141 – Survival Analysis of Newly Diagnosed Transplant-Eligible Multiple Myeloma Patients in the Randomized Forte Trial
Author(s): Francesca Gay, MD, Pellegrino Musto, Delia Rota Scalabrini*, Monica Galli*, Angelo Belotti, MD*, Elena Zamagni, MD*, Luca Bertamini, MD*, Renato Zambello*, Micol Quaresima*, Giovanni De Sabbata*, Giuseppe Pietrantuono*, Mattia D’Agostino, MD*, Daniela Oddolo*, Andrea Capra*, Anna Marina Liberati*, Salvatore Palmieri*, Franco Narni, Massimo Offidani, Michele Cavo* and Mario Boccadoro
GIMEMA, European Myeloma Network, Italy
Disclosures: Gay: Celgene: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Bristol-Myers Squibb: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Roche: Membership on an entity’s Board of Directors or advisory committees; AbbVie: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Adaptive Biotechnologies: Membership on an entity’s Board of Directors or advisory committees; Oncopeptides: Membership on an entity’s Board of Directors or advisory committees; GSK: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Amgen: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Takeda: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity’s Board of Directors or advisory committees. Musto: Celgene: Honoraria; Amgen: Honoraria. Galli: BMS: Honoraria; Celgene: Honoraria; Janssen: Honoraria; Takeda: Honoraria. Belotti: Jannsen: Membership on an entity’s Board of Directors or advisory committees; Amgen: Membership on an entity’s Board of Directors or advisory committees; Celgene: Membership on an entity’s Board of Directors or advisory committees. Zamagni: Sanofi: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses, Speakers Bureau; BMS: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses, Speakers Bureau; Amgen: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; Takeda: Honoraria, Other: Travel, Accommodations, Expenses, Speakers Bureau; Janssen: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses, Speakers Bureau; Celgene Corporation: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau. Zambello: Janssen: Membership on an entity’s Board of Directors or advisory committees; Celgene: Membership on an entity’s Board of Directors or advisory committees. De Sabbata: Celgene: Membership on an entity’s Board of Directors or advisory committees; Amgen: Membership on an entity’s Board of Directors or advisory committees. D’Agostino: GSK: Membership on an entity’s Board of Directors or advisory committees. Liberati: VERASTEM: Honoraria, Research Funding; ROCHE: Honoraria, Research Funding; PFIZER: Honoraria, Research Funding; ONCOPEPTIDES AB: Honoraria, Research Funding; TAKEDA: Honoraria, Research Funding; MORPHOSYS: Honoraria, Research Funding; ONCONOVA: Honoraria, Research Funding; ABBVIE: Honoraria, Research Funding; NOVARTIS: Honoraria, Research Funding; KARYOPHARM: Honoraria, Research Funding; INCYTE: Honoraria; JANSSEN: Honoraria; CELGENE: Honoraria; AMGEN: Honoraria; BMS: Honoraria; BEIGENE: Honoraria; ARCHIGEN: Honoraria; BIOPHARMA: Honoraria; FIBROGEN: Honoraria. Offidani: Janssen: Consultancy, Honoraria; BMS: Consultancy, Honoraria; Celgene: Consultancy, Honoraria. Cavo: AbbVie: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; GlaxoSmithKline: Honoraria, Speakers Bureau; Sanofi: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; BMS: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; Celgene: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Travel accomodations, Speakers Bureau; Novartis: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; Karyopharm: Honoraria; Amgen: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; Janssen: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Travel accomodations, Speakers Bureau. Boccadoro: AbbVie: Honoraria; Bristol-Myers Squibb: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Janssen: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Research Funding; Celgene: Honoraria, Research Funding; GlaxoSmithKline: Membership on an entity’s Board of Directors or advisory committees; Mundipharma: Research Funding; Amgen: Honoraria, Research Funding; Sanofi: Honoraria, Research Funding.
OffLabel Disclosure: The presentation includes discussion of off-label use of a drug or drugs for the treatment of multiple myeloma (including carfilzomib, cyclophosphamide, lenalidomide and dexamethasone).
Background. Proteasome inhibitor (PI)-based induction/consolidation proved to be effective in newly diagnosed multiple myeloma (NDMM) patients (pts) eligible for melphalan 200 mg/m2 plus autologous stem-cell transplantation (MEL200-ASCT). High response rates have been reported with carfilzomib (K) plus lenalidomide-dexamethasone (KRd) or cyclophosphamide-dexamethasone (KCd). Lenalidomide (R) alone is a standard of care for post-ASCT maintenance; K maintenance showed promising results in phase I/II studies, but no data on KR maintenance vs R are available.
Aims. The aims of this analysis were to evaluate the progression-free survival (PFS) of KRd induction-ASCT-KRd consolidation (KRd_ASCT) vs 12 cycles of KRd (KRd12) vs KCd induction-ASCT-KCd consolidation (KCd_ASCT) and the PFS of KR vs R maintenance. Secondary aims were efficacy in different subgroups of pts and safety of the maintenance phase.
