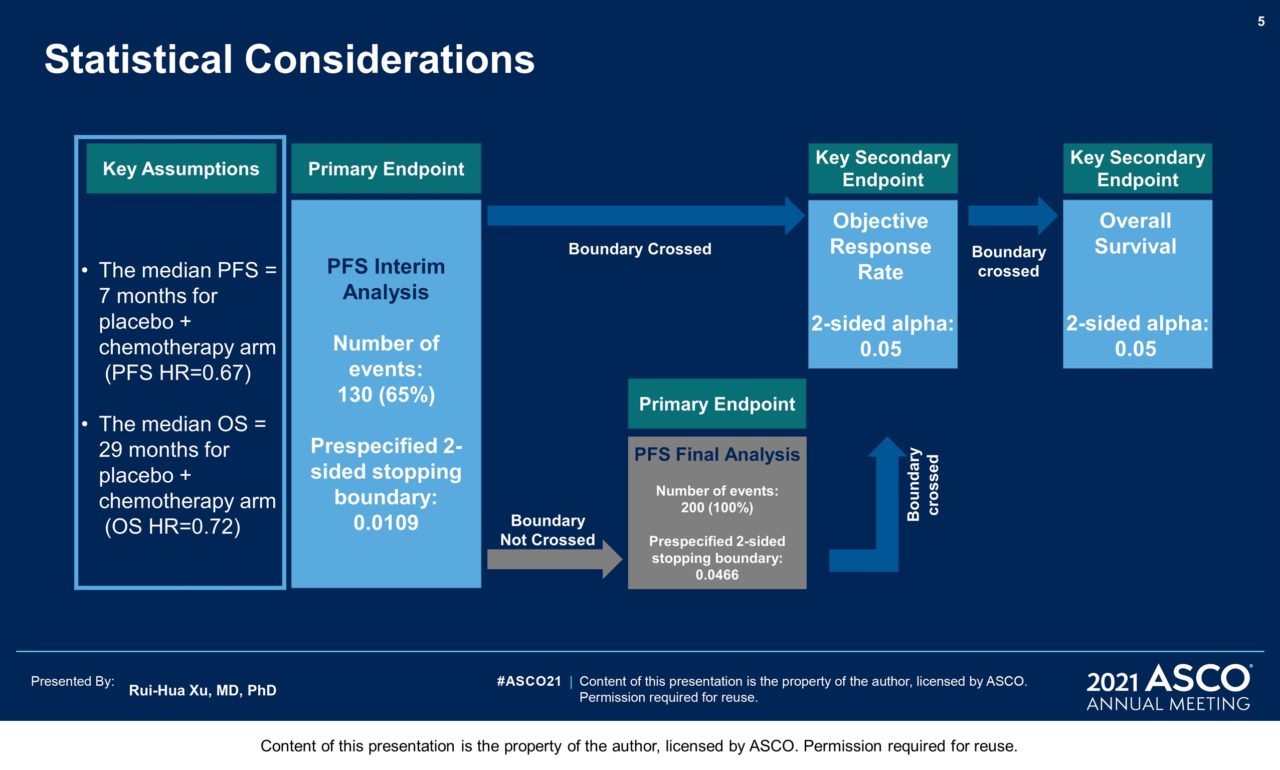
Methods: Pts with advanced NPC with no prior chemotherapy in the r/m setting were randomized (1:1) to receive toripalimab 240 mg or placebo d1 in combination with gemcitabine 1000 mg/m2 d1, d8 and cisplatin 80 mg/m2 d1 every 3 weeks (Q3W) for up to 6 cycles, followed by monotherapy with toripalimab or placebo Q3W until disease progression, intolerable toxicity, or completion of 2 years of treatment. Stratification factors were ECOG PS (0 vs. 1) and extent of disease (recurrent vs. primary metastatic) at enrollment. Progression-free survival (PFS) and response were assessed by independent review committee (IRC) per RECIST v1.1. The primary endpoint was PFS by IRC in the ITT population. Secondary end points included ORR, DOR and OS. There was one prespecified interim analysis of PFS at 130 PFS events with a planned final analysis at 200 PFS events.
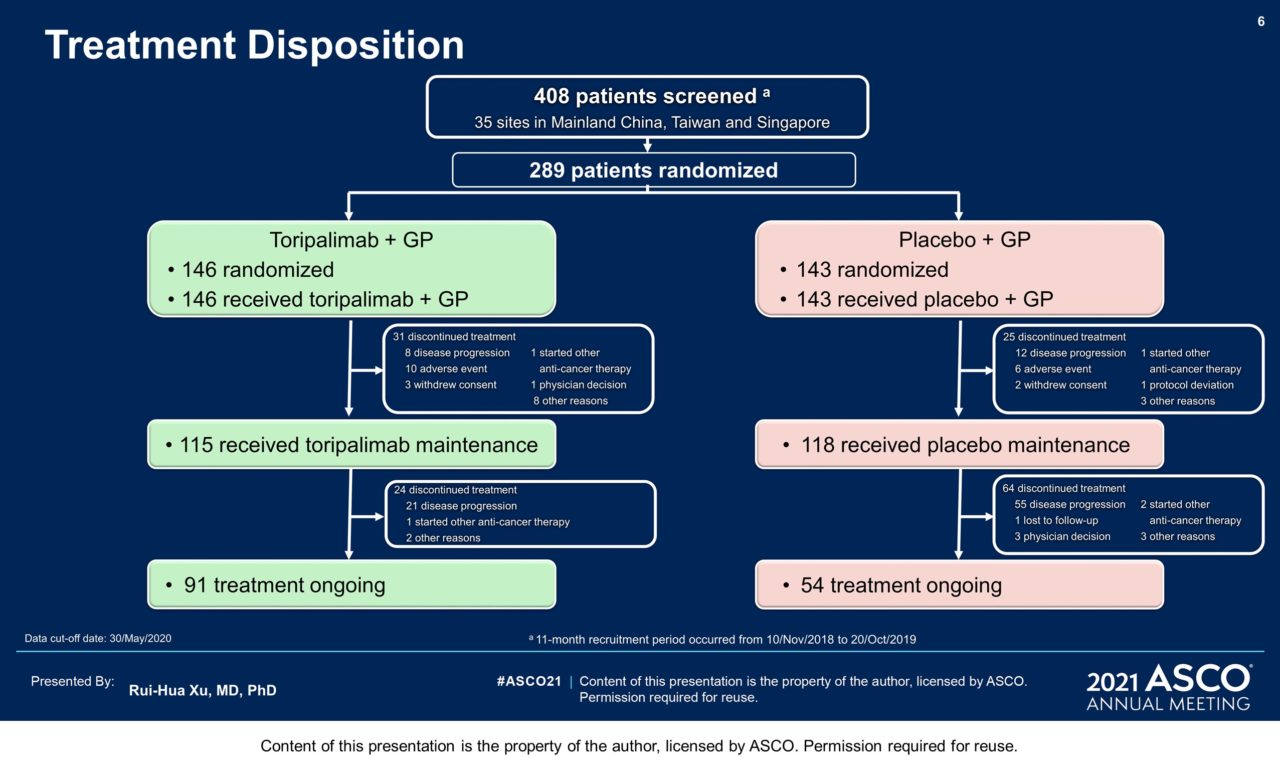
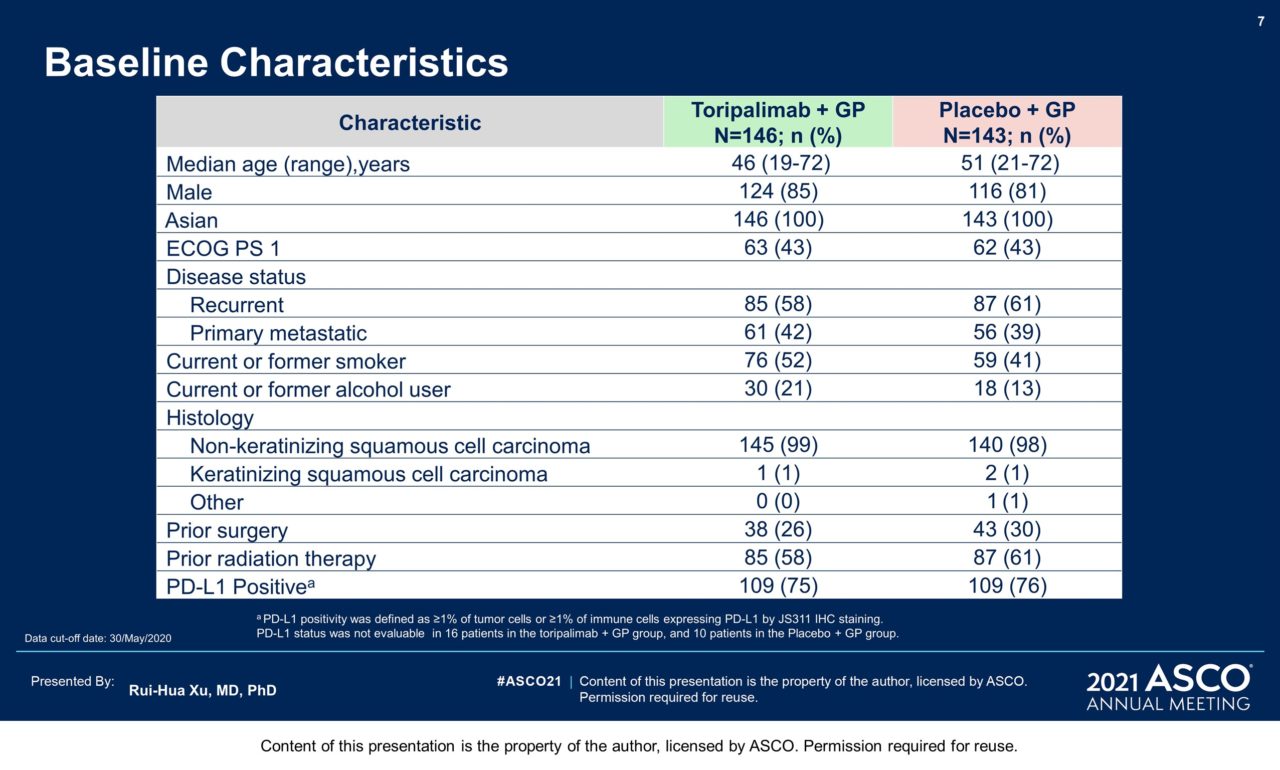
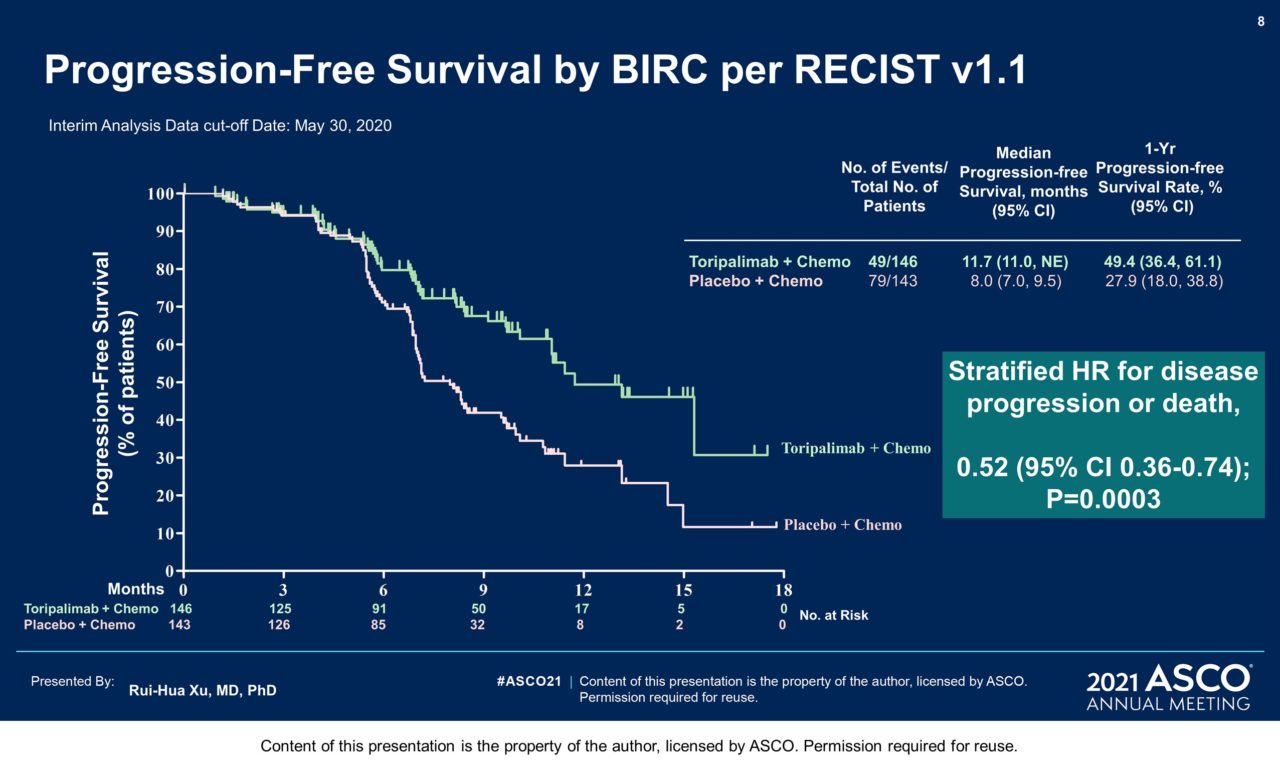
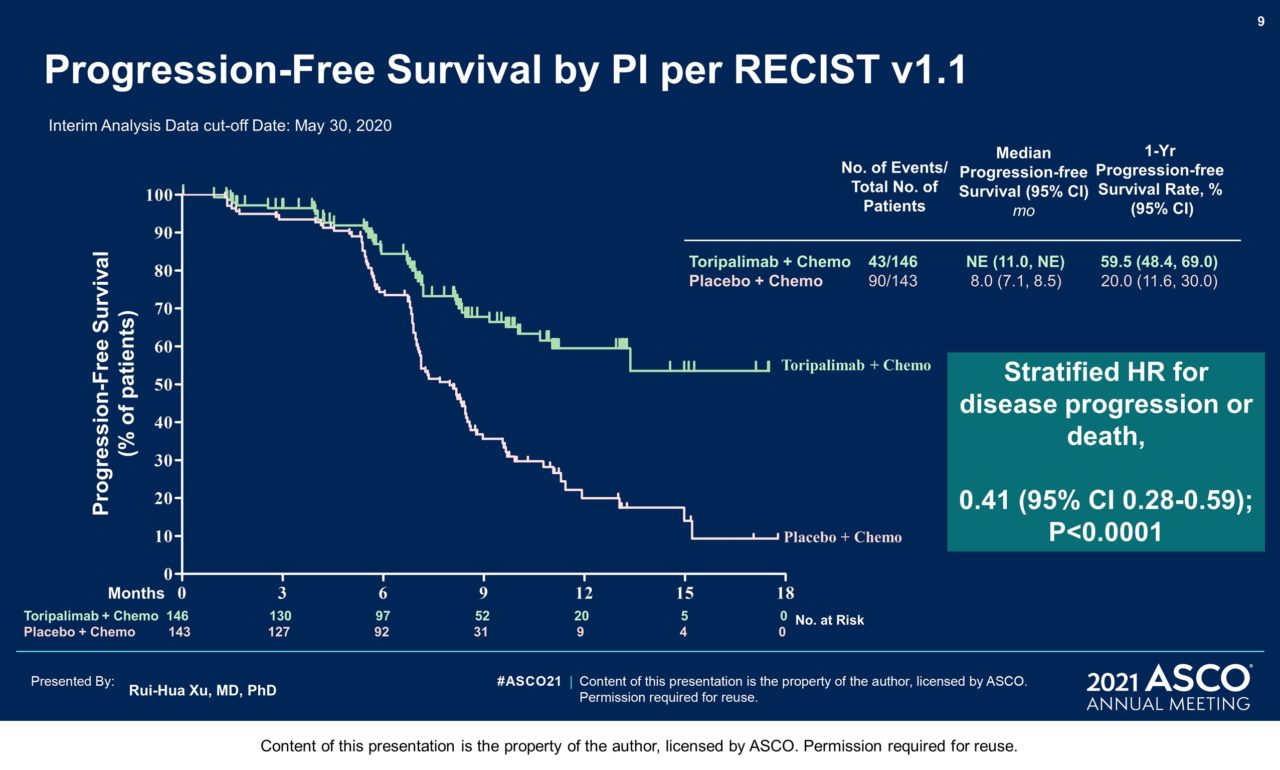
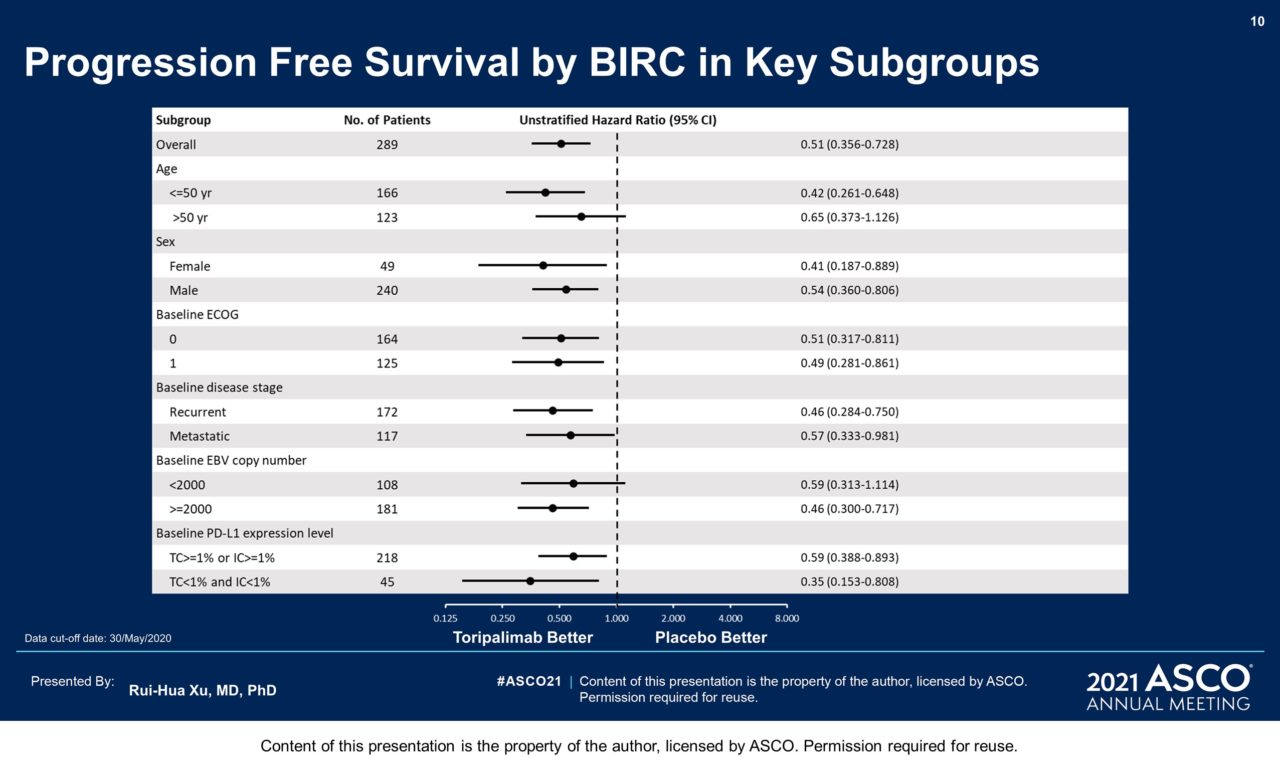
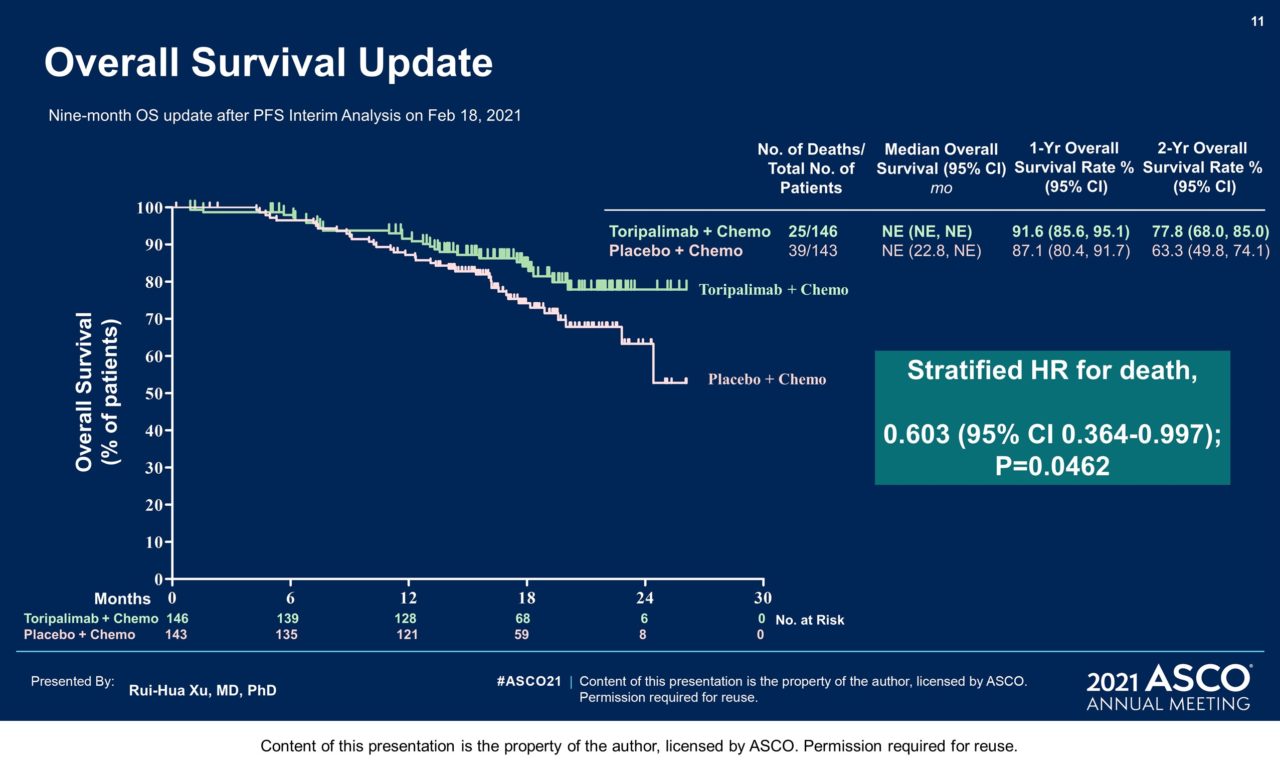
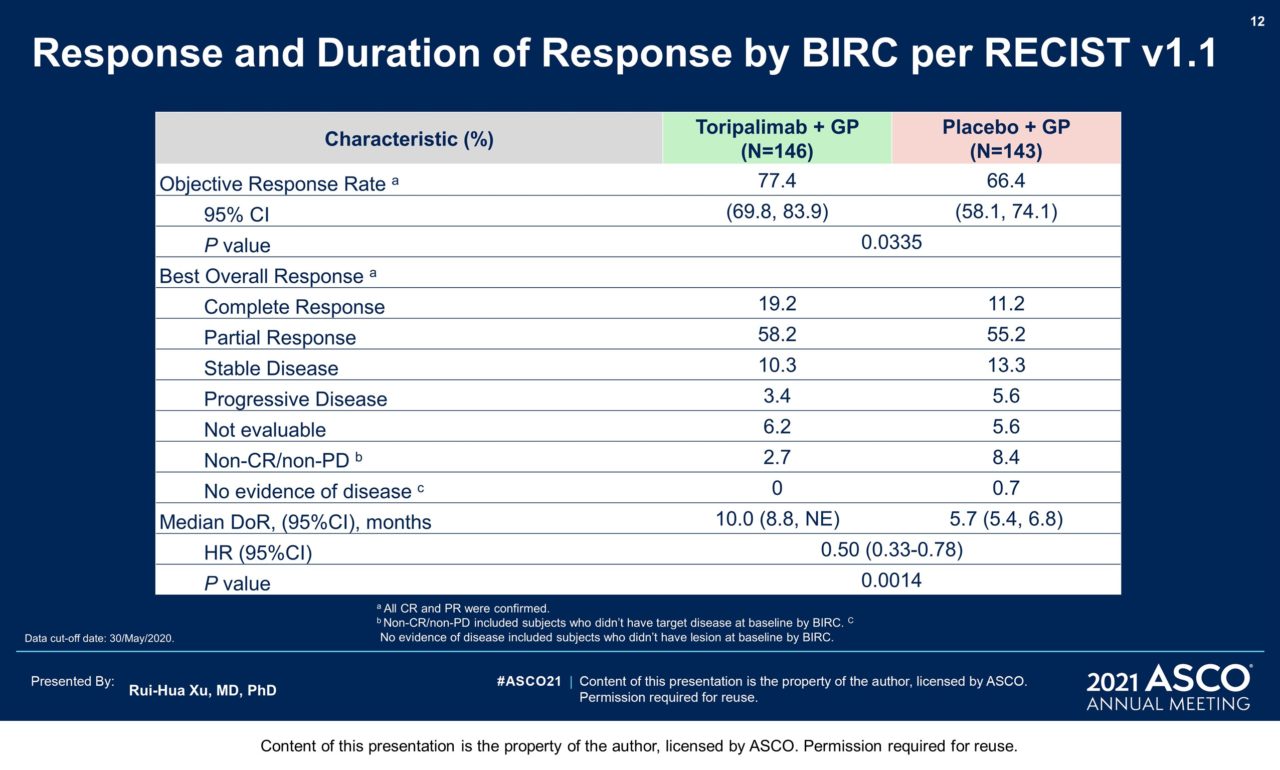
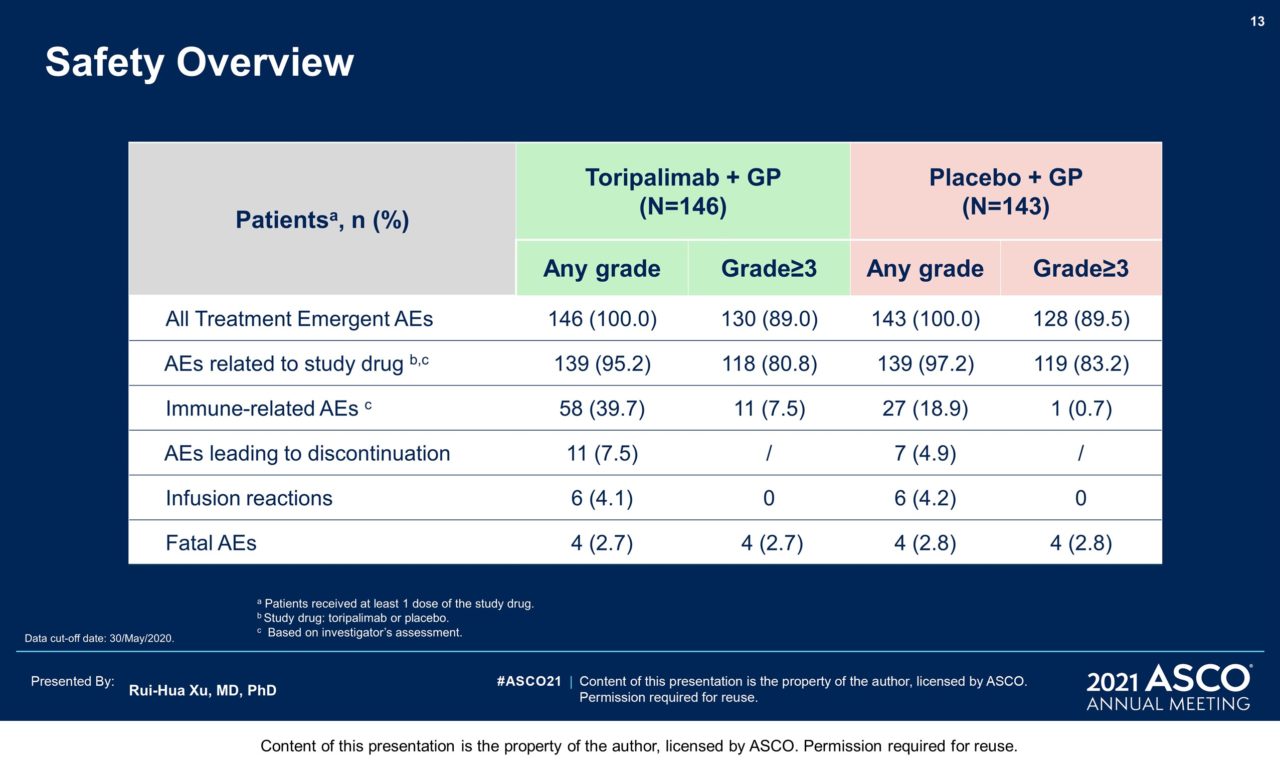
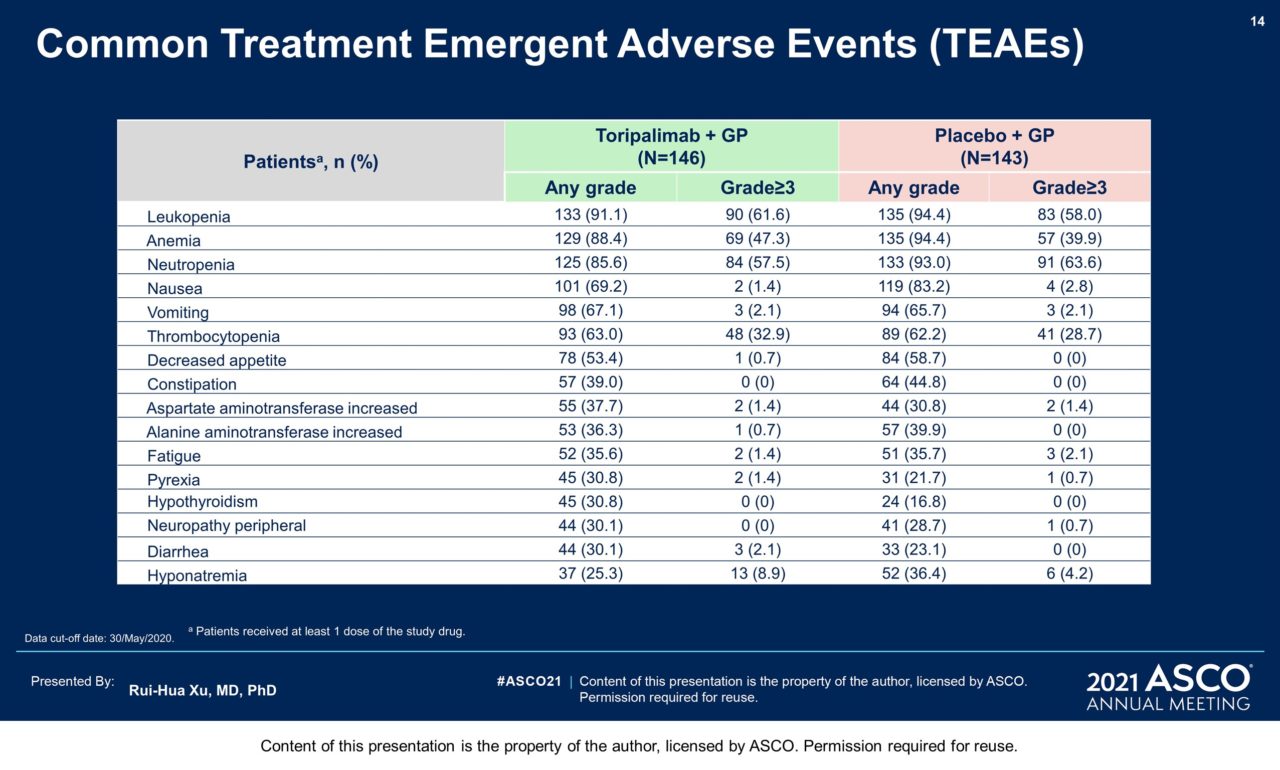
Results: 289 pts were randomized: 146 to the toripalimab arm and 143 to the placebo arm. By May 30, 2020 as the interim analysis cutoff date, the median treatment duration was 39 weeks in the toripalimab arm and 36 weeks in the placebo arm. A significant improvement in PFS was detected for the toripalimab arm compared to the placebo arm (HR = 0.52 [95% CI: 0.36-0.74] two-sided p = 0.0003), with median PFS of 11.7 vs. 8.0 months. The 1-year PFS rates were 49% and 28% respectively. An improvement in PFS was observed across relevant subgroups, including all PD-L1 subgroups. The ORR was 77.4% vs. 66.4% (P = 0.033) and the median DOR was 10.0 vs. 5.7 months (HR = 0.50 [95% CI: 0.33-0.78]). As of Jan 15, 2021, OS was not mature, with 25 deaths in the toripalimab arm and 35 in the placebo arm (HR = 0.68 [95% CI: 0.41-1.14], P = 0.14). The incidence of Grade ≥3 adverse events (AEs) (89.0% vs 89.5%); AEs leading to discontinuation of toripalimab/placebo (7.5% vs 4.9%); and fatal AEs (2.7% vs 2.8%) were similar between two arms; however, immune-related (irAEs) (39.7% vs. 18.9%) and Grade ≥3 irAEs (7.5% vs. 0.7%) were more frequent in the toripalimab arm.