Methods. NDMM pts ≤65 years were randomized [R1: 1:1:1, stratification International Staging System (ISS) and age] to: KRd_ASCT: 4 28-day cycles with KRd induction (K 20/36 mg/m2 IV days 1,2,8,9,15,16; R 25 mg days 1-21; dexamethasone [d] 20 mg days 1,2,8,9,15,16) followed by MEL200-ASCT and 4 KRd consolidation cycles; KRd12: 12 KRd cycles; KCd_ASCT: 4 28-day induction cycles with KCd (K 20/36 mg/m2 IV days 1,2,8,9,15,16; cyclophosphamide 300 mg/m2 days 1,8,15; d 20 mg days 1,2,8,9,15,16) followed by MEL200-ASCT and 4 KCd consolidation cycles. Thereafter, pts were randomized (R2) to maintenance with KR (K 36 mg/m2 days 1,2,15,16, subsequently amended to 70 mg/m2 days 1,15 for up to 2 years; plus R 10 mg days 1-21 every 28 days until progression) or R alone (10 mg days 1-21 every 28 days until progression). Centralized minimal residual disease (MRD) evaluation (8-color second-generation flow cytometry, sensitivity 10-5) was performed in pts achieving ≥very good partial response before maintenance and every 6 months (m) during maintenance. Data cut-off was June 30, 2020.
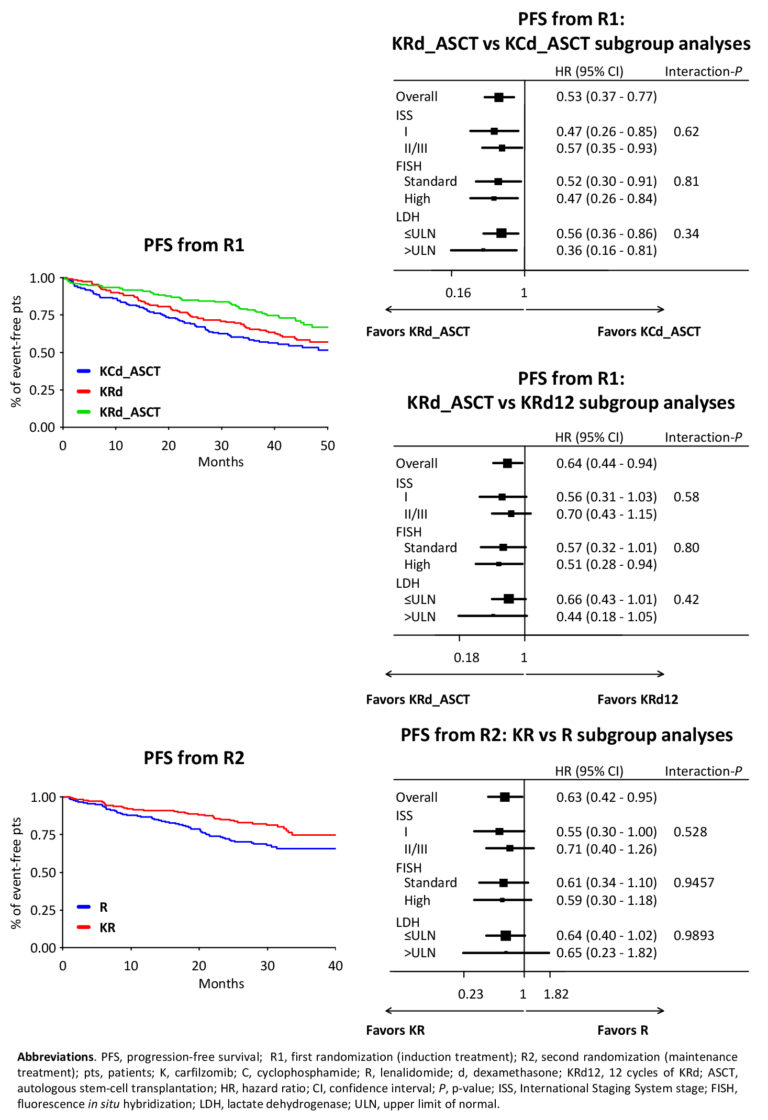
Results. 474 NDMM pts were randomized (KRd_ASCT, n=158; KRd12, n=157; KCd_ASCT, n=159) and analyzed. Pt characteristics were well balanced. Intention-to-treat (ITT) data of pre-maintenance MRD (KRd_ASCT, 62%; KRd12 56%, KCd_ASCT 43%) and safety of the induction/consolidation phases in the 3 arms were already reported (F. Gay et al. ASH 2018; S. Oliva et al. ASH 2019). After a median follow-up from R1 of 45 m, median PFS was not reached with KRd_ASCT, 57 m with KRd12 and 53 m with KCd_ASCT (KRd_ASCT vs KCd_ASCT: HR 0.53, P<0.001; KRd_ASCT vs KRd12: HR 0.64, P=0.023; KRd12 vs KCd_ASCT: HR 0.82, P=0.262). The benefit of KRd_ASCT vs both KCd_ASCT and KRd12 was observed in most subgroups (Figure). 3-year overall survival (OS) was 90% with KRd_ASCT and KRd12 vs 83% with KCd. 356 pts (KR, n=178; R, n=178) were randomized to maintenance; pt characteristics, pre-maintenance response (≥complete response [CR]: KR 62% vs R 59%; stringent CR: KR 50% vs R 48%) including MRD negativity (KR 65% vs R 66%) in the 2 groups were well balanced. After a median follow-up from R2 of 31 m and a median duration of maintenance of 27 m in both arms, 46% of MRD-positive pts at randomization turned negative in KR vs 32% in R (P=0.04). By ITT analysis, 3-year PFS from R2 was 75% with KR vs 66% with R (HR 0.63; P=0.026). The benefit of KR vs R was observed in most subgroups (Figure). 3-year OS was 90% in both arms.