Conclusions: The addition of toripalimab to GP chemotherapy as 1st-line treatment for pts with advanced NPC provided superior PFS and ORR and longer DOR than GP alone with a manageable safety profile. These results support the use of toripalimab with GP chemotherapy as the new standard care for this population. Clinical trial information: NCT03581786
9512 – A phase 2 clinical trial of neoadjuvant anti-PD-1 ab (toripalimab) plus axitinib in resectable mucosal melanoma
Authors: Chuanliang Cui, Xuan Wang, Bin Lian, Lu Si, Zhihong Chi, Xinan Sheng, Yan Kong, Lili Mao, Xue Bai, Bixia Tang, Xieqiao Yan, Siming Li, Li Zhou, Jun Guo
Organizations: Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Renal Cancer and Melanoma, Peking University Cancer Hospital & Institute, Beijing, China, Department of Renal Cancer and Melanoma, Peking University Cancer Hospital & Institute, Beijing, China
Research Funding: Shanghai Junshi Biosciences
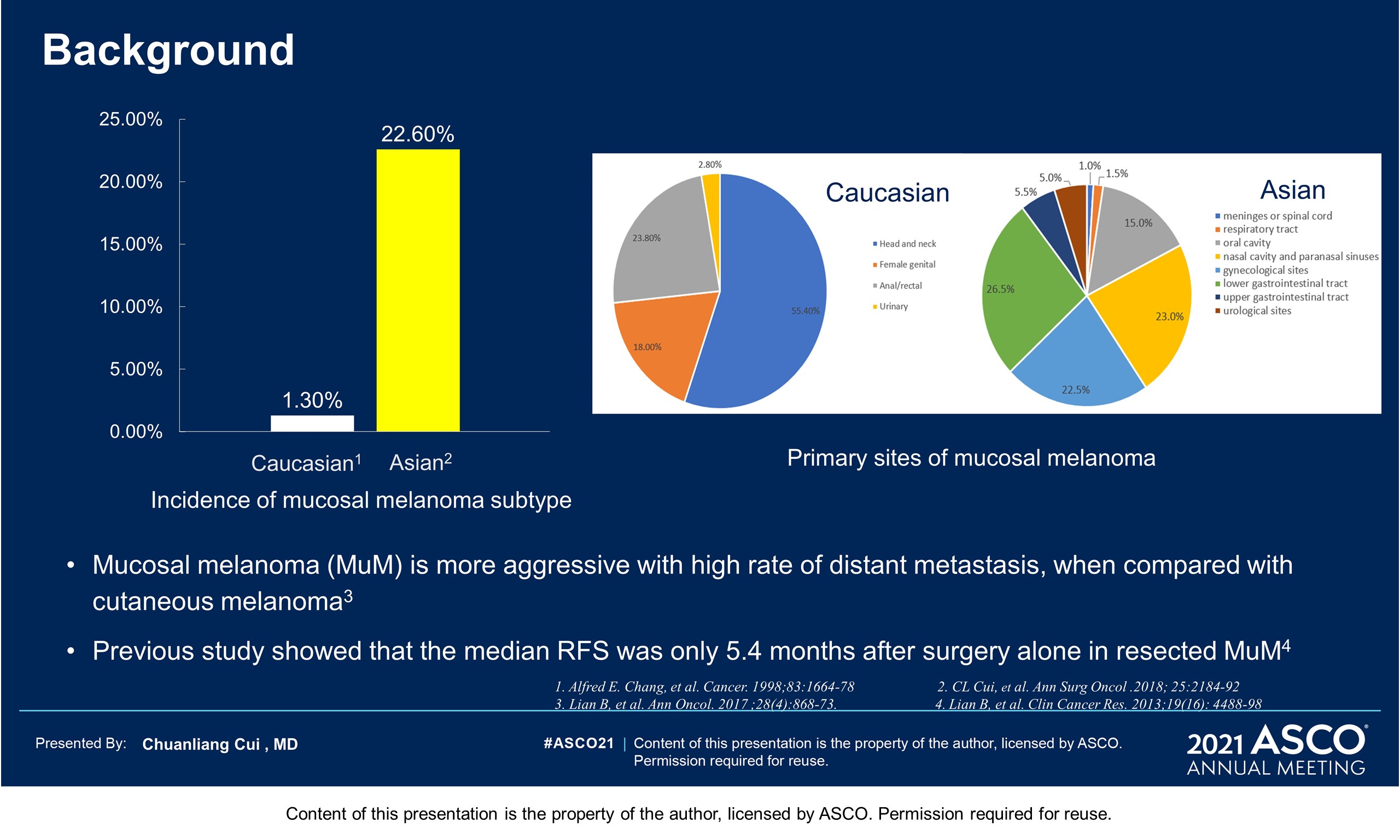
Background: The outcome of patients (pts) with resectable mucosal melanoma (MM) is still poor. Toripalimab combined with axitinib has shown impressive results in metastatic MM with an ORR of 48.3% and a median PFS of 7.5 months in a phase 1b trial. It was hypothesized that this combination therapy might cause pathologic response in neoadjuvant setting for resectable MM, so we conducted this single arm phase 2 trial.
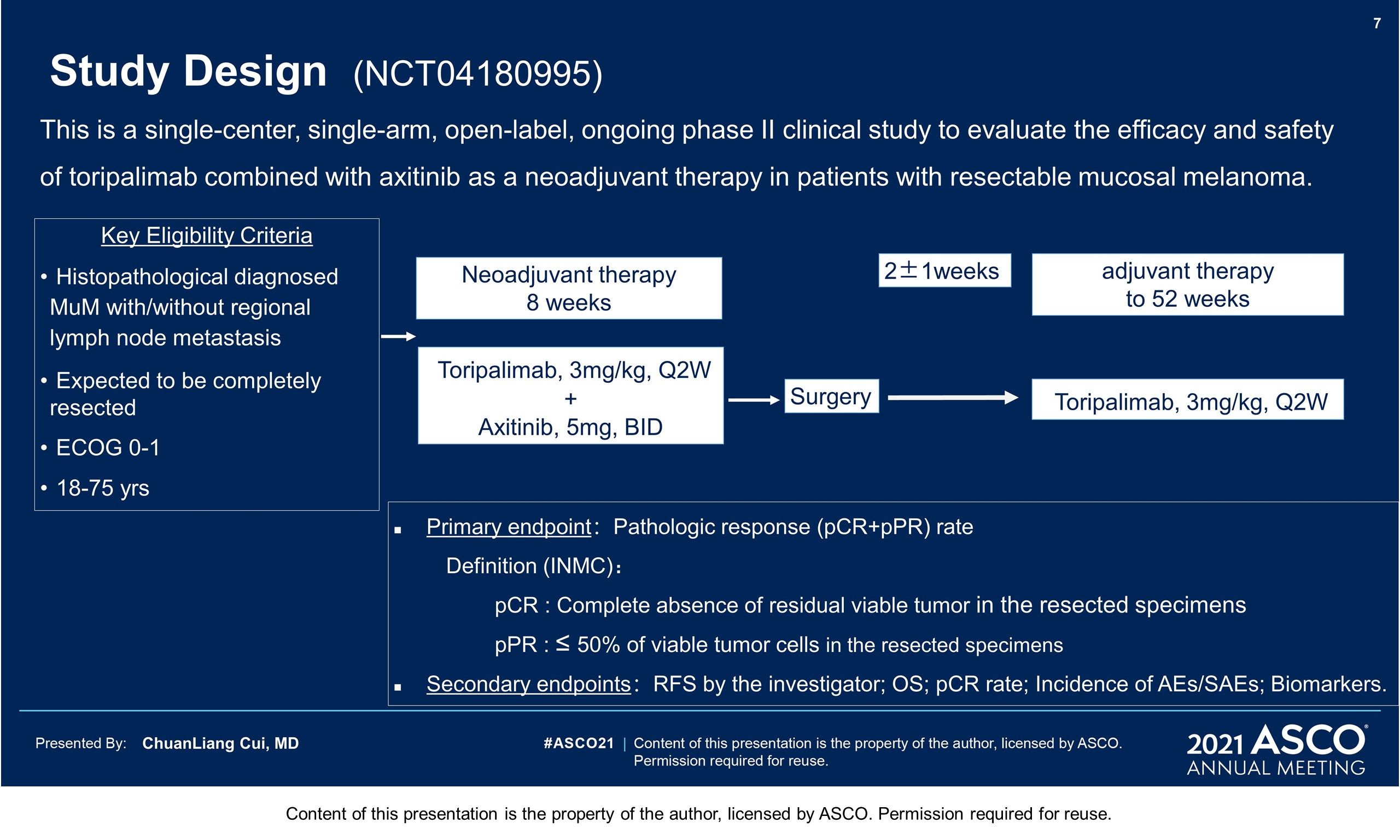
Methods: Eligible pts were adults (aged 18 to 75) with histologically confirmed resectable (localized or regional lymph node metastasis) MM disease. Exclusion criteria included ocular or unknown primary melanoma, distant metastatic disease or previous use of anti PD-1 ab. Pts received toripalimab 3 mg/kg Q2W plus axitinib 5 mg BID for 8 weeks as neoadjuvant therapy, then surgery and the adjuvant toripalimab 3 mg/kg Q2W starting 2±1week after surgery for totally 52 weeks. The primary end point is pathologic response rate according to the International Neoadjuvant Melanoma Consortium (pCR+pPR, pCR is defined as the complete absence of residual viable tumor and pPR ≤ 50% of viable tumor cells). The secondary end point is RFS in the ITT population. Clinical trial information: NCT04180995.
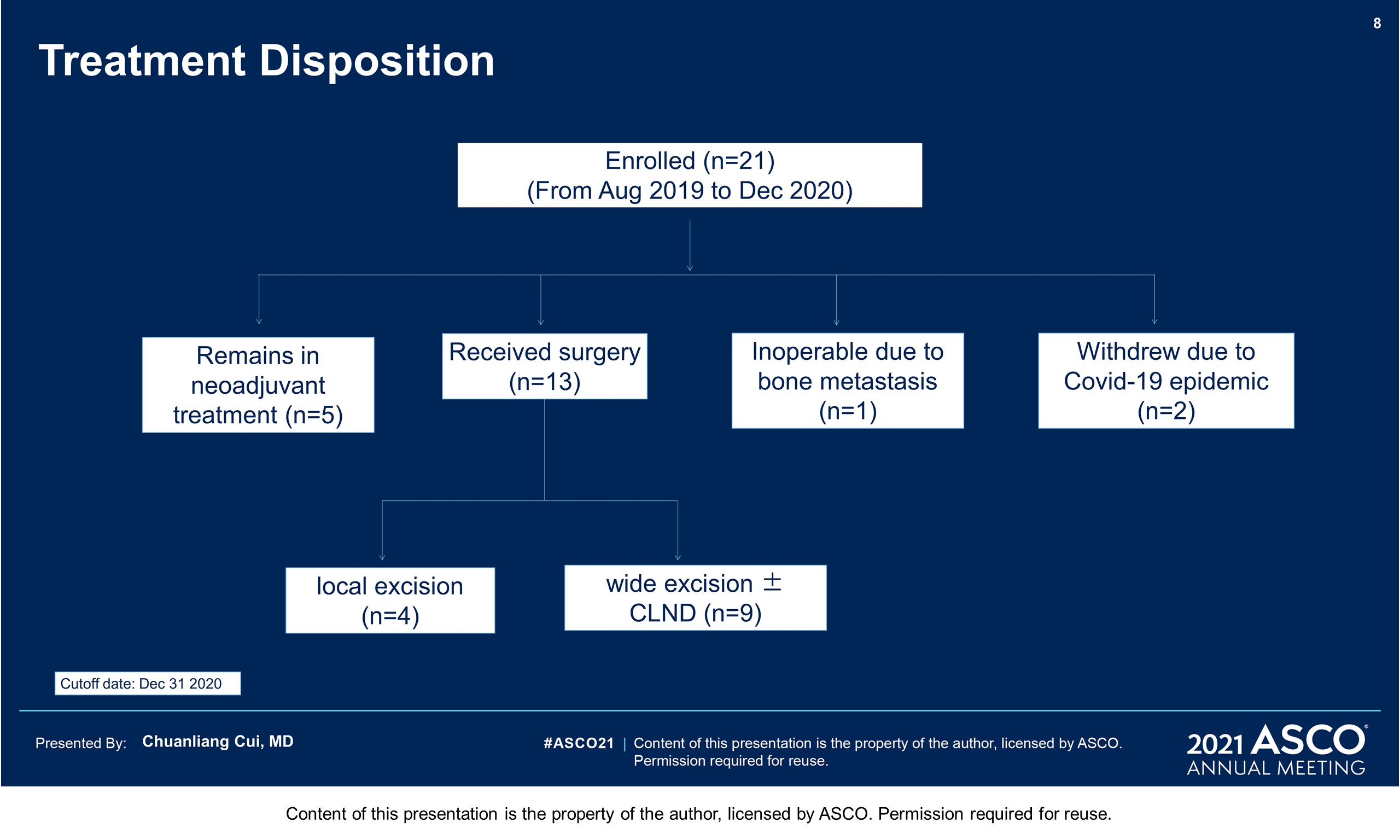
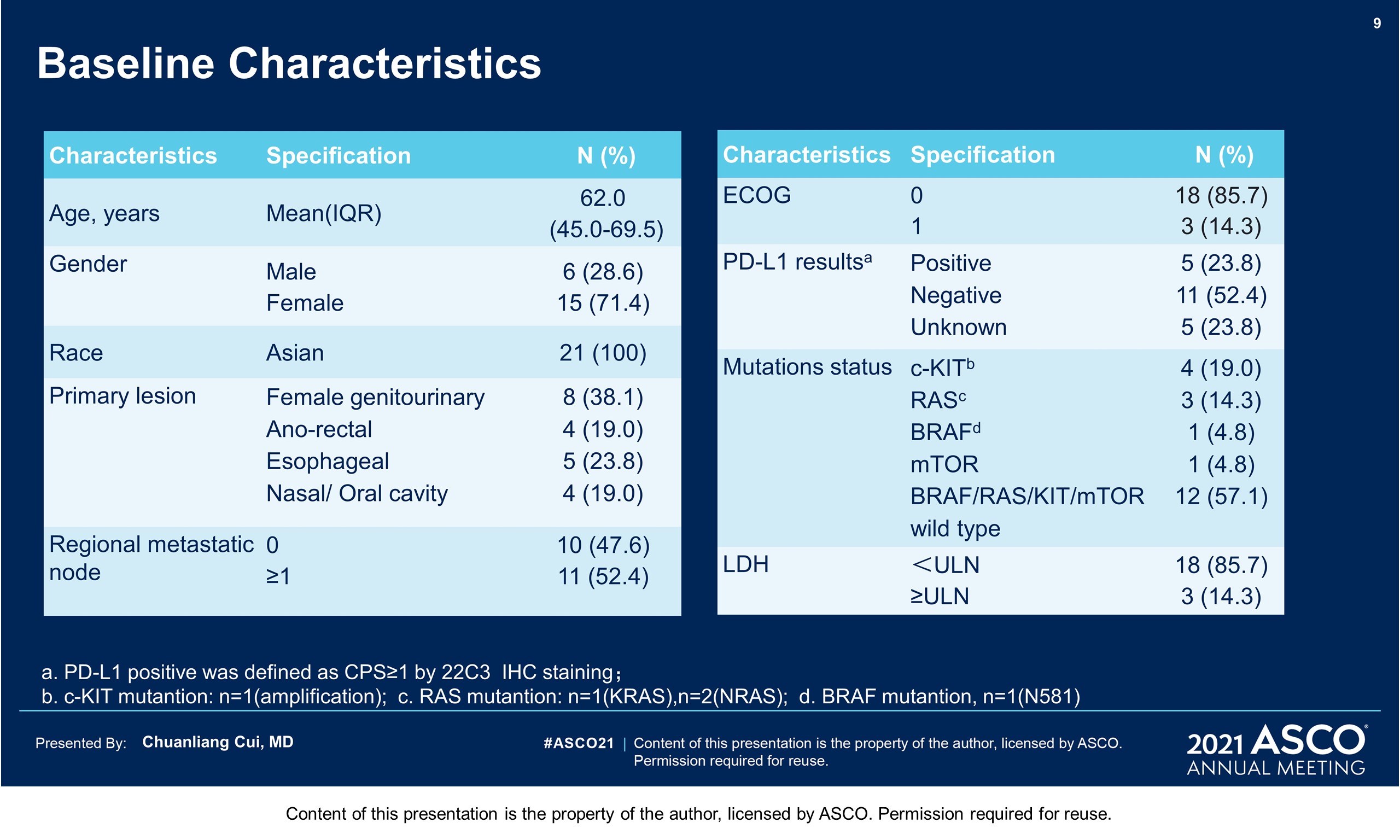
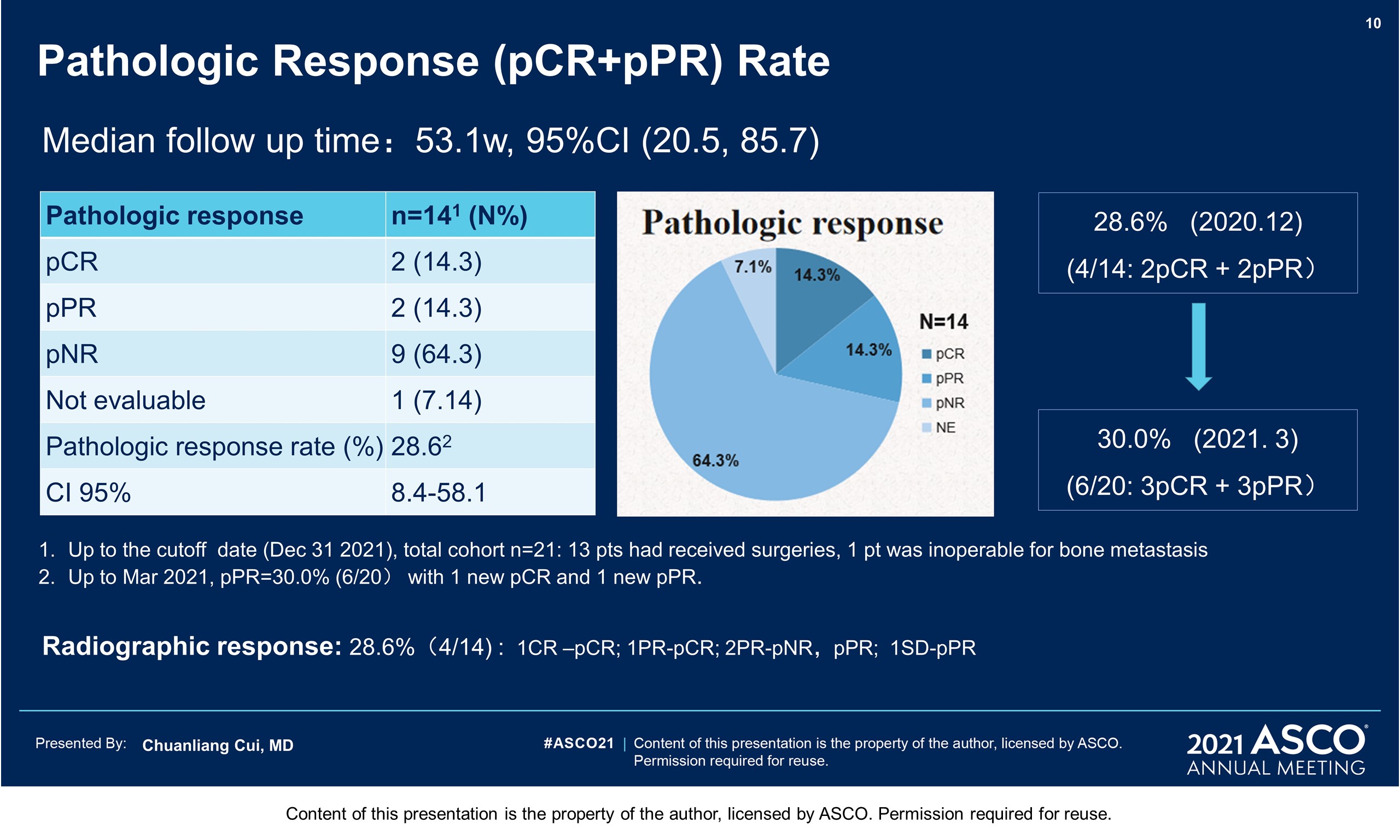
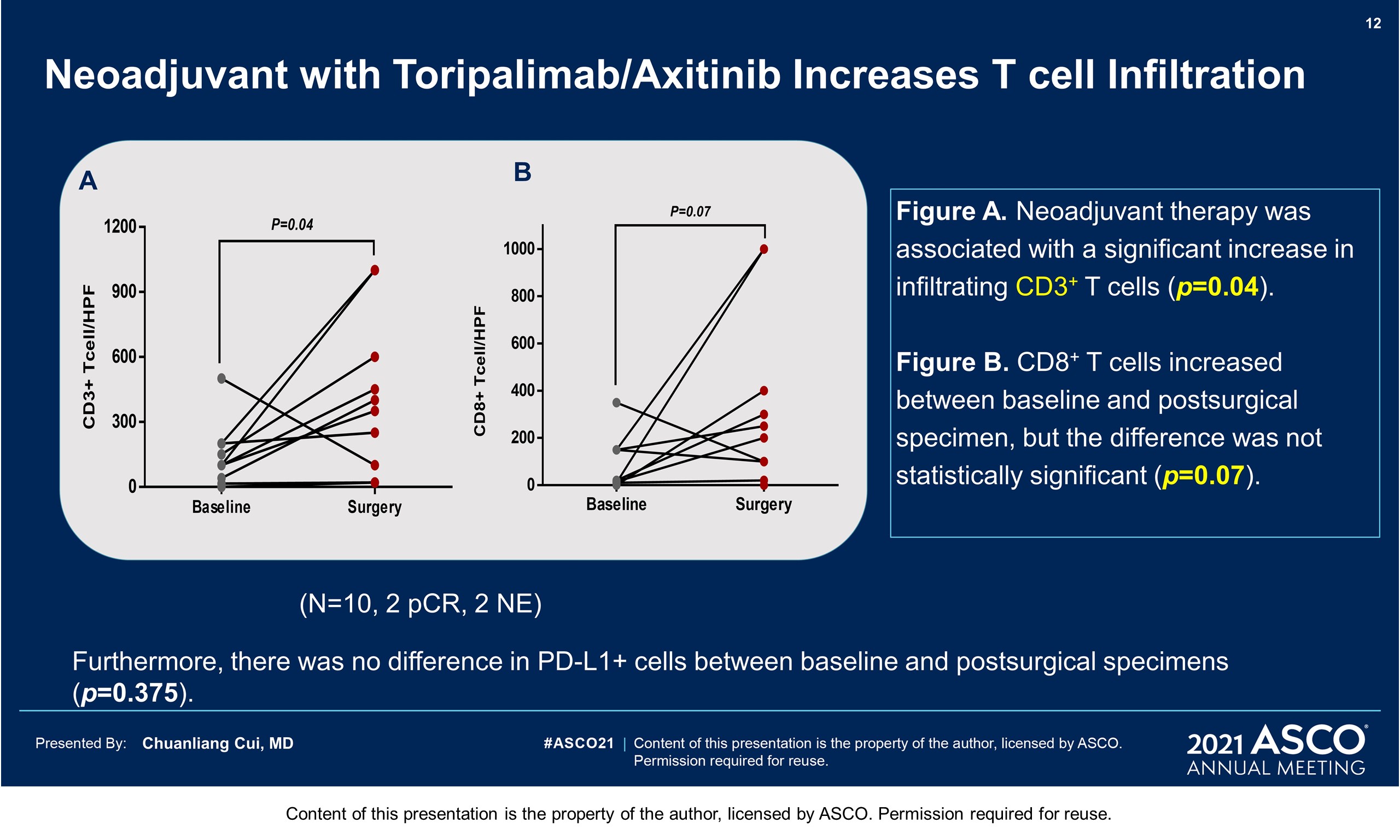
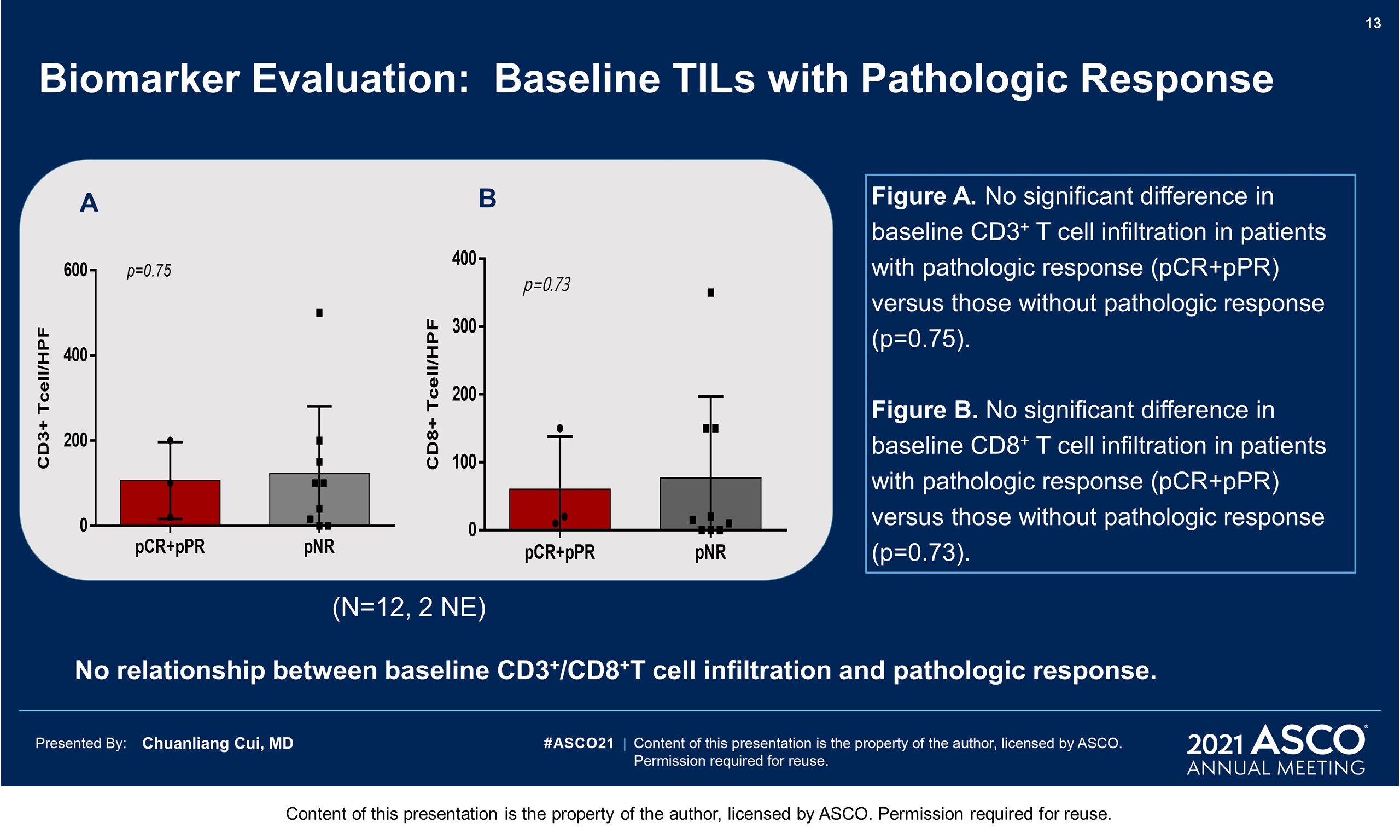
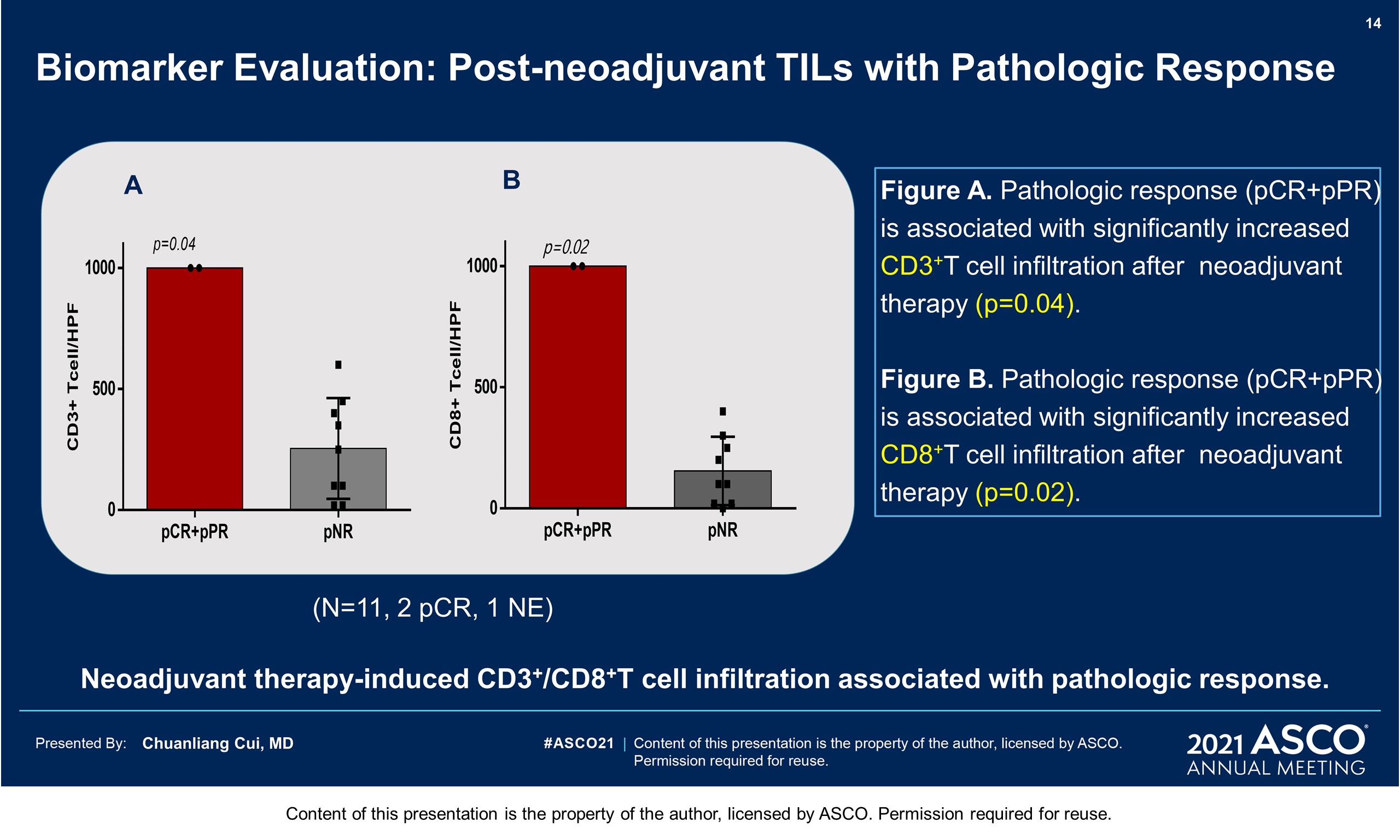
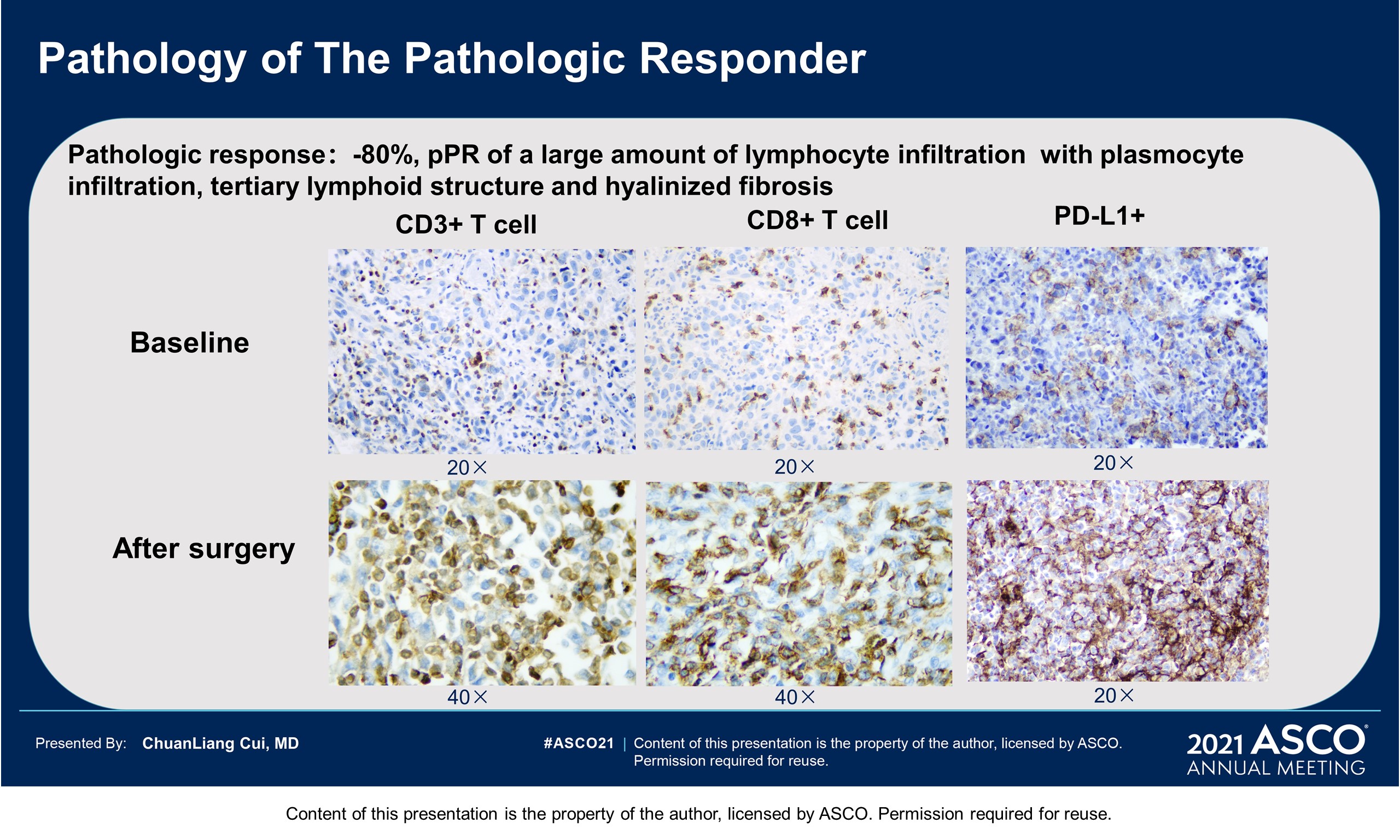
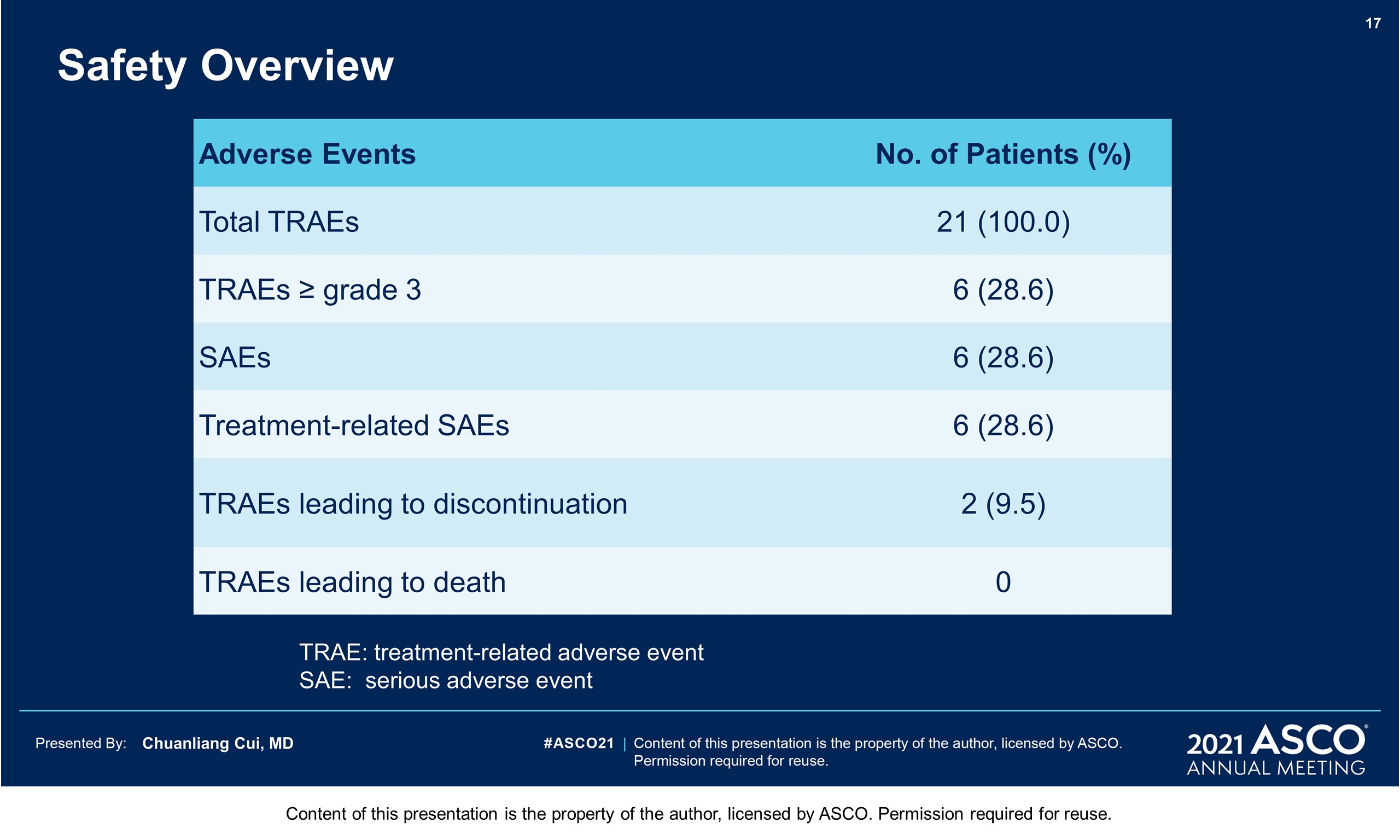
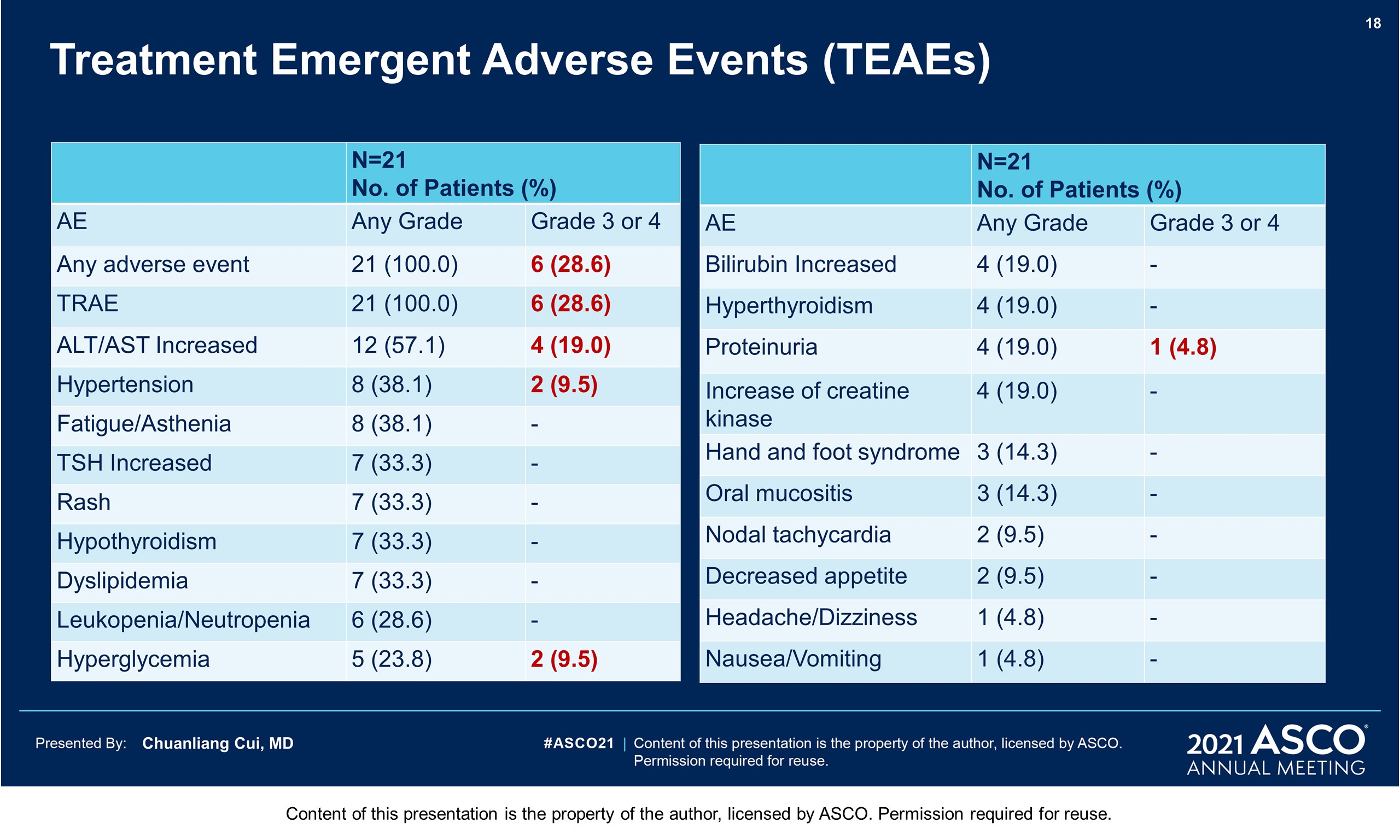
Results: From Aug 2019 to Dec 2020, 21 pts have been eligible and enrolled. Basic characteristics: median age 62 years; M: F 28.6% : 71.4%; primary sites 8 femal genital(1urethra, 7vagina), 5 esophagus, 4 ano-rectal, 4 head & neck(3 nasal,1 oral), in which 47.6% localized disease (T3/4 60%), 52.4% regional lymphatic disease; Gene mutation: 4 cKit (1 amplification), 2 Nras,1 Braf (N581), 1mTOR. This therapy was tolerable with grade 3-4 treatment related AEs of 23.8% (liver dysfunction 14.3%, hyperglycemia 9.5% and hypertension 4.8%). 13 pts had received surgeries (local excision 30.8%, wide excision ± CLND72.7%)and 5 pts still in neoadjuvant treatment. One patient was inoperable for bone metastasis, and 2 pts withdrew for covid 19 epidemic. At a median follow up time of 59 weeks, the pathologic response rate was 28.6% (4/14, 2 pCR, 2pPR). Of the post-surgical specimens, 61.5% (8/13) showed significant TIL infiltration, with 38.5% Brisk and 23.1% Nonbrisk according to the definition of AJCC 8th edition. Plenty of plasma cells, histiocyte and pigment with hyaline fibrosis were also found in responders. No recurrence or metastasis was observed in responders until now, with a RFS reaching more than 58weeks. 5 pts with pNR( > 50% viable tumor cells) got disease progression, with 1 local recurrence, 1 regional lymphatic metastasis, and 3 distant metastases. The median RFS has not been reached.
Conclusions: Neoadjuvant toripalimab plus axitinib in resectable MM has shown promising pathologic responses with good tolerance, which supports further investigation of neoadjuvant therapies in MM. Survival is still in follow-up. Clinical trial information: NCT04180995
6023 – Toripalimab plus intensity-modulated radiotherapy for recurrent nasopharyngeal carcinoma: An open-label single-arm, phase II trial
Authors: Mingyuan Chen, Yijun Hua, Rui You, Zhi-Qiang Wang, Peiyu Huang, Mei Lin, Yan-Feng Ouyang
Organizations: Sun Yat-sen University Cancer Center, Guangzhou, China, Sun Yat-sen Univerisity Cancer Center, Guangzhou, China
Research Funding: Key-Area Research and Development of Guangdong Province
Background: Toripalimab is a humanized immunoglobulin G4 monoclonal antibody against programmed death 1 (PD-1). We aimed to investigate the efficacy and safety of toripalimab in combination with intensity-modulated radiotherapy (IMRT) for recurrent nasopharyngeal carcinoma (rNPC).