During maintenance, a similar proportion of pts experienced ≥1 grade (G)3-4 hematologic adverse events (AEs)/serious AEs (SAEs) in the 2 arms (KR 22% vs R 23%); the most frequent were neutropenia (KR 18% vs R 21%) and thrombocytopenia (KR 3% vs R 3%). Rate of ≥1 G3-4 non-hematologic AEs/SAEs was higher with KR (27%) compared with R (15%), P=0.012; the most frequent were infections (KR 4% vs R 7%); all other events were reported in ≤5% of pts and included: gastrointestinal (KR 5% vs R 2%), cardiac (KR 4% vs R 1%), hypertension (KR 3% vs R 0%), and thrombotic microangiopathy (3% vs 0%). 4 pts developed a second primary malignancy in KR (breast 1 pt; thyroid 1 pt; myelodysplastic syndrome 1 pt; non-melanoma skin cancer 1pt) vs 1 pt in R (acute lymphoblastic leukemia). Dose reductions of R were reported in 23% of KR and 29% of R pts; dose reductions of K were reported in 20% of pts. The rate of discontinuation due to AEs was similar in the 2 arms (KR 10% vs R 9%).
Conclusions. Treatment with KRd_ASCT significantly improved PFS compared with both KRd12 and KCd_ASCT. Maintenance with KR also improved PFS vs R.
549 – Daratumumab (DARA) Plus Lenalidomide, Bortezomib, and Dexamethasone (RVd) in Patients with Transplant-Eligible Newly Diagnosed Multiple Myeloma (NDMM): Updated Analysis of Griffin after 12 Months of Maintenance Therapy
Author(s): Jonathan L. Kaufman, MD1, Jacob P. Laubach2*, Douglas Sborov, MD, MS3, Brandi Reeves, MD4, Cesar Rodriguez, MD5, Ajai Chari6*, Rebecca W. Silbermann, MD7, Luciano J. Costa, MD, PhD8, Larry D. Anderson Jr., MD, PhD9, Nitya Nathwani, MD10, Nina Shah, MD11, Yvonne A. Efebera, MD, MPH12, Sarah A. Holstein, MD, PhD13, Caitlin Costello14*, Andrzej Jakubowiak, MD, PhD15, Tanya M. Wildes, MD, MSc16, Robert Z. Orlowski, MD, PhD17, Kenneth H. Shain, MD, PhD 18, Andrew J. Cowan19*, Yana Lutska, PharmD20*, Padma Bobba20*, Huiling Pei, PhD21*, Jon Ukropec, PhD22, Jessica Vermeulen, MD, PhD23, Thomas S. Lin, MD, PhD20, Paul G. Richardson, MD2 and Peter M. Voorhees24*
1Winship Cancer Institute, Emory University, Atlanta, GA
2Dana-Farber Cancer Institute, Boston, MA
3Huntsman Cancer Institute, University of Utah School of Medicine, Salt Lake City, UT
4University of North Carolina-Chapel Hill, Chapel Hill, NC
5Wake Forest University School of Medicine, Winston-Salem, NC
6Tisch Cancer Institute, Mount Sinai School of Medicine, New York, NY
7Knight Cancer Institute, Oregon Health and Science University, Portland, OR
8University of Alabama at Birmingham, Birmingham, AL
9Simmons Comprehensive Cancer Center, UT Southwestern Medical Center, Dallas, TX
10Judy and Bernard Briskin Center for Multiple Myeloma Research, City of Hope Comprehensive Cancer Center, Duarte, CA
11Department of Medicine, University of California San Francisco, San Francisco, CA
12The Ohio State University Comprehensive Cancer Center, Columbus, OH
13Division of Oncology & Hematology, University of Nebraska Medical Center, Omaha, NE
14Moores Cancer Center, University of California San Diego, La Jolla, CA
15University of Chicago Medical Center, Chicago, IL
16Division of Oncology, Section Medical Oncology, Washington University School of Medicine, St. Louis, MO
17Department of Lymphoma–Myeloma, The University of Texas M.D. Anderson Cancer Center, Houston, TX
18Department of Malignant Hematology, H. Lee Moffitt Cancer Center, Tampa, FL
19Division of Medical Oncology, University of Washington, Seattle, WA
20Janssen Scientific Affairs, LLC, Horsham, PA
21Janssen Research & Development, LLC, Titusville, NJ
22Janssen Global Medical Affairs, Horsham, PA