Methods: We conducted a single-arm, phase II trial with rNPC patients who had biopsy-proven disease and were unsuitable for local surgery. Eligible patients received IMRT in combination with toripalimab administered via intravenous infusion of 240 mg once every 3 weeks for a maximum of seven cycles. The primary endpoint was the objective response rate (ORR). The secondary endpoints included safety profiles, progression-free survival (PFS).
Results: Between May 2019 and January 2020, a total of 25 rNPC patients were enrolled (18 men [72.0%] and 7 women [28.0%]; median [IQR] age, 49.0 [43.5-52.5] years). With a median (IQR) follow-up duration of 14.6 months (13.1-16.2) months, 19 patients (79.2%) achieved an overall response, and disease control was achieved in 23 (95.8%) patients at 3 months post radiotherapy. The 12-month progression-free survival was 91.8% (95% CI 91.7% – 91.9%). The incidences of acute (grade ≥3) blood triglyceride elevation, creatine phosphokinase elevation, skin reaction, and mucositis were 1 (4.0%), 1 (4.0%), 2 (8.0%), and 1 (4.0%), respectively. The incidences of late severe (grade ≥3) nasopharyngeal wall necrosis, nasal bleeding, and trismus were 28.0%, 12.0%, and 4.0%, respectively.
Conclusions: Toripalimab combined with IMRT was tolerable and showed promising antitumor activity in rNPC patients. Clinical trial information: NCT03854838
9573 – Adjuvant anti-PD-1 ab (Toripalimab) versus high-dose IFN-a2b in resected mucosal melanoma: A phase II randomized trial
Authors: Chuanliang Cui, Bin Lian, Lu Si, Zhihong Chi, Xinan Sheng, Yan Kong, Xuan Wang, Hui Tian, Lili Mao, Xue Bai, Bixia Tang, Xieqiao Yan, Siming Li, Li Zhou, Jun Guo
Organizations: Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Renal Cancer and Melanoma, Peking University Cancer Hospital & Institute, Beijing, China, Peking University Cancer Hospital and Institute, Beijing, China, Department of Renal Cancer and Melanoma, Peking University Cancer Hospital & Institute, Beijing, China
Research Funding: Shanghai Junshi biosciences Co
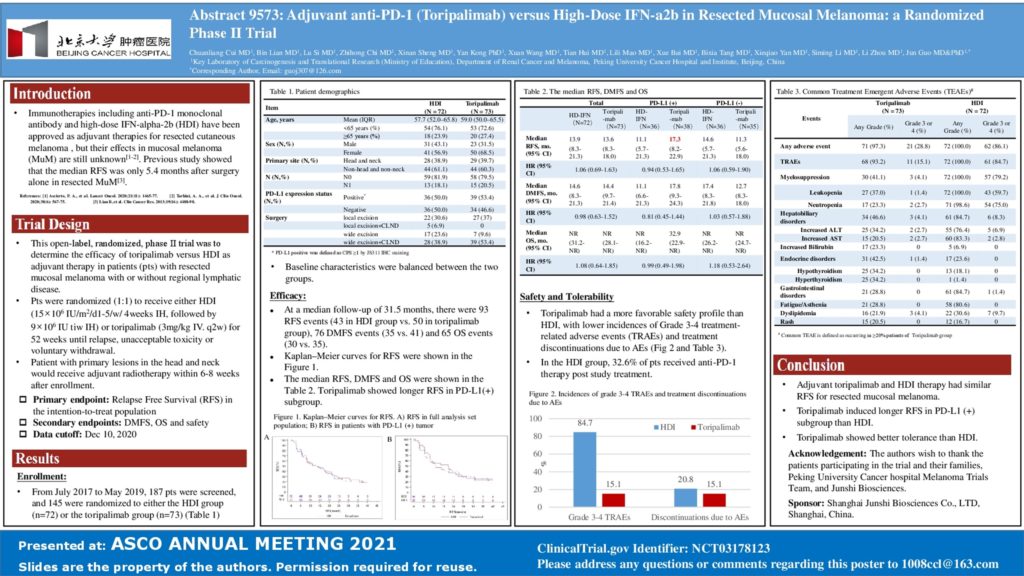
Background: Immunotherapies including anti PD-1 ab and high-dose IFN-a2b (HDI) have been approved for adjuvant therapies in resected cutaneous melanoma, but their roles in mucosal melanoma are still unknown. To determine the efficacies of toripalimab versus HDI as adjuvant therapy in patients (pts) with resected mucosal melanoma, this open-label, phase randomized trial was conducted.
Methods: Mucosal melanoma pts who have undergone complete resections of localized with or without regional lymphatic disease were randomly (1:1) assigned to receive HDI (15×106 IU/m2/d1-5/w/ 4weeks IH, followed by 9×106 IU tiw IH) or toripalimab (3mg/kg intravenously q2w) for 52 weeks unless disease recurrence, unacceptable toxicity or consent withdrawal. Head and neck primary would receive adjuvant radiotherapy within 6-8weeks after enrollment. The primary end point was RFS in the intention-to-treat population. Data cutoff was December 10, 2020. Clinical trial information: NCT03178123.