23Janssen Research & Development, LLC, Leiden, Netherlands
24Levine Cancer Institute, Atrium Health, Charlotte, NC
*signifies non-member of ASH
Disclosures: Kaufman: Tecnopharma: Consultancy, Honoraria; Karyopharm: Membership on an entity’s Board of Directors or advisory committees; Celgene: Consultancy, Honoraria; Sanofi/Genyzme: Consultancy, Honoraria; AbbVie: Consultancy; Amgen: Consultancy, Honoraria; Bristol-Myers Squibb: Consultancy, Honoraria; TG Therapeutics: Consultancy, Membership on an entity’s Board of Directors or advisory committees; Takeda: Consultancy, Honoraria; Incyte: Consultancy, Membership on an entity’s Board of Directors or advisory committees; Janssen: Consultancy, Honoraria; Pharmacyclics: Membership on an entity’s Board of Directors or advisory committees. Sborov: University of Utah: Current Employment; Celgene, Janssen: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Personal fees. Reeves: Incyte: Honoraria; Takeda: Honoraria; Bristol Myers Squibb: Speakers Bureau. Rodriguez: BMS, Takeda, Amgen: Consultancy, Speakers Bureau. Chari: Janssen, Celgene, Novartis, Amgen, Bristol-Myers Squibb, Karyopharm, Sanofi, Genzyme, Seattle Genetics, Oncopeptides, Millennium/Takeda, Antengene, Glaxo Smith Kline, Secura Bio: Consultancy; Janssen, Celgene, Novartis, Amgen, Pharmacyclics, Seattle Genetics, Millennium/Takeda: Research Funding. Silbermann: Karyopharm: Consultancy; Janssen: Consultancy; Sanofi-Aventis: Consultancy, Research Funding. Costa: AbbVie: Consultancy; Sanofi: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; Janssen: Consultancy, Honoraria, Research Funding; Amgen: Consultancy, Honoraria, Research Funding; Genentech: Consultancy; BMS: Consultancy, Honoraria. Anderson: Amgen: Consultancy, Honoraria, Research Funding; GSK: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Research Funding; BMS: Consultancy, Honoraria, Research Funding; Karyopharm: Consultancy, Honoraria, Research Funding. Shah: GSK, Amgen, Indapta Therapeutics, Sanofi, BMS, CareDx, Kite, Karyopharm: Consultancy; BMS, Janssen, Bluebird Bio, Sutro Biopharma, Teneobio, Poseida, Nektar: Research Funding. Efebera: Pharmacyclics: Research Funding; Ohio State University: Current Employment; Celgene: Research Funding; Takeda: Honoraria, Speakers Bureau. Holstein: Sanofi: Consultancy; Adaptive Biotechnologies: Consultancy; Oncopeptides: Consultancy, Research Funding; Genentech: Consultancy; GSK: Consultancy; Celgene: Consultancy; Sorrento: Consultancy; Takeda: Consultancy. Costello: Takeda, Celgene: Consultancy, Honoraria. Jakubowiak: Adaptive, Juno: Consultancy, Honoraria; AbbVie, Amgen, BMS/Celgene, GSK, Janssen, Karyopharm: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees. Wildes: Seattle Genetics: Consultancy; Carevive Systems: Consultancy; Janssen: Research Funding. Orlowski: Founder of Asylia Therapeutics, Inc., with associated patents and an equity interest, though this technology does not bear on the current submission.: Current equity holder in private company, Patents & Royalties; STATinMED Research: Consultancy; Sanofi-Aventis, Servier, Takeda Pharmaceuticals North America, Inc.: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Amgen, Inc., AstraZeneca, BMS, Celgene, EcoR1 Capital LLC, Forma Therapeutics, Genzyme, GSK Biologicals, Ionis Pharmaceuticals, Inc., Janssen Biotech, Juno Therapeutics, Kite Pharma, Legend Biotech USA, Molecular Partners, Regeneron Pharmaceuticals, Inc.,: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Laboratory research funding from BioTheryX, and clinical research