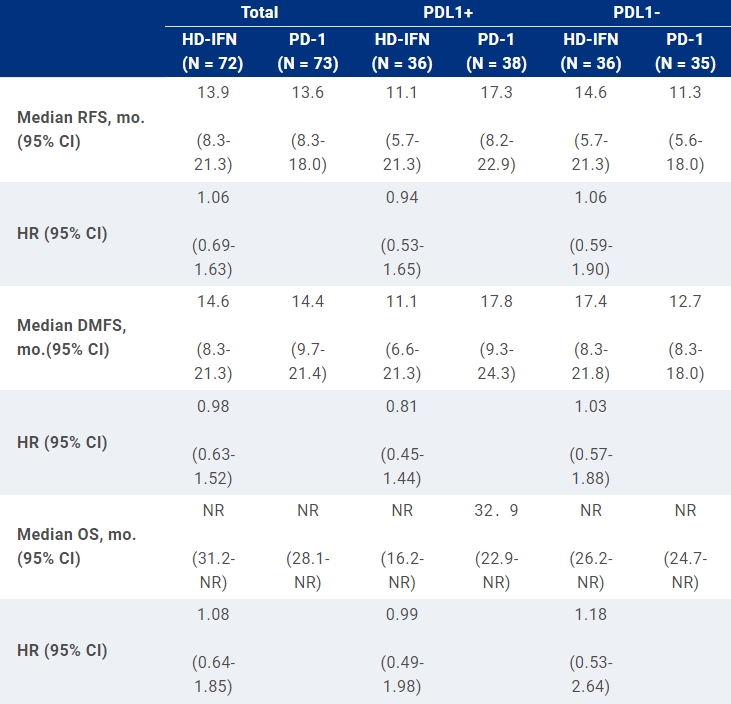
Results: From Jul 2017 to May 2019, 187 pts were screened, and 145 were randomized into HDI group (n = 72) and toripalimab group (n = 73). The median age was 58years; M:F 37.2%: 62.8%; localized disease 80.7%, regional lymphatic disease 19.3%; local excision± CLND 37.2%, wide excision ± CLND 62.8%; head and neck primary 39.3%( 87.5% received adjuvant radiotherapy); PDL-1positive 51.0%(CPS≥1%, 22C3), PDL-1 negative 49.0%. There was no difference in baseline characteristics between two groups. At a median follow-up of 31.5 months, there were 93 RFS events (43 in HDI group vs. 50 in toripalimab group), 76 DMFS events (35 vs. 41respectively) and 65 OS events (30 vs. 35 respectively). The median RFS, DMFS and OS were shown in the table. In the HDI group, 32.6% of pts received anti PD-1 ab in the following treatment. Grade 3/4 AEs were reported in 83.3% of pts in HDI group (most decrease of leukocytes or neutrophils) and 15.1% of pts in toripalimab group (mainly increase of amylase or liver enzymes). Discontinuations of treatment due to any AE occurred in 20.8% of HDI group and 15.1% of toripalimab group.
Conclusions: Both adjuvant toripalimab and HDI therapy improve RFS of mucosal melanoma. Toripalimab shows longer RFS in PDL1 (+) subgroup and better tolerance than HDI. Clinical trial information: NCT03178123
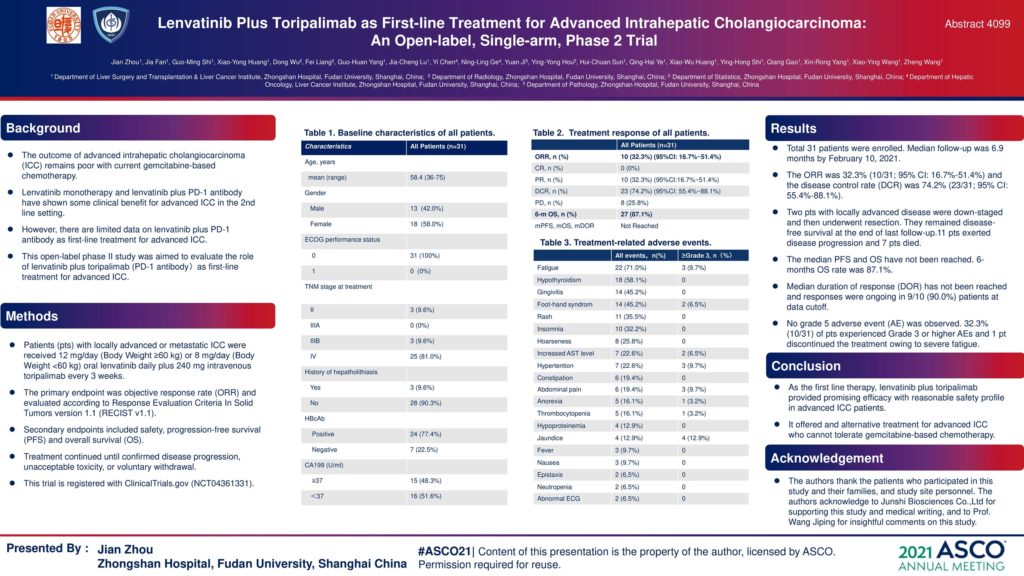
4099 – Lenvatinib plus toripalimab as first-line treatment for advanced intrahepatic cholangiocarcinoma: A single-arm, phase 2 trial
Authors: Zhou Jian, Jia Fan, Guo-Ming Shi, Xiao-Yong Huang, Dong Wu, Fei Liang, Guo-Huan Yang, Jia-Cheng Lu, Yi Chen, Ning-Ling Ge, Yuan Ji, YY Hou, Hui-Chuan Sun, Shuang-Jian Qiu, Qing-Hai Ye, Xiao-Wu Huang, Ying-Hong Shi, Qiang Gao, Xin-Rong Yang, Xiao-Ying Wang
Organizations: Department of Liver Surgery and Transplantation, Liver Cancer Institute, Zhongshan Hospital, Fudan University, Shanghai, China, Fudan University ZhongShan Hospital, Shanghai, China, Fudan University, Shanghai, China, Zhongshan Hospital, Fudan University, Shanghai, China, Department of Statistics, Zhongshan Hospital, Fudan University, Shanghai, China, Department of Liver Surgery, Zhongshan Hospital, Fudan University, Shanghai, China, Zhongshan Hospital, Shanghai, China, Department of Hepatic Oncology, Liver Cancer Institute and Zhongshan Hospital, Fudan University, Shanghai, China, Department of Pathology, Zhongshan Hospital, Fudan University, Shanghai, China, Liver Cancer Institute and Zhongshan Hospital, Fudan University, Shanghai, China
Research Funding: None
Background: Lenvatinib monotherapy and lenvatinib plus PD-1 antibody have shown some clinical benefit for advanced intrahepatic cholangiocarcinoma (ICC) in the second-line setting. Our study assesses the role of lenvatinib plus toripalimab (PD-1 antibody) for advanced ICC patients as the first line therapy.
Methods: Patients (pts) with locally advanced or metastatic ICC received 12 mg/day (Body Weight ≥60 kg) or 8 mg/day (Body Weight <60 kg) oral lenvatinib daily plus 240 mg intravenous toripalimab every 3 weeks. The primary endpoint was objective response rate (ORR) and evaluated according to Response Evaluation Criteria In Solid Tumors version 1.1 (RECIST v1.1). Secondary endpoints included safety, progression-free survival (PFS) and overall survival (OS). Treatment continued until confirmed disease progression, unacceptable toxicity, or voluntary withdrawal. This trial is registered with ClinicalTrials.gov (NCT04361331).
Results: From March 2020 to Sep. 2020, 31 pathologically confirmed advanced ICC pts with a mean age of 58.4 (range, 25-73) years, including 18 women (58.0%), were enrolled at Zhongshan Hospital, Fudan University. At the end of last follow-up (February 10, 2021), the ORR was 32.3% (10/31; 95% CI: 16.7%-51.4%) and the disease control rate (DCR) was 74.2% (23/31; 95% CI: 55.4%-88.1%). Median follow-up was 6.9 months. Two pts with locally advanced disease were down-staged and then underwent resection. They remained disease-free survival at the end of last follow-up. 11 pts exerted disease progression and 7 pts died. The median PFS and OS have not been reached. Median duration of response (DOR) has not been reached and responses were ongoing in 9/10 (90.0%) pts at data cutoff. 6-months OS rate was 87.1%. No grade 5 adverse event (AE) was observed in present study. 32.3% (10/31) of pts experienced Grade 3 or higher AEs and 1 pts discontinued the treatment owing to severe fatigue.
Conclusions: As the first-line therapy, lenvatinib plus toripalimab provided promising efficacy with reasonable safety profile in advanced ICC patients. It offered an alternative treatment for advanced ICC who cannot tolerate gemcitabine-based chemotherapy. Clinical trial information: NCT04361331
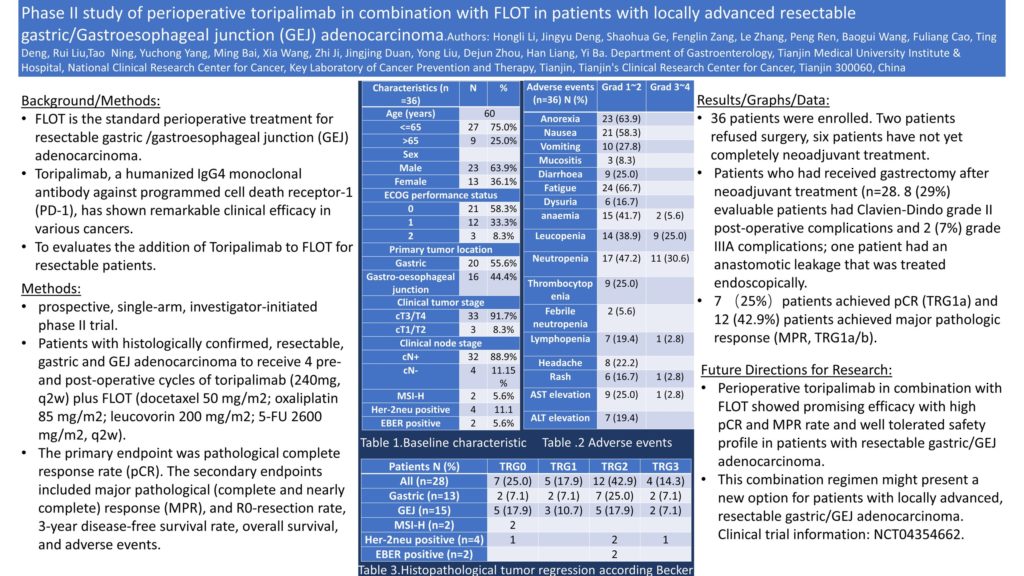
4050 – Phase II study of perioperative toripalimab in combination with FLOT in patients with locally advanced resectable gastric/gastroesophageal junction (GEJ) adenocarcinoma
Authors: Hongli Li, Jingyu Deng, Shaohua Ge, Fenglin Zang, Le Zhang, Peng REN, Baogui Wang, Fuliang Cao, Ting Deng, Rui Liu, Tao Ning, Yuchong Yang, Ming Bai, Xia Wang, Zhi Ji, Jingjing Duan, Yong Liu, Dejun Zhou, Han Liang, Yi Ba
Organizations: Department of Gastrointestinal Medical Oncology, Tianjin Medical University Cancer Institute & Hospital, National Clinical Research Center for Cancer, Key Laboratory of Cancer Prevention and Therapy, Tianjin’s Clinical Research Center for Cancer, Tianjin, China, Department of Gastroenterology (Surgical Oncology), Tianjin Medical University Institute & Hopspital, Tianjin, China, Department of Gastroenterology, Tianjin Medical University Institute & Hospital, National Clinical Research Center for Cancer, Key Laboratory of Cancer Prevention and Therapy, Tianjin’s Clinical Research Center for Cancer, Tianjin, China, Department of Pathology, Tianjin Medical University Institute & Hopspita, Tianjin, China, Department of Esophageal Cancer, Tianjin Medical University Institute & Hopspital., Tianjin, China, Department of Endoscopy, Tianjin Medical University Institute & Hopspital, Tianjin, China, Department of Gastrointestinal Medical Oncology, Tianjin Medical University Cancer Institute & Hospital, National Clinical Research Center for Cancer, Key Laboratory of Cancer Prevention and Therapy, Tianjin, Tianjin’s Clinical Research Center for Cancer, Tianjin, China, Department of Gastrointestinal Medical Oncology, Tianjin Medical University Cancer Institute & Hospital, National Clinical Research Center for Cancer, Key Laboratory of Cancer Prevention and Therapy, Tianjin’s Clinical Research Center for Cancer, Tianjing, China
Research Funding: None
Background: FLOT is the standard perioperative treatment for resectable gastric /gastroesophageal junction (GEJ) adenocarcinoma. However, patient’s outcome is still poor. Toripalimab, a humanized IgG4 monoclonal antibody against programmed cell death receptor-1 (PD-1), has shown remarkable clinical efficacy in various cancers. This trial evaluates the addition of Toripalimab to FLOT for resectable patients.
Methods: This is a prospective, single-arm, investigator-initiated phase II trial. Patients with histologically confirmed, resectable, gastric and GEJ adenocarcinoma (≥cT2 or cN+) were enrolled to receive 4 pre-and post-operative cycles of toripalimab (240mg, q2w) plus FLOT (docetaxel 50 mg/m2; oxaliplatin 85 mg/m2; leucovorin 200 mg/m2; 5-FU 2600 mg/m2, q2w). The primary endpoint was pathological complete response rate (pCR). The secondary endpoints included major pathological (complete and nearly complete) response (MPR), and R0-resection rate, 3-year disease-free survival rate, overall survival, and adverse events.
Results: In total, of 36 patients were enrolled from June 2019 through Dec 2020. Male, 66.7%; median age, 60y; cT3 8.3%, T4, 83.3%; cN+ 88.9%; GEJ 47%; MSI-H, 5.6%, Her-2neu-positive, 5.6%, EBER-positive, 5.6%). Two patients refused surgery, six patients have not yet completely neoadjuvant treatment. 100% of patients completed the 4 pre-cycle. Patients who had received gastrectomy after neoadjuvant treatment (n=28) were included in this analysis. 6 (21%) patients had operations involving a thoracic approach (oesophagogastrectomy with two field lymphadenectomy), 21 (75%) gastrectomy with D2 lymphadenectomy. 8 (29%) evaluable patients had Clavien-Dindo grade II post-operative complications and 2 (7%) grade IIIA complications; one patient had an anastomotic leakage that was treated endoscopically. There were no emergency re-operations. All 28 patients achieved R0-resection and were discharged home after a median of 12 days (range:7-63) in hospital. 7 (25%)patients achieved pCR (TRG1a) and 12 (42.9%) patients achieved major pathologic response (MPR, TRG1a/b). Treatment-related adverse events (TRAEs) to any drug were reported in 30 (94%) patients. Mostly TRAEs were grade 1-2, the grade 3 or 4 TRAEs included neutropenia (34%), neutropenia (25%), lymphopenia (3%), Alanine aminotransferase increased (3%), hypokalemia (3%) and anaemia (3%).
Conclusions: Perioperative toripalimab in combination with FLOT showed promising efficacy with high pCR and MPR rate and well tolerated safety profile in patients with resectable gastric/GEJ adenocarcinoma. This combination regimen might present a new option for patients with locally advanced, resectable gastric/GEJ adenocarcinoma. Clinical trial information: NCT04354662
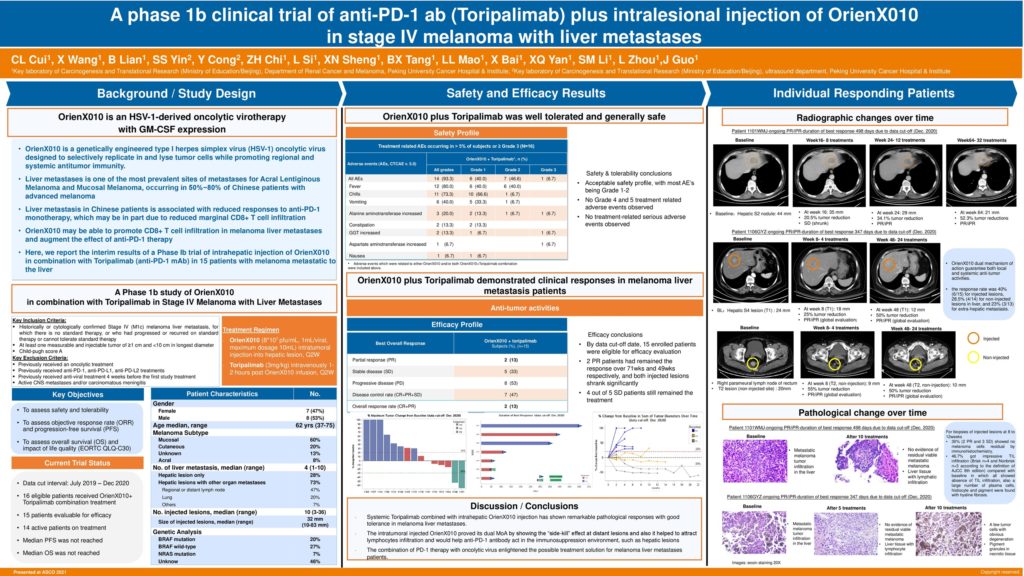
9559 – A phase 1b clinical trial of anti-PD-1 ab (Toripalimab) plus intralesional injection of OrienX010 in stage IV melanoma with liver metastases
Authors: Jun Guo, Chuanliang Cui, Xuan Wang, Bin Lian, Shanshan Yin, Yue Cong, Zhihong Chi, Lu Si, Xinan Sheng, Bixia Tang, Lili Mao, Xue Bai, Xieqiao Yan, Siming Li, Li Zhou
Organizations: Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Renal Cancer and Melanoma, Peking University Cancer Hospital & Institute, Beijing, China, Department of Ultrasound, Peking University Cancer Hospital & Institute, Beijing, China, Department of Renal Cancer and Melanoma, Peking University Cancer Hospital & Institute, Beijing, China
Research Funding: Oriengene Biotechnology Ltd
Background: Liver metastasis was associated with reduced responses and PFS in melanoma patients (pts) treated with anti-PD-1 monotherapy, which is probably due to reduced marginal CD8+ T cell infiltration. Oncolytic virotherapy was found to increase CD8+ T cell infiltration in the injected lesions and improve the efficacy of anti-PD-1 ab in a phase 1b trial. We hypothesized that intratumoral oncolytic virus injection for liver metastasis in melanoma combined with systemic anti-PD-1 therapy might improve the efficacy, thus conducting this phase 1b trial with intratumoral OrienX010 – a HSV-1-derived oncolytic virotherapy with expression of GM-CSF combined toripalimab in liver metastatic melanoma pts.
Methods: Eligible pts included those over 18 with injectable liver metastasis confirmed by biopsy with or without extra-hepatic metastasis; ocular melanoma and brain metastasis were excluded. Pts received intravenous toripalimab Q2W combined with ultrasound guided intratumoral injection of OrienX010 Q2W (8×107 pfu/ml, 10ml per injection) until intolerance or disease progression per iRECIST criteria. Liver biopsy would be performed at baseline and first tumor evaluation (8-12weeks). The primary endpoint was toxicity; secondary endpoints included ORR, DCR and PFS. Clinical trial information: NCT04206358.
Results: From Jul 2019 to Dec 2020, 15 pts were eligible and enrolled. Baseline characteristics: median age 62 yrs; primary: mucosal 60%, cutaneous 20%, unknown primary 13.3%, acral 6.7%; gene mutation status: Braf 20%, Nras 6.7%; 73.3% got extra-hepatic metastasis: regional or distant lymph node 46.7%, lung 20.0%; LDH>ULN 20%; median size of injected lesions: 32mm(10-83mm); median number of liver metastasis: 4(1-10); median number of injection: 10 (3-36). AEs were all grade 1/2: pyrexia 86.7%, rigor 66.7%, elevated transaminase 53.3%, nausea/vomiting 40.0%, fatigue 26.7%. No grade 3-4 AEs. The ORR by investigator was 13.3% (2/15), DCR 46.7% (7/15); the response rate was 40%(6/15) for injected lesions, 28.5%(4/14) for non-injected lesions in liver, and 23% (3/13) for extra-hepatic metastasis. For biopsies of injected lesions at 8 to 12weeks, 30%( 2 PR and 3 SD) showed no melanoma cells residual by immunohistochemistry, 46.7% got impressive TIL infiltration (Brisk n = 4 and Nonbrisk n = 3 according to the definition of AJCC 8th edition) compared with baseline in which all showed absence of TIL infiltration, also a large number of plasma cells, histiocyte and pigment were found with hyaline fibrosis. The PFS has reached 72 weeks for one PR pt. The median PFS was not reached.
Conclusions: Systemic toripalimab combined with intrahepatic OrienX010 injection has shown remarkable pathological responses with good tolerance in melanoma liver metastases. Survival is still in follow-up. Clinical trial information: NCT04206358
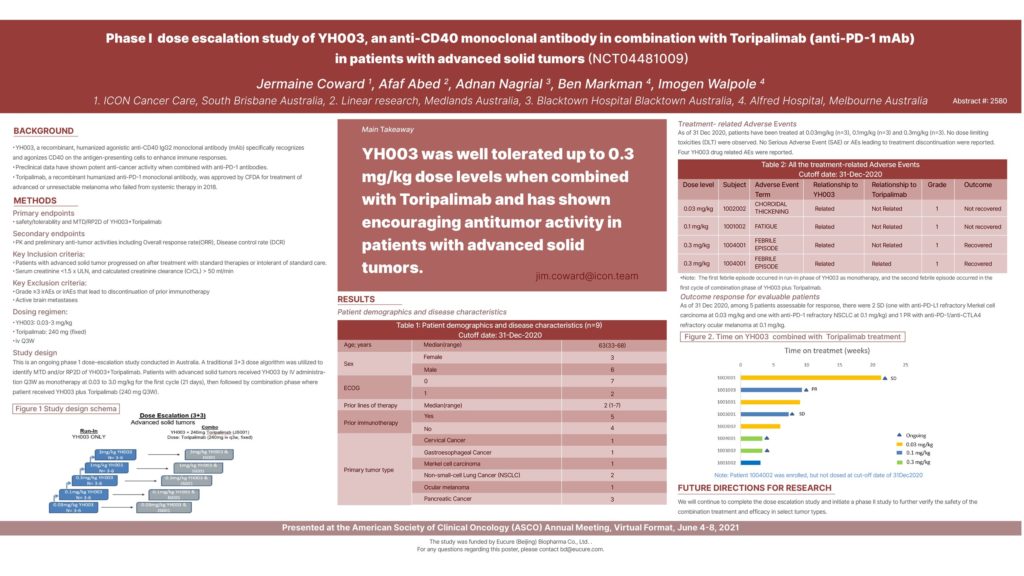
2580 – Phase I open-label, dose escalation of YH003, an anti-CD40 monoclonal antibody in combination with toripalimab (anti-PD-1 mAb) in patients with advanced solid tumors
Authors: Jermaine Coward, Afaf Abed, Adnan Nagrial, Ben Markman
Organizations: ICON Cancer Care, South Brisbane, QLD, Australia, Linear Research, Nedlands, WA, Australia, Blacktown Hospital Cancer and Haematology Centre, Blacktown, NSW, Australia, Alfred Hospital, Melbourne, VIC, Australia
Research Funding: Eucure (Beijing) Biopharma Co., Ltd
Background: YH003, a recombinant, humanized agonistic anti-CD40 IgG2 monoclonal antibody (mAb) specifically recognizes and agonizes CD40 on the antigen-presenting cells to enhance immune responses. Preclinical data have shown potent anti-cancer activity when combined with anti-PD-1 antibodies.
Methods: This is an ongoing phase 1 dose-escalation study. Patients with advanced solid tumors receive YH003 by IV administration Q3W as monotherapy at 0.03 to 3.0 mg/kg for the first cycle (21 days) then in combination with Toripalimab at 240 mg Q3W for the 4 subsequent cycles in an accelerated “3+3” design. The safety, tolerability and preliminary efficacy data will be analyzed.
Results: As of 31 Dec 2020 data cutoff, 9 patients (pts) were enrolled and treated at 0.03 mg/kg (n = 3), 0.1mg/kg (n = 3), and 0.3mg/kg (n = 3). The median age was 63 years (range 33-68). Baseline ECOG scores were 0 (7 pts) and 1 (2 pts) with a median of 2 prior lines therapy (range 1-7). 5 pts had received prior immunotherapy (PD-1/PD-L1 or PD-1+CTLA-4). As of data cutoff, no dose limiting toxicities (DLT) were observed. No Serious Adverse Event (SAE) or AEs leading to treatment discontinuation were reported. Four drug related AEs were reported including one Grade 1 (G1) choroidal thickening (related to YH003) at 0.03 mg/kg, one G1 fatigue (related to YH003) at 0.1 mg/kg, two G1 febrile episodes (one related to YH003 and the other related to combination treatment) at 0.3 mg/kg. Among 5 patients assessable for response, there were 2 SD (one with anti-PDL1 refractory Merkel cell carcinoma at 0.03 mg/kg and one with anti-PD1 refractory NSCLC at 0.1 mg/kg) and 1 PR with anti-PD1/anti-CTLA4 refractory ocular melanoma at 0.1 mg/kg.
Conclusions: YH003 was well tolerated up to 0.3 mg/kg dose levels when combined with Toripalimab and has shown encouraging antitumor activity in patients with advanced solid tumors. Clinical trial information: NCT04481009
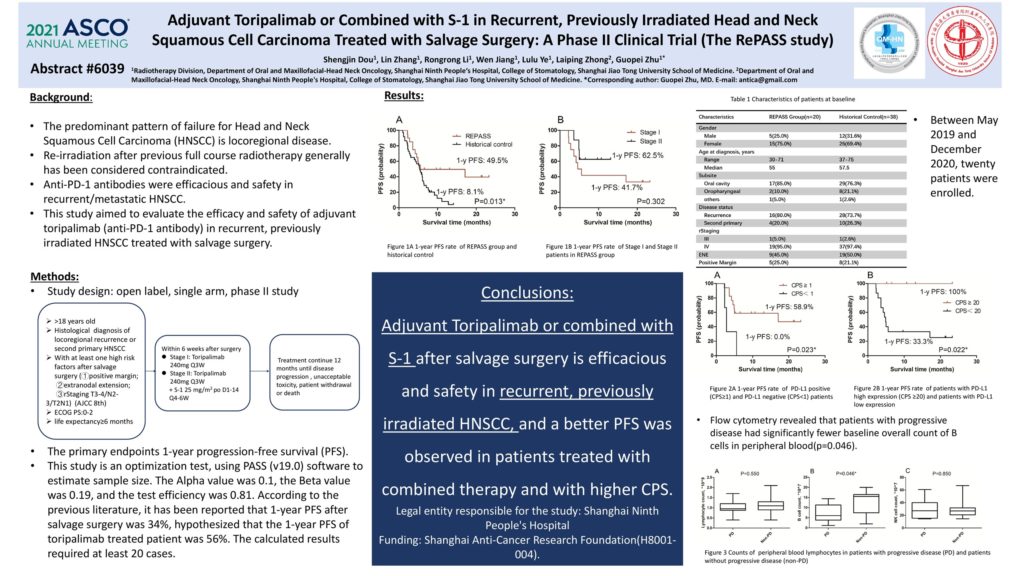
6039 – Adjuvant toripalimab or combined with S-1 in recurrent, previously irradiated head and neck squamous cell carcinoma treated with salvage surgery: A phase II clinical trial (The RePASS study)
Authors: Shengjin Dou, Rongrong Li, Lin Zhang, Wen Jiang, Lulu Ye, Guopei Zhu
Organizations: Radiotherapy Division, Department of Oral and Maxillofacial-Head Neck Oncology, Shanghai Ninth People’s Hospital, College of Stomatology, Shanghai Jiao Tong University School of Medicine, Shanghai, China
Research Funding: Eucure (Beijing) Biopharma Co., Ltd
Background: The predominant pattern of failure for Head and Neck Squamous Cell Carcinoma (HNSCC) is locoregional disease. Salvage surgery remains the standard of care for operable disease. Re-irradiation after previous full course radiotherapy generally has been considered contraindicated. Since anti-PD-1 antibodies were efficacious and safety in recurrent/metastatic HNSCC, this study aimed to evaluate the efficacy and safety of adjuvant toripalimab (anti-PD-1 antibody) in recurrent, previously irradiated HNSCC treated with salvage surgery.
Methods: This study was a single-arm, phase II study. Patients with HNSCC occurring in an area of previously irradiated and with at least one high risk factors after salvage surgery (1- positive margin; 2- extranodal extension; 3- rStaging T3-4/N2-3/T2N1) were enrolled. In the Stage I of 12 patients, patients received toripalimab 240mg once every 3 weeks until confirmed disease progression or unacceptable toxicity, for 12 months. In the stage II of 8 patients with PD-L1 CPS≥1, patients received toripalimab combined with S-1, which was given orally at 25 mg/m2, twice daily, on day 1 to 14, repeated every 21 days for 4-6 cycles. The primary endpoint was 1-year progression-free survival (PFS). We hypothesized a 1-year PFS of at least 56% and assumed a null hypothesis of 34%. A retrospective cohort of 16 patients was compared.
Results: Between May 2019 and December 2020, 20 patients were enrolled. High-risk factors included ENE (35%), positive margin (25%), T3-4(30%) and T2N+(10%). Seventeen patients have PD-L1 CPS≥1 and 3 patients have CPS<1. With a median follow-up of 11.2 months, estimated 1-year PFS and overall survival was 57.0% (95% confidence interval, 32%– 77%) and 79.2% (51%–91%). The primary PFS endpoint has exceeded the hypothesis and its median has not been reached. When compared to the retrospective cohort, the PFS was significantly better(p=0.001),even for Stage I patients(Median PFS: 5.1 vs 3.7 months, p=0.03 ). Stage II patients resulted a better PFS and OS compare to stage I (p=0.02 and p=0.002). For patients with CPS≥1, 1-year PFS and OS was 79.1% (95% confidence interval, 51%–91%) and 91.7%(68%–99%), which were significantly better than patients with CPS<1 (p=0.001 and p=0.05). Adjuvant Toripalimab or combine with S-1 was well-tolerated with no grade 3-4 toxicity and dose interruption as a result of treatment-related adverse event only occurred in 2 patients. Flow cytometry revealed that patients with short PFS had fewer baseline overall count of B cells(p=0.09).
Conclusions: Adjuvant Toripalimab or combined with S-1 after salvage surgery is efficacious and safety in recurrent, previously irradiated HNSCC, and a better PFS was observed in patients treated with combined therapy and with CPS≥1. Further randomized trials are warranted. Clinical trial information: NCT04126460
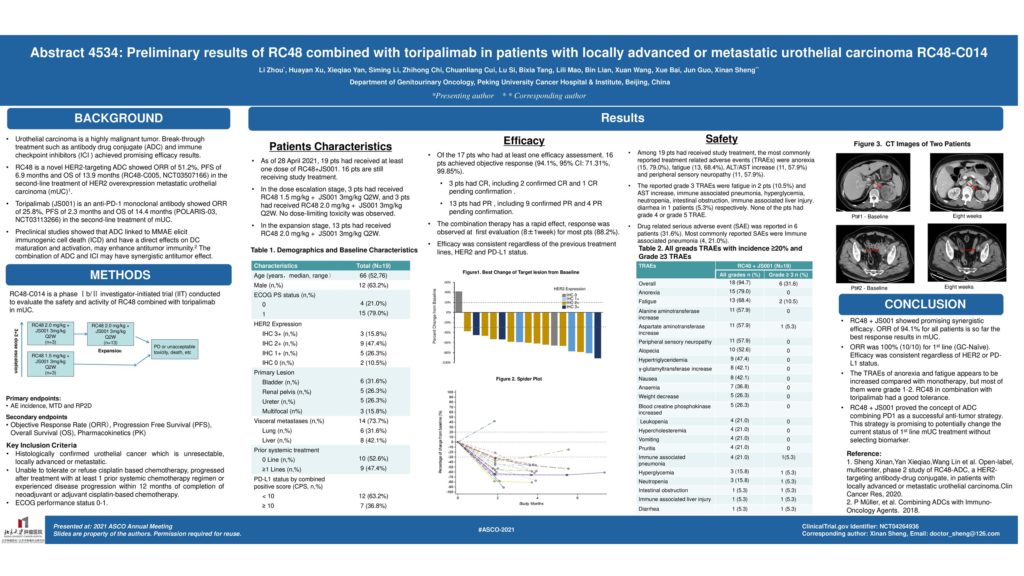
4534 – RC48-ADC combined with toripalimab, an anti-PD-1 monoclonal antibody (Ab), in patients with locally advanced or metastatic urothelial carcinoma (UC): Preliminary results of a phase Ib/II study
Authors: Li Zhou, Huayan Xu, Xieqiao Yan, Zhihong Chi, Chuanliang Cui, Lu Si, Bixia Tang, Lili Mao, Bin Lian, Xuan Wang, Siming Li, Xue Bai, Jun Guo, Xinan Sheng
Organizations: Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Renal Cancer and Melanoma, Peking University Cancer Hospital & Institute, Beijing, China, Department of Renal Cancer and Melanoma, Peking University Cancer Hospital & Institute, Beijing, China
Research Funding: Remegen
Background: RC48-ADC is a novel humanized anti-HER2 antibody-drug conjugate (ADC), which showed promising data in HER2-positive and even negative patients (pts). Anti-PD-1 Abs have durable antitumor effect for mUC especially in PD-L1 positive patients. The combination may have synergistic antitumor effect. This phase 1b/II study evaluated the safety and activity of RC48-ADC combined with toripalimab in mUC.
Methods: In dose-escalation cohort, pts received 1.5 or 2 mg/kg RC48-ADC + 3mg/kg toripalimab with the traditional 3+3 escalation design. In expansion cohort, patients received the recommended dose of RC48-ADC + toripalimab every 2 weeks. The primary endpoints were safety/tolerability and recommended RC48-ADC dose; secondary endpoints included pharmacokinetics, ORR per RECIST 1.1, PFS, and OS stratified by HER2 and PD-L1 expression. HER2 positivity was determined by IHC and in situ hybridization (ISH). PD-L1 expression was tested with IHC 22C3 pharmDx assay.
Results: As of 8 Jan 2021 (data cutoff), 14 mUC pts (9 males, median age 66 y [52-76]) were enrolled. Most pts were systemic treatment naïve (57%) in the locally advanced or metastatic setting. The primary site was in upper tract UC in 50%; 50% had visceral metastases (mets), including 36% with liver mets; HER2 expression was positive (IHC 3+ or 2+ ISH+) in 28%, and 43% PD-L1 CPS≥10. A total of 36 pts is anticipated to be enrolled by Apr 2021. No dose limiting toxicity was reported and the recommended dose for RC48-ADC was 2mg/kg. At data cutoff, 10/14 patients were evaluable for response, with 8 PR, 1 SD (tumor shrinking), and 1 PD. The objective response rate (ORR) was 80%, and disease control rate (DCR) was 90%. All responsive patients have durable efficacy and are still on treatment. Follow-up continues for PFS and OS. Most common treatment-related AEs were grade 1-2, including aminotransferase level increased (7/14, 50%), weight loss (6/14, 43%), alopecia (6/14, 43%), asthenia (4/14, 29%), anemia (3/14, 21%), leukopenia (21%), peripheral sensory neuropathy (21%), hypothyroidism (21%), blood triglycerides increased (21%), and creatine phosphokinase increase (21%). One pt had G3 intestinal obstruction attributed to study drug and went back to treatment after recovery.
Conclusions: RC48-ADC in combination with toripalimab had a good tolerance and promising anti-tumor activity in pts with mUC. Further evaluation of safety and efficacy is ongoing. Clinical trial information: NCT04264936
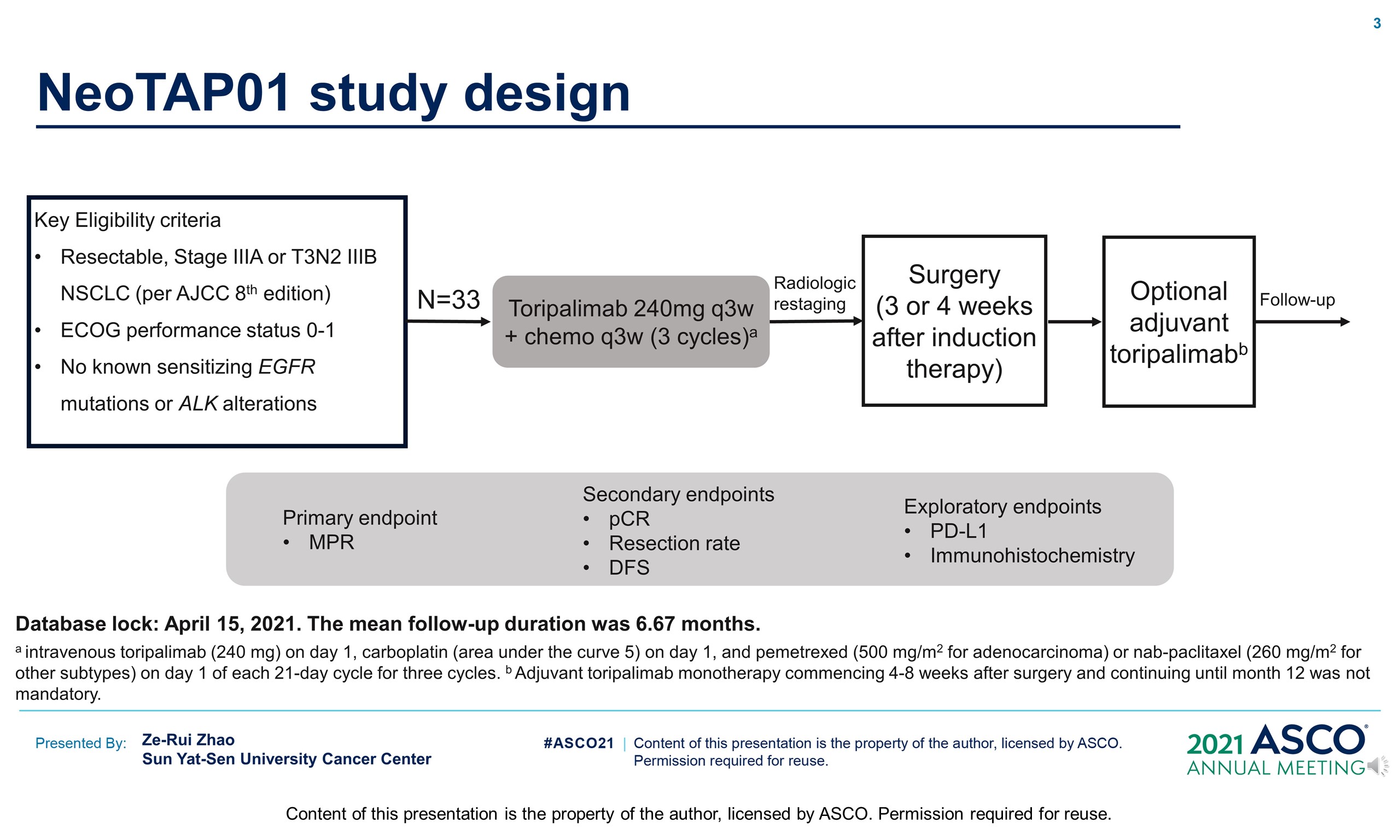
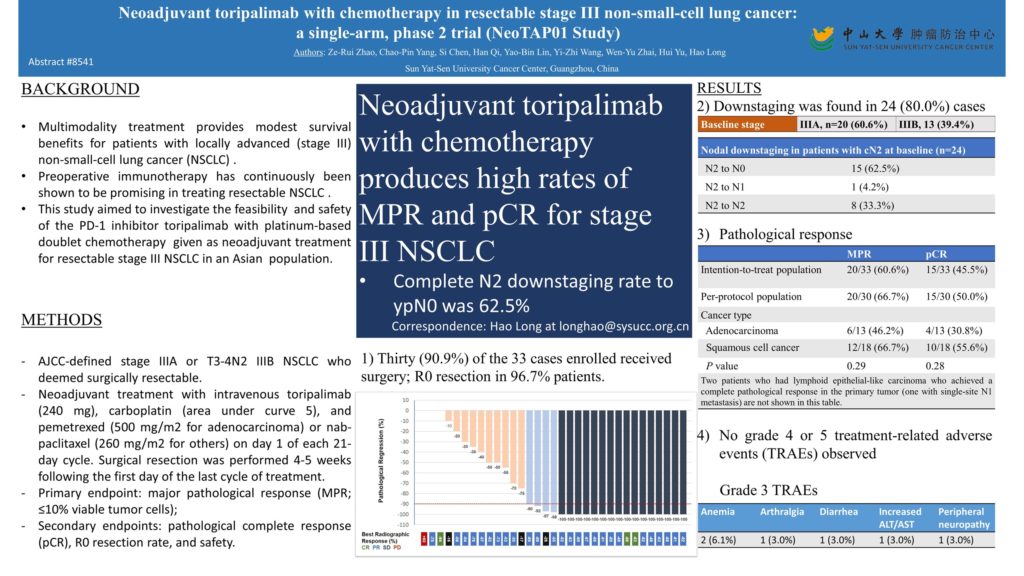
8541 – Phase II trial of toripalimab plus chemotherapy as neoadjuvant treatment in resectable stage III non-small cell lung cancer (NeoTPD01 Study)
Authors: Zerui Zhao, Si Chen, Han Qi, Chao-Pin Yang, Yao-Bin Lin, Jie-Tian Jin, Shan-Shan Lian, Yi-Zhi Wang, Hui Yu, Long Hao
Organizations: Department of Thoracic Surgery, Sun Yat-Sen University Cancer Center, Guangzhou, VA, China, Department of Thoracic Surgery, Sun Yat-Sen University Cancer Center, Guangzhou, China, Department of Minimally Invasive Interventional Therapy, Sun Yat-Sen University Cancer Center, Guangzhou, China, Department of Biotherapy, Sun Yat-Sen University Cancer Center, Guangzhou, China, Department of Pathology, Sun Yat-Sen University Cancer Center, Guangzhou, China, Guangzhou, China, Department of Radiology, Sun Yat-Sen University Cancer Center, Guangzhou, China, Guangzhou, China
Research Funding: None
Background: Multi-modality treatment provides modest survival benefits for locally advanced non-small-cell lung cancer (NSCLC) patients. Preoperative immunotherapy has continuously been shown to be promising in treating resectable NSCLC. The current study aimed to investigate the activity and safety of the PD-1 inhibitor, toripalimab, with chemotherapy given as neoadjuvant treatment for resectable stage III NSCLC in Asian population.