funding from CARsgen Therapeutics, Celgene, Exelixis, Janssen Biotech, Sanofi-Aventis, Takeda Pharmaceuticals North America, Inc.: Research Funding. Shain: BMS: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; Celgene: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; Janssen: Honoraria, Speakers Bureau; AbbVie: Research Funding; GlaxoSmithKline: Speakers Bureau; Adaptive: Consultancy, Honoraria; Sanofi/Genzyme: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; Takeda: Honoraria, Speakers Bureau; Karyopharm: Research Funding, Speakers Bureau; Amgen: Speakers Bureau. Cowan: Nektar: Research Funding; Janssen: Consultancy, Research Funding; Abbvie: Research Funding; Bristol-Myer Squibb: Research Funding; Celgene: Consultancy, Research Funding; Cellectar: Consultancy; Sanofi-Aventis: Consultancy. Lutska: Janssen: Current Employment. Bobba: Janssen: Current Employment. Pei: Janssen: Current Employment, Current equity holder in publicly-traded company. Ukropec: Janssen: Current Employment, Current equity holder in publicly-traded company. Vermeulen: Janssen: Current Employment, Current equity holder in publicly-traded company. Lin: Janssen Scientific Affairs: Current Employment, Current equity holder in publicly-traded company. Richardson: Celgene/BMS, Oncopeptides, Takeda, Karyopharm: Research Funding. Voorhees: TeneoBio: Other: Advisory Board; Oncopeptides: Consultancy, Honoraria; Novartis: Consultancy; Janssen: Other: Advisory Board; GSK: Honoraria; BMS: Other: Advisory Board; Adaptive Biotechnologies: Other: Advisory Board.
OffLabel Disclosure: The specific regimen combination is not yet approved, but individual components are.
Introduction: DARA, a human IgGκ monoclonal antibody targeting CD38, is approved as monotherapy and in combination with standard-of-care regimens for relapsed/refractory multiple myeloma and NDMM. In the primary analysis of the phase 2 GRIFFIN study (NCT02874742) in patients with transplant-eligible NDMM, DARA plus RVd (D-RVd) significantly improved rates of stringent complete response (sCR) by the end of post-transplant consolidation therapy versus RVd (Voorhees P, Blood 2020). Here, we present updated efficacy and safety results following 12 months of maintenance therapy with lenalidomide (R) or DARA plus R (D-R).
Methods: Patients with NDMM eligible for high-dose therapy (HDT) and autologous stem cell transplant (ASCT) were randomized 1:1 to RVd ± DARA, stratified by ISS stage and creatinine clearance rate. Patients received 4 induction cycles, HDT, ASCT, 2 consolidation cycles, and maintenance with R ± DARA for 24 months. During induction and consolidation, patients received R 25 mg PO on Days 1‐14; V 1.3 mg/m2 SC on Days 1, 4, 8, and 11; and d 40 mg QW every 21 days. DARA 16 mg/kg IV was given on Days 1, 8, and 15 of Cycles 1‐4 and Day 1 of Cycles 5‐6. During maintenance (Cycles 7-32), patients received R 10 mg (15 mg in Cycles 10+ if tolerated) on Days 1‐21 every 28 days ± DARA 16 mg/kg IV Q8W (or Q4W per patient decision after Amendment 2). The primary endpoint was rate of sCR at the end of post-ASCT consolidation per IMWG criteria, evaluated by a validated computer algorithm. Key secondary endpoints included progression-free survival (PFS) and rate of minimal residual disease (MRD) negativity (10‑5 threshold per IMWG criteria) assessed by next-generation sequencing (clonoSEQ; Adaptive Biotechnologies). The primary hypothesis was tested at a 1-sided alpha of 0.10. All secondary analyses were evaluated using a 2-sided P value (alpha 0.05) and were not adjusted for multiplicity.