Methods: Eligible patients recruited were aged 18 years or older with histologically confirmed AJCC-defined stage IIIA or T3-4N2 IIIB NSCLC who deemed surgically resectable. Patients received 3 cycles of neoadjuvant treatment with intravenous toripalimab (240 mg), carboplatin (area under curve 5), and pemetrexed (500 mg/m2 for adenocarcinoma) or nab-paclitaxel (260 mg/m2 for others) on day 1 of each 21-day cycle. Surgical resection was performed 4-5 weeks following the first day of the last cycle of treatment. The primary endpoint was major pathological response (MPR; ≤10% viable tumor cells). Secondary endpoints included pathological complete response (pCR), R0 resection rate, disease-free survival and safety. Paired primary tumor +/- lymph node and blood samples at baseline and surgery were obtained for exploratory study. This study is registered with ClinicalTrials.gov, NCT04304248.
Results: Between August 2019 and July 2020, 33 patients (median age: 61, IQR: 56-66; female: 6, 18.2%) were enrolled and received neoadjuvant treatment. 18 (54.5%) patients had squamous cell lung cancer, and 13 (39.4%) had T3-4N2 stage IIIB disease. Two patients refused surgery and one had progressive disease after treatment. 30 (91.9%) patients underwent resection (median interval between neoadjuvant treatment and surgery: 36.5 days, IQR 30-42.5) and all except one achieved R0 resection (29/30, 96.7%). 20 patients (20/30, 66.7%) had an MPR, including 15 patients (15/30, 50.0%) had a pCR in the per-protocol population. Surgical complications included three arrhythmias, one prolonged air leak, and one chylothorax. 24 patients (80.0%) had pathological downstaging following treatment, and complete lymph node clearances (ypN0) were seen in 70.0% (21/30) of patients. The most common grade 3-4 treatment-related adverse events in the intention-to-treat population were anemia (2, [6.0%]). Severe treatment-related adverse event included one (3.0%) patient with grade 3 peripheral neuropathy (Guillain-Barré syndrome) and resulted in surgery cancellation. At the time of data cutoff (Feb 7, 2021), the median duration of follow-up was 4.13 months, and there were no treatment-related deaths.
Conclusions: Toripalimab plus platinum-based doublet yields a high MPR rate, manageable treatment-related toxicity, and feasible surgical resection in resectable stage III NSCLC. Ongoing analysis of biomarker will be available at the meeting. Clinical trial information: NCT04304248
4083 – A phase II trial of lenvatinib plus toripalimab and hepatic arterial infusion chemotherapy as a first-line treatment for advanced hepatocellular carcinoma (LTHAIC study)
Authors: MinKe He, Shi Ming, Zhicheng Lai, QiJiong Li
Organizations: Department of Hepatobiliary Oncology, Sun Yat-sen University Cancer Center, State Key Laboratory of Oncology in South China, Collaborative Innovation Center for Cancer Medicine, Guangzhou, China, Department of Hepatobiliary Oncology, Sun Yat-sen University Cancer Center, State Key Laboratory of Oncology in South China, Collaborative Innovation Center for Cancer Medicine, Guangzhou, VA, China
Research Funding: None
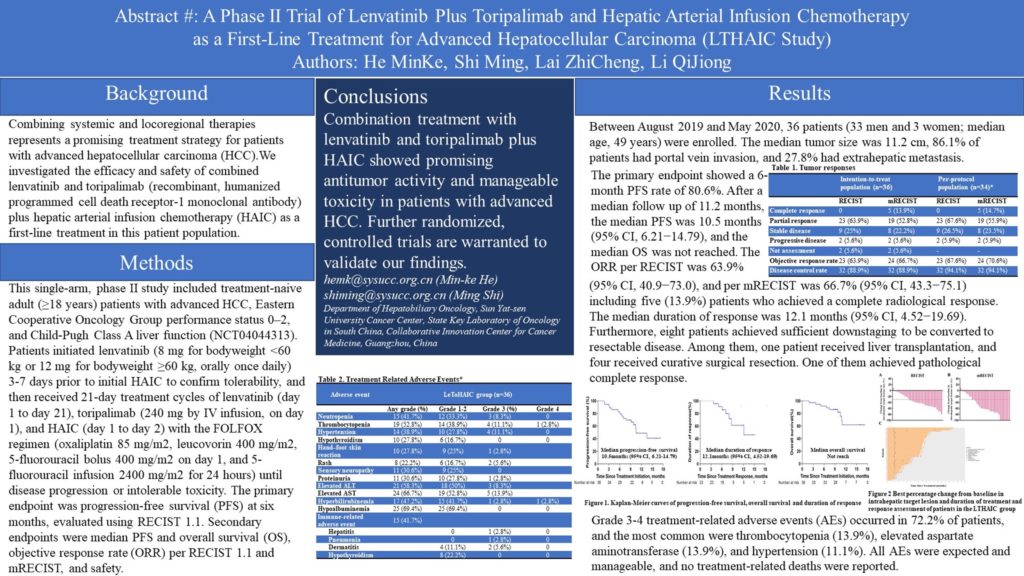
Background: Combining systemic and locoregional therapies represents a promising treatment strategy for patients with advanced hepatocellular carcinoma (HCC). We investigated the efficacy and safety of combined lenvatinib and toripalimab (recombinant, humanized programmed cell death receptor-1 monoclonal antibody) plus hepatic arterial infusion chemotherapy (HAIC) as a first-line treatment in this patient population.
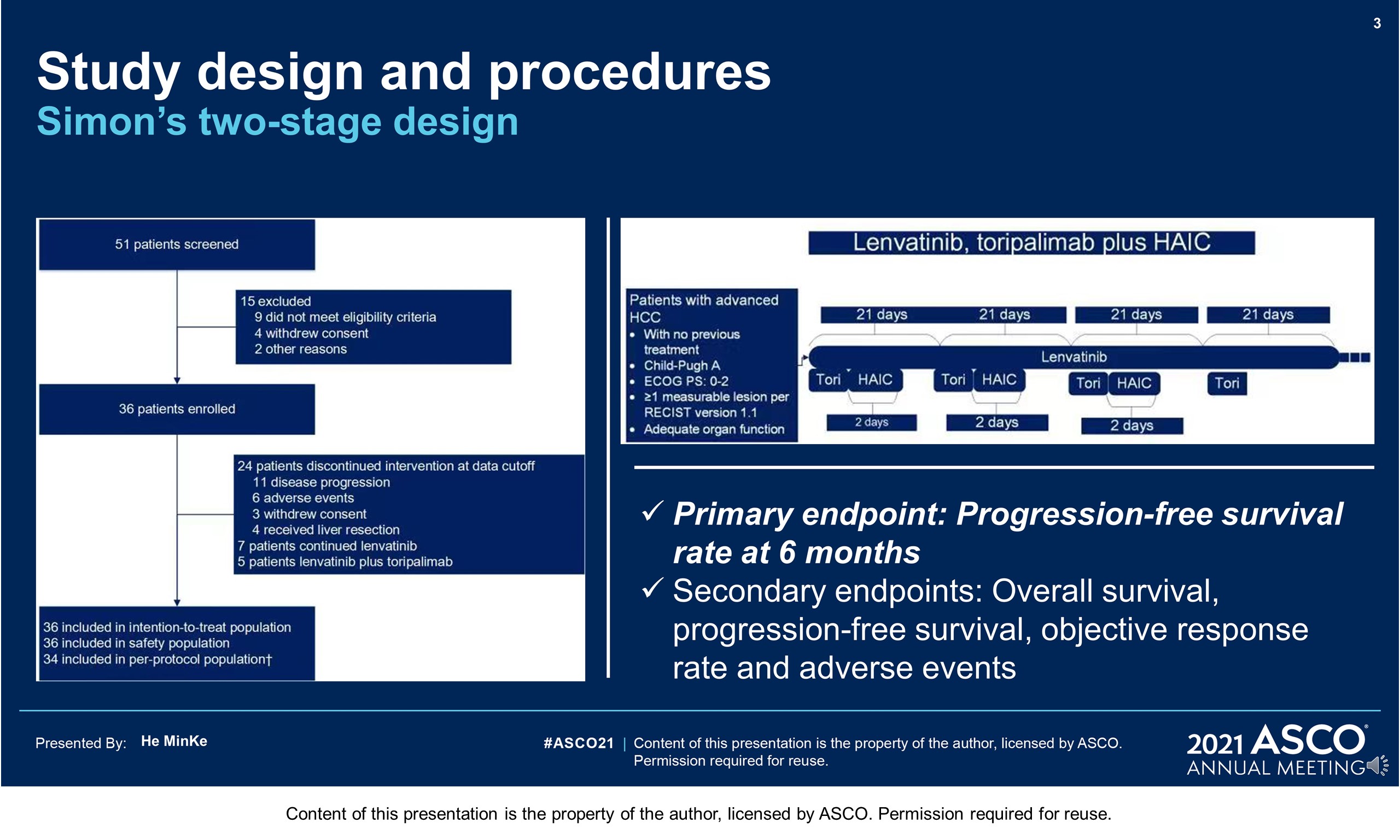
Methods: This single-arm, phase II study included treatment-naive adult (≥18 years) patients with advanced HCC, Eastern Cooperative Oncology Group performance status 0–2, and Child-Pugh Class A liver function (NCT04044313). Patients initiated lenvatinib (8 mg for bodyweight < 60 kg or 12 mg for bodyweight ≥60 kg, orally once daily) 3-7 days prior to initial HAIC to confirm tolerability, and then received 21-day treatment cycles of lenvatinib (day 1 to day 21), toripalimab (240 mg by IV infusion, on day 1), and HAIC (day 1 to day 2) with the FOLFOX regimen (oxaliplatin 85 mg/m2, leucovorin 400 mg/m2, 5-fluorouracil bolus 400 mg/m2 on day 1, and 5-fluorouracil infusion 2400 mg/m2 for 24 hours) until disease progression or intolerable toxicity. The primary endpoint was progression-free survival (PFS) at six months, evaluated using RECIST 1.1. Secondary endpoints were median PFS and overall survival (OS), objective response rate (ORR) per RECIST 1.1 and mRECIST, and safety.
Results: Between August 2019 and May 2020, 36 patients (33 men and 3 women; median age, 49 years) were enrolled. The median tumor size was 11.2 cm, 86.1% of patients had portal vein invasion, and 27.8% had extrahepatic metastasis. The primary endpoint showed a 6-month PFS rate of 80.6%. After a median follow up of 11.2 months, the median PFS was 10.5 months (95% CI, 6.21−14.79), and the median OS was not reached. The ORR per RECIST was 63.9% (95% CI, 40.9−73.0), and per mRECIST was 66.7% (95% CI, 43.3−75.1) including five (13.9%) patients who achieved a complete radiological response. The median duration of response was 12.1 months (95% CI, 4.52−19.69). Furthermore, eight patients achieved sufficient downstaging to be converted to resectable disease. Among them, one patient received liver transplantation, and four received curative surgical resection. One of them achieved pathological complete response. Grade 3-4 treatment-related adverse events (AEs) occurred in 72.2% of patients, and the most common were thrombocytopenia (13.9%), elevated aspartate aminotransferase (13.9%), and hypertension (11.1%). All AEs were expected and manageable, and no treatment-related deaths were reported.
Conclusions: Combination treatment with lenvatinib and toripalimab plus HAIC showed promising antitumor activity and manageable toxicity in patients with advanced HCC. Further randomized, controlled trials are warranted to validate our findings. Clinical trial information: NCT04044313
9570 – A phase Ib clinical trial of neoadjuvant OrienX010, an oncolytic virus, in combination with toripalimab in patients with resectable stage IIIb to stage IVM1a acral melanoma
Authors: Xuan Wang, Chuanliang Cui, Lu Si, Caili Li, Jie Dai, Lili Mao, Xue Bai, Zhihong Chi, Xinan Sheng, Yan Kong, Bin Lian, Bixia Tang, Xieqiao Yan, Li Zhou, Siming Li, Robert H.I. Andtbacka, Jun Guo
Organizations: Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Renal Cancer and Melanoma, Peking University Cancer Hospital & Institute, Beijing, China, Department of Renal Cancer and Melanoma, Peking University Cancer Hospital & Institute, Beijing, China, Huntsman Cancer Institute at the University of Utah, Salt Lake City, UT
Research Funding: Oriengene Biotechnology Ltd
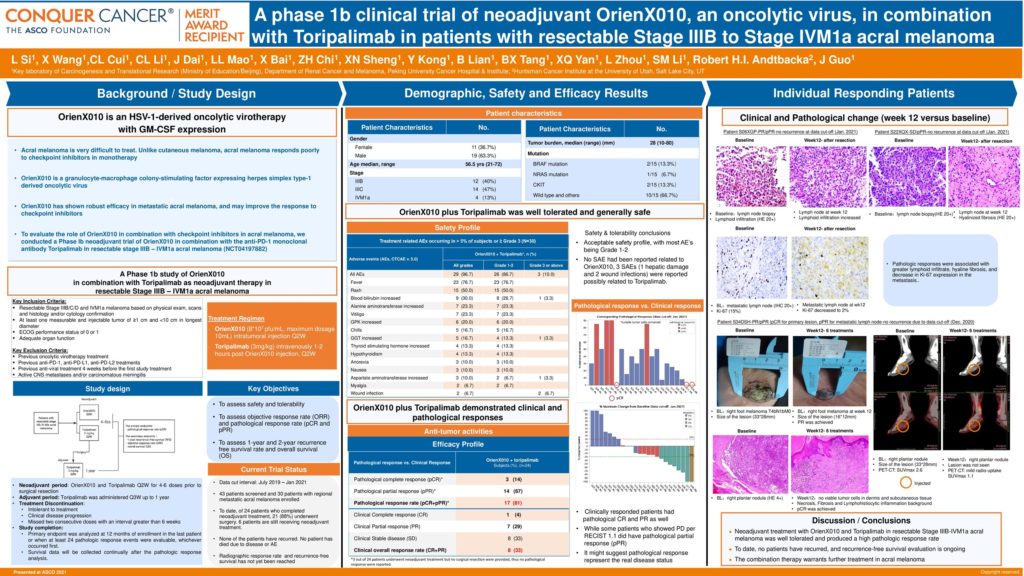
Background: Metastatic acral melanoma is very difficult to treat. Unlike cutaneous melanoma, acral melanoma responds poorly to checkpoint inhibitors in monotherapy. OrienX010 is a granulocyte-macrophage colony-stimulating factor expressing herpes simplex type-1 derived oncolytic virus. It has shown robust efficacy in metastatic acral melanoma, and may improve the response to checkpoint inhibitors. To evaluate the role of OrienX010 in combination with checkpoint inhibitors in acral melanoma, we conducted a Phase Ib neoadjuvant trial of OrienX010 in combination with the anti-PD-1 monoclonal antibody toripalimab in resectable stage IIIB-IVM1a acral melanoma (NCT04197882).
Methods: Patients with resectable stage IIIB-IV M1a acral melanoma received neoadjuvant intratumoral OrienX010 up to 10 mL of 8 x 107 pfu/mL and intravenous toripalimab 3 mg/kg every 2 weeks for 4 – 6 doses prior to surgical resection. After resection, adjuvant toripalimab 3 mg/kg was administered every 3 weeks for up to 1 year. The primary endpoints were radiographic response rate per RECIST 1.1 and pathological response rate (pCR and pPR). The secondary endpoints were 1- and 2-year recurrence-free survival, and safety.
Results: Between July 2019 and Jan 2021, 30 patients with regional metastatic acral melanoma were enrolled. Median age was 56.5 years, 14 (47%) were male, 19 (63%) had recurrent disease, and stage IIIB 12 (40%), IIIC 14 (47%), and IVM1a 4 (13%). Median tumor burden was 28mm (range, 10-80mm), and only 5 (17%) patients had melanoma mutations (2 cKIT, 1 NRAS, 2 BRAF). To date, of 24 patients who completed neoadjuvant treatment, 21 (88%) underwent surgery. Three (12%) patients did not undergo surgery due to disease progression prior to surgery and 6 patients are still receiving neoadjuvant treatment. Radiographic responses were seen in 10 (33%) patients. However, 17 of 21 (81%) patients showed pathologic responses in resected metastases, with 3 (14%) showing a pCR and 14 (67%) a pPR. Pathologic responses were associated with greater lymphoid infiltrate, hyaline fibrosis, and decrease in Ki-67 expression in the metastasis. At a median follow-up of 8.9 months, none of the patients who underwent resection have recurred. The neoadjuvant treatment was well tolerated, with all patients experiencing at least 1 treatment related adverse event (TRAE) and Grade 1 fever was most common. Three (10%) patients had a grade 3-4 TRAE, including 1 alanine aminotransferase increase and 2 wound infections.
Conclusions: Neoadjuvant treatment with OrienX010 and toripalimab in resectable stage IIIB-IVM1a acral melanoma was well tolerated and produced a high pathologic response rate. To date, no patients have recurred, and recurrence-free survival evaluation is ongoing. This combination therapy warrants further evaluation in acral melanoma. Clinical trial information: NCT04197882
2577 – A first-in-human phase I dose escalation of YH001, an anti-CTLA-4 monoclonal antibody (mAb) in combination with toripalimab (anti-PD-1 mAb) in patients with advanced solid tumors
Authors: Vinod Ganju, Adam Cooper, Bo Gao, Kate Wilkinson
Organizations: Peninsula and Southeast Oncology, Frankston, VIC, Australia, St George Private Hospital, Kogarah, NSW, Australia, Blacktown and Westmead Hospital, Blacktown, NSW, Australia, Sydney Southwest Private Hospital & St George Private Hospital, Liverpool, NSW, Australia
Research Funding: Eucure (Beijing) Biopharma Co., Ltd
Background: YH001 is a humanized anti -hCTLA-4 IgG1 mAb that relieves CTLA-4-mediated immunosuppression, and thereby enhances the T-cell-mediated antitumor immune response. Pre-clinical data have shown potent anti-cancer activity when combined with anti-PD-1 mAb.