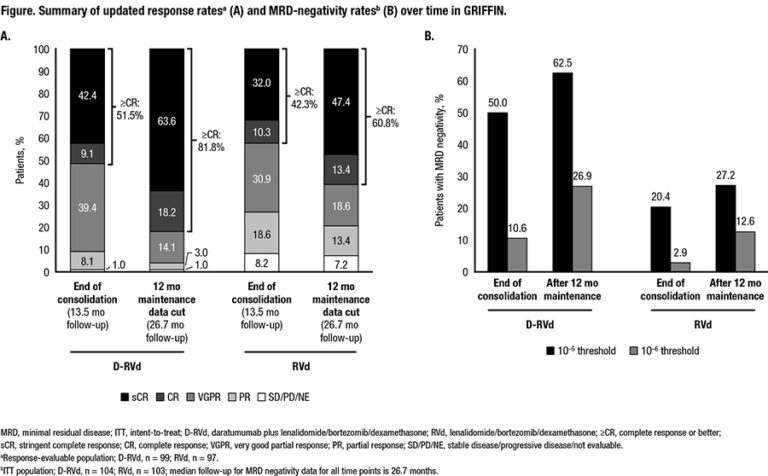
Results: In total, 207 patients were randomized (D-RVd, n=104; RVd, n=103). Baseline demographics and disease characteristics were well balanced between arms. At the end of post-transplant consolidation (median follow-up, 13.5 months) in the response-evaluable population, the sCR rate favored D-RVd versus RVd (42.4% [42/99] vs 32.0% [31/97]; 1-sided P=0.0680). With additional D-R or R maintenance therapy, responses continued to deepen and remained higher for the D-RVd group versus the RVd group. At the 12-months-of-maintenance therapy data cut (median follow-up, 26.7 months), the sCR rate still favored D-RVd versus RVd (63.6% [63/99] vs 47.4% [46/97], 2-sided P=0.0253; Figure). MRD-negativity (10‒5) rates in the ITT population favored D-RVd versus RVd (62.5% [65/104] vs 27.2% [28/103], P<0.0001; Figure), as well as among patients who achieved complete response (CR) or better at that time (76.5% [62/81] vs 42.4% [25/59], P<0.0001). Similarly, MRD-negativity (10‒6) rates favored D-RVd versus RVd in the ITT population (26.9% [28/104] vs 12.6% [13/103], P=0.0140; Figure), as well as among patients who achieved CR or better at that time (34.6% [28/81] vs 18.6% [11/59], P=0.0555). Estimated 24-month PFS rates were 94.5% and 90.8% for the D-RVd and RVd groups, respectively. In total, 14 deaths occurred (n=7 per group), and 9 were due to progressive disease (D-RVd, n=5; RVd, n=4). With longer follow-up, no new safety concerns were observed. 84.8% (84/99) of patients in the D-RVd group and 79.4% (81/102) in the RVd group had grade 3/4 treatment-emergent adverse events (TEAEs). One grade 5 TEAE occurred in the RVd group, which was unrelated to study therapy (unknown cause). Infusion-related reactions occurred in 43.4% (43/99) of patients, with the majority being grade 1 or 2 and occurring in the first cycle.
Conclusions: After 26.7 months of median follow-up, the addition of DARA to RVd induction and consolidation, followed by D-R maintenance in patients with transplant-eligible NDMM continued to demonstrate deep and improved responses, including higher sCR and MRD negativity rates, compared with lenalidomide alone. Maintenance therapy increased sCR and MRD negativity rates, compared to post-consolidation rates. No new safety concerns were observed with longer follow-up.
Support: Alliance Foundation Trials; https://acknowledgments.alliancefound.org; Janssen Oncology
131 – Idecabtagene Vicleucel (ide-cel, bb2121), a BCMA-Directed CAR T Cell Therapy, in Patients with Relapsed and Refractory Multiple Myeloma: Updated Results from Phase 1 CRB-401 Study
Author(s): Yi Lin, MD, PhD1, Noopur S. Raje, MD2, Jesus G. Berdeja, MD3, David S. Siegel, MD4, Sundar Jagannath, MD5, Deepu Madduri, MD5*, Michaela Liedtke, MD6, Jacalyn Rosenblatt, MD7, Marcela V. Maus, MD, PhD2, Monica Massaro, MPH8*, Fabio Petrocca, MD9*, Andrea Caia, MS10*, Zhihong Yang, PhD10*, Timothy B. Campbell, MD, PhD10*, Kristen Hege, MD10, Nikhil C. Munshi, MD11 and James N. Kochenderfer, MD12
1Mayo Clinic, Rochester, MN
2Massachusetts General Hospital Cancer Center, Boston, MA
3Sarah Cannon Research Institute and Tennessee Oncology, Nashville, TN
4Hackensack University Medical Center, Hackensack, NJ
5Mount Sinai Medical Center, New York, NY
6Stanford University Medical Center, Stanford, CA
7Beth Israel Deaconess Medical Center, Boston, MA
8bluebird bio, Inc., Cambridge, MA
9bluebird bio, Cambridge, MA
10Bristol Myers Squibb, Princeton, NJ
11Dana-Farber Cancer Institute, Boston, MA
12Surgery Branch, National Cancer Institute/National Institutes of Health, Bethesda, MD
*signifies non-member of ASH
Disclosures: Lin: Novartis: Consultancy; Janssen: Consultancy, Research Funding; Vineti: Consultancy; Sorrento: Consultancy, Membership on an entity’s Board of Directors or advisory committees; Gamida Cells: Consultancy; Takeda: Research Funding; Merck: Research Funding; Legend BioTech: Consultancy; Juno: Consultancy; Celgene: Consultancy, Research Funding; Bluebird Bio: Consultancy, Research Funding; Kite, a Gilead Company: Consultancy, Research Funding. Raje: Caribou: Membership on an entity’s Board of Directors or advisory committees; BMS: Consultancy; Bluebird, Bio: Consultancy, Research Funding; Takeda: Consultancy; Immuneel: Membership on an entity’s Board of Directors or advisory committees; Janssen: Consultancy; Karyopharm: Consultancy; Celgene: Consultancy; Astrazeneca: Consultancy; Amgen: Consultancy. Berdeja: Novartis: Research Funding; Lilly: Research Funding; CURIS: Research Funding; Prothena: Consultancy; Celgene: Consultancy, Research Funding; Servier: Consultancy; Genentech, Inc.: Research Funding; EMD Sorono: Research Funding; Cellularity: Research Funding; BMS: Consultancy, Research Funding; Bioclinica: Consultancy; Bluebird: Research Funding; Acetylon: Research Funding; Amgen: Consultancy, Research Funding; Abbvie: Research Funding; Vivolux: Research Funding; Poseida: Research Funding; CRISPR Therapeutics: Consultancy, Research Funding; Teva: Research Funding; Legend: Consultancy; Kite Pharma: Consultancy; Glenmark: Research Funding; Karyopharm: Consultancy; Janssen: Consultancy, Research Funding; Constellation: Research Funding; Kesios: Research Funding; Takeda: Consultancy, Research Funding. Siegel: Karyopharma: Consultancy, Honoraria; Takeda: Consultancy, Honoraria, Speakers Bureau; BMS: Consultancy, Honoraria, Speakers Bureau; Janssen: Consultancy, Honoraria, Speakers Bureau; Merck: Consultancy, Honoraria, Speakers Bureau; Amgen: Consultancy, Honoraria, Speakers Bureau; Celulatiry: Consultancy. Jagannath: Janssen: Consultancy, Honoraria; Sanofi: Consultancy, Honoraria; Legend Biotech: Consultancy, Honoraria; Karyopharm: Consultancy, Honoraria; BMS: Consultancy, Honoraria; Takeda: Consultancy, Honoraria. Madduri: Legend: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Speaking Engagement, Speakers Bureau; Celgene: Consultancy, Honoraria; Kinevant: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Speaking Engagement, Speakers Bureau; Foundation Medicine: Consultancy, Honoraria; AbbVie: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; GSK: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: Speaking Engagement, Speakers Bureau. Liedtke: Pfizer: Honoraria; Jazz Pharmaceuticals: Membership on an entity’s Board of Directors or advisory committees; Janssen: Membership on an entity’s Board of Directors or advisory committees; GSK: Membership on an entity’s Board of Directors or advisory committees; Celgene: Membership on an entity’s Board of Directors or advisory committees; Caelum: Membership on an entity’s Board of Directors or advisory committees; Adaptive: Membership on an entity’s Board of Directors or advisory committees. Rosenblatt: Celgene: Research Funding. Maus: Novartis: Consultancy, Research Funding; arcellx: Consultancy, Research Funding; kite: Consultancy, Research Funding; century therapeutics: Current equity holder in private company; tcr2: Consultancy, Current equity holder in publicly-traded company. Massaro: bluebird, bio: Current Employment, Current equity holder in publicly-traded company. Petrocca: bluebird, bio: Current Employment, Current equity holder in publicly-traded company. Caia: Celgene a BMS company: Current Employment, Current equity holder in publicly-traded company. Yang: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company; Celgene: Ended employment in the past 24 months. Campbell: BMS: Current Employment, Current equity holder in publicly-traded company. Hege: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Patents & Royalties: numerous, Research Funding; Celgene (acquired by Bristol Myers Squibb): Ended employment in the past 24 months; Mersana Therapeutics: Current equity holder in publicly-traded company, Membership on an entity’s Board of Directors or advisory committees; Arcus Biosciences (Former Board of Directors): Divested equity in a private or publicly-traded company in the past 24 months. Munshi: BMS: Consultancy; OncoPep: Consultancy, Current equity holder in private company, Membership on an entity’s Board of Directors or advisory committees, Patents & Royalties; AbbVie: Consultancy; Karyopharm: Consultancy; Takeda: Consultancy; Adaptive: Consultancy; Janssen: Consultancy; C4: Current equity holder in private company; Amgen: Consultancy; Legend: Consultancy. Kochenderfer: Celgene: Patents & Royalties, Research Funding; bluebird, bio: Patents & Royalties; Kite, a Gilead company: Patents & Royalties, Research Funding.
Background: Ide-cel, a BCMA-directed CAR T cell therapy, showed tolerability and promising efficacy in patients with relapsed and/or refractory multiple myeloma (RRMM) in the first-in-human phase 1 CRB-401 study (Raje et al. N Engl J Med. 2019;380:1726) and the pivotal phase 2 KarMMa study (Munshi et al. J Clin Oncol. 2020;38[suppl, abstr]:8503). Ide-cel demonstrated a favorable benefit-risk profile with an overall response rate (ORR) of 85%, a complete response (CR) rate of 45%, and a median progression-free survival (PFS) of 11.8 months in the first 33 patients treated in CRB-401. Reported here are updated safety and efficacy results for 62 patients who received ide-cel in the ongoing CRB-401 study.