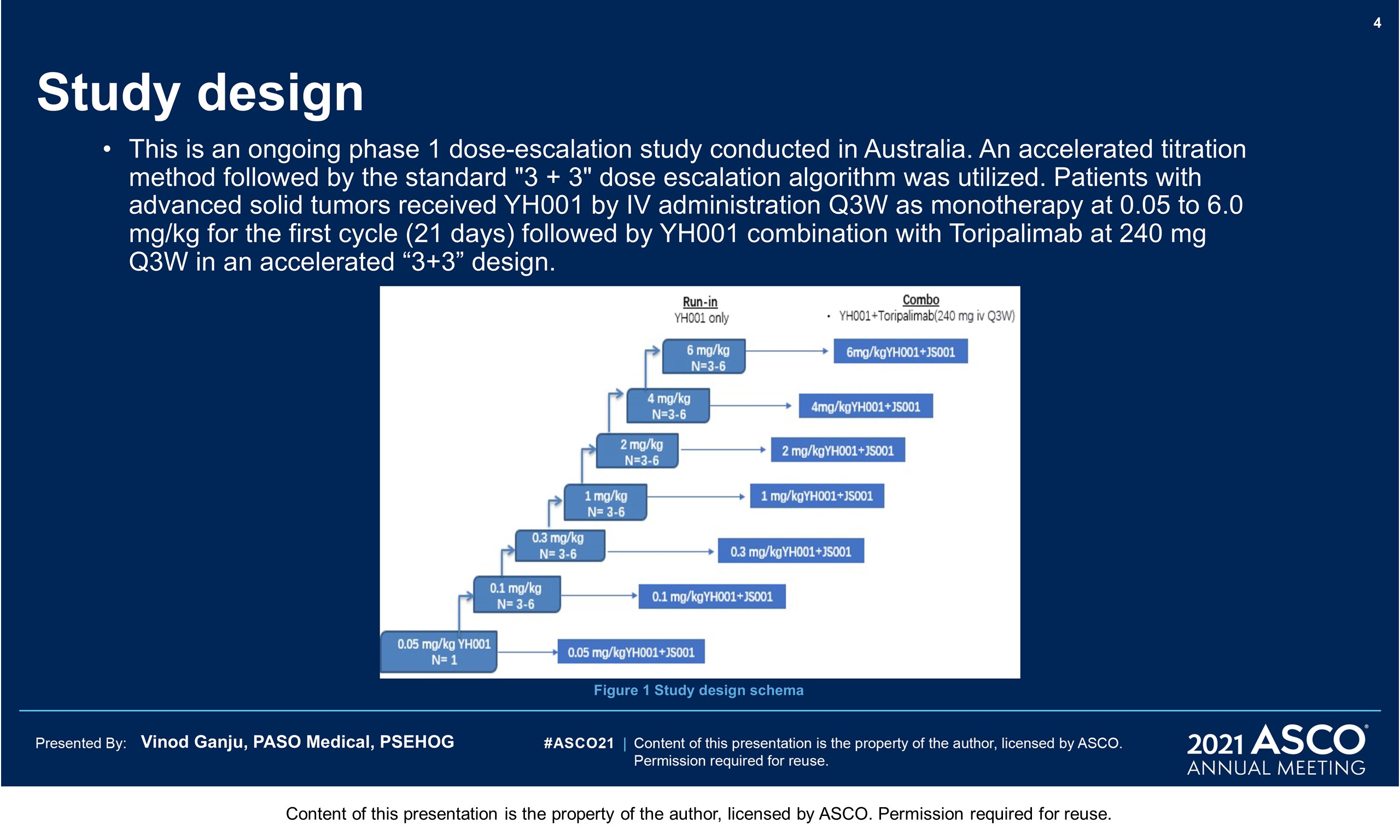
Methods: This is an ongoing phase 1 dose-escalation study. Patients (pts) with advanced solid tumors received YH001 by IV administration at 0.05 to 6.0 mg/kg for 1 cycle (21 days) then in combination with Toripalimab (anti-PD-1 mAb) at 240 mg Q3W for 4 cycles. An accelerated titration method followed by the standard “3+3” design was utilized to evaluate safety, tolerability and preliminary efficacy.
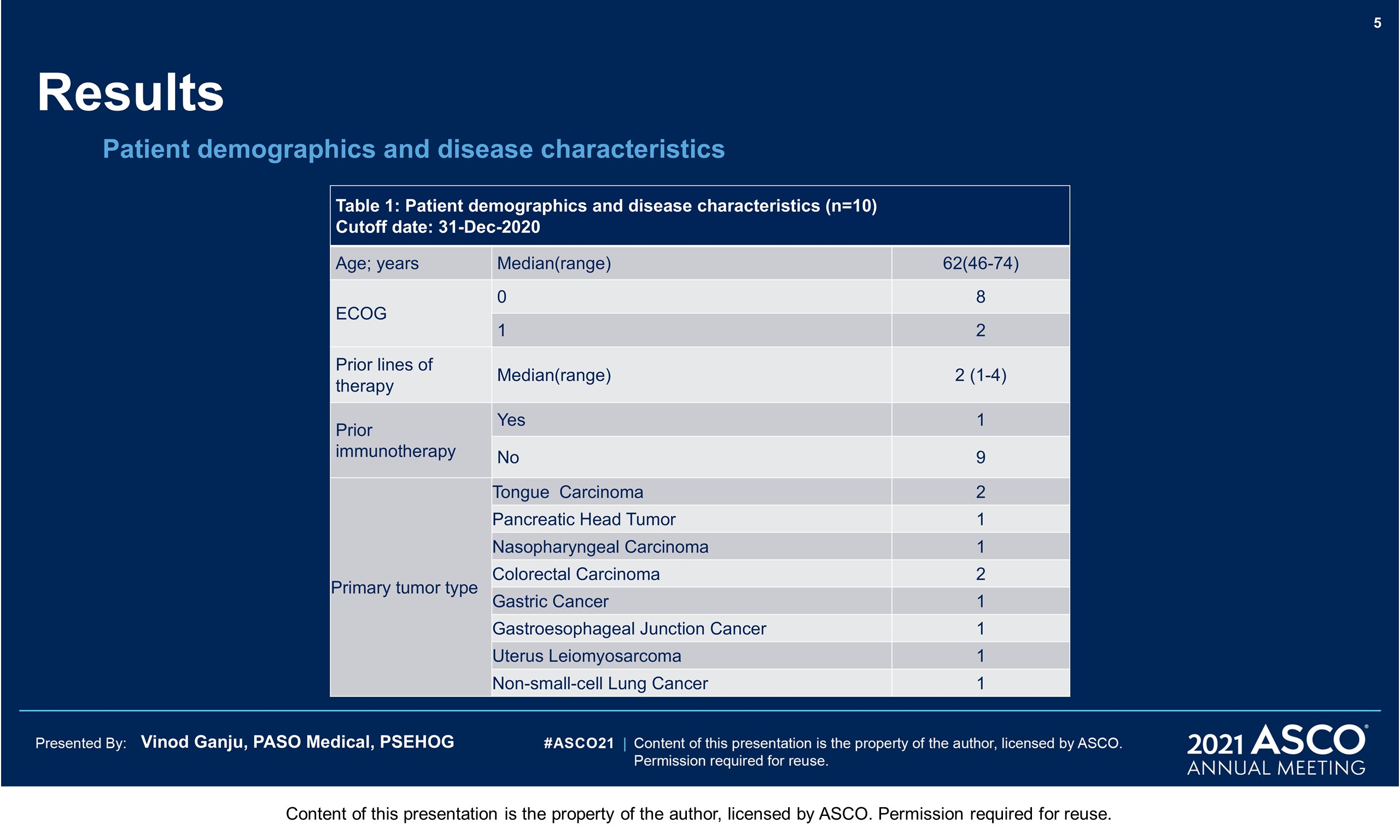
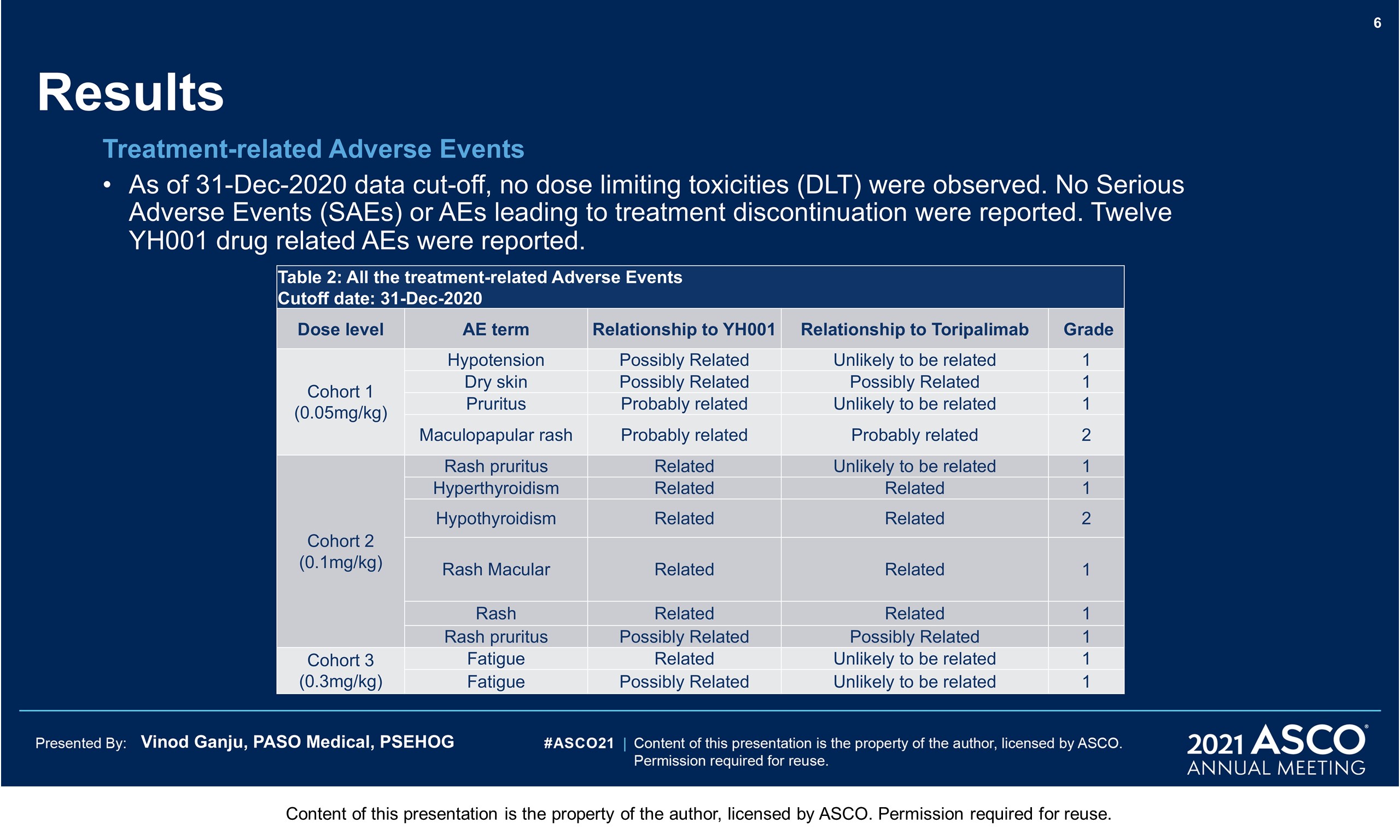
Results: As of 31-Dec-2020 data cut-off, 10 pts were enrolled and treated at 0.05 mg/kg (n = 2), 0.1 mg/kg (n = 3), 0.3 mg/kg (n = 3) and 1 mg/kg (n = 2). The median age was 62 years (range 46-74). Baseline ECOG scores were 0 (n = 8), 1(n = 2) with all pts progressed after a median of 2 prior lines of available standard therapy (range 1-4) including 1 pt progressed after immunotherapy of pembrolizumab. There were no dose limiting toxicities (DLT) observed. No severe adverse events (SAEs), Grade (G) 3 or above adverse events (AEs) and AEs leading to treatment discontinuation were reported. Twelve drug related AEs were all G1/2 events including 2 G2 AEs (1 rash maculopapular at 0.05mg/kg, 1 hypothyroidism at 0.1mg/kg), 10 G1 AEs (1 hypotension, 1 dry skin, 1 pruritus at 0.05mg/kg; 1 rash, 1 rash macular, 1 hyperthyroidism, 2 rash pruritus at 0.1mg/kg, 2 fatigues at 0.3mg/kg). Among 7 patients having imaging tumor assessment by RECIST v1.1, there were 4 SD, including 1 at 0.05 mg/kg with tongue carcinoma at week 8 assessment, 1 at 0.1 mg/kg with nasopharyngeal carcinoma at week 8 and 15 assessment, 2 at 0.3 mg/kg with gastroesophageal junction cancer and uterus leiomyosarcoma at week 8.
Conclusions: YH001 combined with Toripalimab is safe and tolerable up to 1 mg/kg dose level. Updated safety and preliminary efficacy data will be presented. Clinical trial information: NCT04481009
4094 – Gemox chemotherapy in combination with anti-PD1 antibody toripalimab and lenvatinib as first-line treatment for advanced intrahepatic cholangiocarcinoma: A phase 2 clinical trial
Authors: Zhou Jian, Jia Fan, Guo-Ming Shi, Xiao-Yong Huang, Dong Wu, Guo-Huan Yang, Yuan Ji, Yi Chen, Fei Liang, Jia-Cheng Lu, Ning-Ling Ge, Hui-Chuan Sun, Xiao-Wu Huang, Shuang-Jian Qiu, Yi-Feng He, Xin-Rong Yang, Yang Xu, Qiang Gao, Jian Sun
Organizations: Department of Liver Surgery and Transplantation, Liver Cancer Institute, Zhongshan Hospital, Fudan University, Shanghai, China, Fudan University ZhongShan Hospital, Shanghai, China, Fudan University, Shanghai, China, Zhongshan Hospital, Fudan University, Shanghai, China, Department of Liver Surgery, Zhongshan Hospital, Fudan University, Shanghai, China, Department of Pathology, Zhongshan Hospital, Fudan University, Shanghai, China, Zhongshan Hospital, Shanghai, China, Department of Statistics, Zhongshan Hospital, Fudan University, Shanghai, China, Department of Hepatic Oncology, Liver Cancer Institute and Zhongshan Hospital, Fudan University, Shanghai, China, Liver Cancer Institute and Zhongshan Hospital, Fudan University, Shanghai, China, Zhongshan Hospital Liver Cancer Institute Fudan University, Shanghai, China
Research Funding: None
Background: The outcome of advanced intrahepatic cholangiocarcinoma (ICC) remains poor with current gemcitabine-based chemotherapy. This study is to evaluate the safety and efficacy of anti-PD1 agent toripalimab, lenvatinib in combination with oxaliplatin and gemcitabine (Gemox) chemotherapy.
Methods: Locally advanced or metastatic ICC patients (pts) were given 240 mg toripalimab Q3W via intravenous (IV) infusion, 8 mg lenvatinib QD orally, and 1g/m² gemcitabine on Day 1 and Day 8, and 85 mg/m² oxaliplatin Q3W by IV for 6 cycles. The primary outcome was objective response rate (ORR), which was evaluated according to RECIST v1.1. Secondary outcomes included safety, progression-free survival (PFS) and overall survival (OS). Treatment would be terminated by confirmed disease progression, unacceptable toxicity, or voluntary withdrawal. Whole exome sequencing was performed on available tumor tissues and PD-L1 expression was determined by immunohistochemistry staining.
Results: From May 2019 to Oct 2019, 30 pathologically-confirmed advanced ICC pts with a mean age of 56.5 (range, 25-73) years, including 11 women (37%), were enrolled at Zhongshan Hospital, Fudan University (one pt withdrawn). At the end of last follow-up (February 1, 2021), the ORR was 80% (24/30; 95% CI: 61.4%-92.3%), and disease control rate (DCR) was 93.3% (28/30; 95% CI:77.9%-99.2%). Median follow-up was 16.6 months. One pt achieved complete response (CR). Three pts with locally advanced disease were down-staged and then underwent resection. They remained disease-free survival at the end of last follow-up. 23 pts experienced disease progression and 12 pts (including one pt withdrawn) have died. The median PFS was 10.0 months and median duration of response (DOR) was 9.8 months. The median OS have not been reached. 12-months OS rate was 73.3% (95% CI: 57.5%-89.2%). No grade 5 adverse event (AE) was observed in present study. Grade 3 or 4 neutropenia and thrombocytopenia observed in 3 (10%) and 1 (3.3%) patient, respectively. Non-hematological toxic effects were jaundice (10 %), rash (6.7 %), proteinuria (6.7 %), increased AST level (3.3%), vomiting (3.3%), upper gastrointestinal hemorrhage (3.3%), sepsis (3.3%), gastrointestinal fistula (3.3%), adrenocortical insufficiency (3.3%), interstitial pneumonia (3.3%), and hyponatremia (3.3%). High ORR was significantly associated with positive PD-L1 expression (p= 0.048) and DNA damage repair (DDR)-related mutations (p= 0.022) in tumor samples.
Conclusions: Gemox chemotherapy in combination with Anti-PD1 antibody Toripalimab and Lenvatinib provided remarkable efficacy with reasonable tolerability in advanced ICC patients. These findings warrant further validation in a large randomized clinical trial. Clinical trial information: NCT03951597
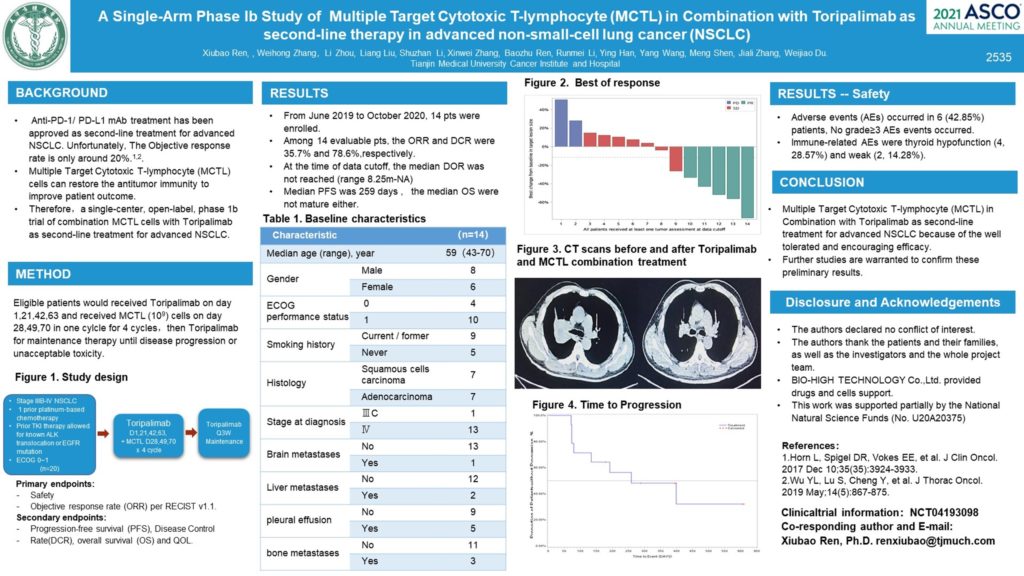
2535 – GA single-arm phase Ib study of multiple target cytotoxic T-lymphocyte (MCTL) in combination with toripalimab as second-line therapy in advanced non-small cell lung cancer (NSCLC)
Authors: Xiubao Ren, Weihong Zhang
Organizations: Department of Biological Therapy, Tianjin Cancer Institute & Hospital, Tianjin, China, Tianjin Medical University Cancer Institute and Hospital, Tianjin, China
Research Funding: National Natural Science Foundation of China grants Awards No. U20A20375
Background: Anti-PD-1/ PD-L1 (programmed cell-death 1) mAb treatment has been approved in the US and in Europe as second-line treatment for advanced NSCLC because of the good tolerance and efficacy in comparison with docetaxel. Unfortunately, The objective response rate is only around 20%. Multiple Target Cytotoxic T-lymphocyte (MCTL) cells can restore the antitumor immunity to improve patient outcome. Combining MCTL cells with anti-PD-1 mAb may strengthen the results as second-line treatment in patients with advanced NSCLC. (NCT04193098).
Methods: This is a single-center, open-label, phase 1b trial of combination MCTL cells with toripalimab (anti-PD-1 mAb)as second-line treatment for advanced NSCLC. Systemic therapy patients received toripalimab every 3 weeks for 12 cycles and received MCTL cells every 3 weeks for 9 cycles, then toripalimab and MCTL cells for maintenance therapy until disease progression or unacceptable toxicity.
Results: From June 2019 to October 2020, 14 pts aged 43-70 years (median age 59 years) were enrolled. The squamous/non-squamous ratio was 50%/50%. 8 (57.1%) were men, 13(92.8%) were ECOG PS=0-1, 5 (35.7%) had pleural effusion, and 3 (21.4%) had bone metastases. Among 13 evaluable pts, the ORR and DCR were 38.4% and 71.4%, At the time of data cutoff, the median DOR was not reached (range 8.25m-NA), the median PFS was 399 days (range 192d-NA), and the median OS were not mature. Adverse events (AEs) occurred in 5 (38.4%), No grade≥3 AEs events occurred. Immune-related AEs were thyroid hypofunction (3, 23%) and weak (2, 15.4%). Biomarkers which correlated with efficacy and AEs are being analyzed.
Conclusions: Multiple Target Cytotoxic T-lymphocyte (MCTL) in combination with toripalimab as second-line treatment for advanced NSCLC because of the well tolerated and encouraging efficacy. Further studies are warranted to confirm these results. Research Sponsor: Tianjin Medical University Cancer Institute and Hospital. Clinical trial information: NCT04193098
Disclaimer
This content was selected by and is provided courtesy of Coherus BioSciences.
ASCO® and American Society of Clinical Oncology® are registered trademarks of the American Society of Clinical Oncology, Inc. Used with permission.
This presentation does not necessarily represent a balanced view or full discussion of any given subject. The ideas and opinions expressed herein do not necessarily reflect those of ASCO. The authors, editors, and ASCO are not responsible for errors or omissions in translations. The mention of any company, product, service, or therapy in this collection of materials does not constitute an endorsement of any kind by ASCO. It is the responsibility of the treating physician or other health care provider, relying on independent experience and knowledge of the patient, to determine drug dosages and the best treatment for the patient. Viewers are advised to check the appropriate medical literature and the product information currently provided by the manufacturer of each drug to be administered to verify, among other matters, the dosage, method, and duration of administration, or contraindications. Viewers are also encouraged to contact the manufacturer with questions about the features or limitations of any products. ASCO assumes no responsibility for any injury or damage to persons or property arising out of or related to any use of the material contained in this publication or to any errors or omissions.
Site provided by Springer Healthcare Limited. Part of the Springer Nature Group.