Methods: CRB-401 (NCT02658929) is a 2-part, phase 1 dose-escalation and -expansion study. The expansion phase enrolled patients who had received ≥3 prior lines of therapy, including a proteasome inhibitor, an immunomodulatory drug, and an anti-CD38 antibody, and were refractory to their last line of therapy. Eligibility criteria for the dose-escalation phase were described previously (Raje et al. N Engl J Med. 2019;380:1726). After lymphodepletion with fludarabine (30 mg/m2/day) and cyclophosphamide (300 mg/m2/day) for 3 days followed by 2 days of rest, patients received ide-cel at target doses of 50, 150, 450, or 800 × 106 CAR+ T cells in the dose-escalation phase and 150 to 450 × 106 CAR+ T cells in the dose-expansion phase. The primary endpoint was safety. Secondary endpoints included tumor response according to the International Myeloma Working Group criteria. Exploratory endpoints included PFS, overall survival (OS), and minimal residual disease (MRD).
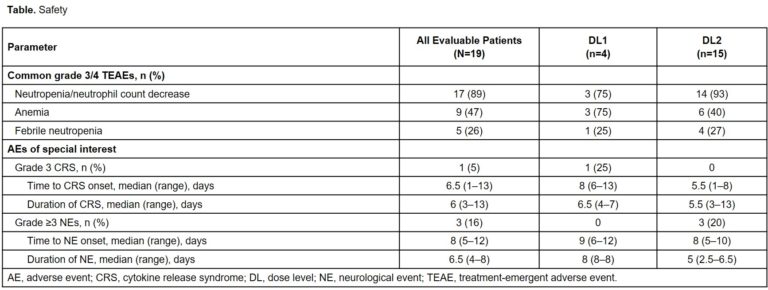
Results: As of January 14, 2020, 21 patients had received ide-cel in the dose-escalation phase, and 41 patients received ide-cel in the dose-expansion phase. The median age was 61 years, and 44% of patients had high tumor burden (≥50% bone marrow CD138+ plasma cells). Of the 62 patients, 45% received >6 prior regimens, 90% were daratumumab-exposed, and 77% were daratumumab-refractory. As of the cutoff date, 13 patients were ongoing, and 49 patients had discontinued the study. Reasons for study discontinuation were progressive disease (58%), withdrawal by patients (10%), and death (10%). Based on safety and efficacy in the dose-escalation phase, target dose levels of 150 to 450 × 106 CAR+ T cells were selected for the dose-expansion phase. The most frequent adverse events (AEs) were neutropenia (92%), cytokine release syndrome (CRS; 76%), anemia (76%), and thrombocytopenia (74%). The most frequent grade 3/4 AEs were neutropenia (89%), leukopenia (61%), anemia (57%), and thrombocytopenia (57%). Most CRS events were grade 1 or 2 (Table). Four patients (7%) had grade 3 CRS; there were no grade >3 CRS events. The incidence of CRS generally increased with target dose level. Neurologic toxicity (NT; clustered term) occurred in 27 patients (44%) and was primarily grade 1/2 with 1 patient having grade 3 and 1 patient having grade 4 NT. Among all 62 patients in the dose-escalation and -expansion phases, the ORR was 76%, including 24 patients (39%) with a CR or better and 40 patients (65%) with a very good partial response or better. The median duration of response was 10.3 months. Of 37 responders evaluable for MRD, 30 were MRD negative (≤10−4 nucleated cells) at 1 or more time point, and 7 responders were MRD positive. With a median follow-up of 14.7 months for all patients in the dose-escalation and dose-expansion phases, median PFS was 8.8 months and median OS was 34.2 months. Overall, a dose-dependent effect was observed on responses and survival outcomes, with greater efficacy reported at ≥150 × 106 CAR+ T cells (Table).
Conclusions: Ide-cel demonstrated deep and durable responses in heavily-pretreated RRMM patients. Efficacy and safety reflect prior reports and support a favorable clinical benefit-risk profile for ide-cel at target dose levels ≥150 × 106 CAR+ T cells.
724 – First Results of Iberdomide (IBER; CC-220) in Combination with Dexamethasone (DEX) and Daratumumab (DARA) or Bortezomib (BORT) in Patients with Relapsed/Refractory Multiple Myeloma (RRMM)
Author(s): Niels W.C.J. Van De Donk1, Rakesh Popat2*, Jeremy Larsen3, Monique C. Minnema4, Sundar Jagannath, MD5*, Albert Oriol6*, Jeffrey Zonder7, Paul G. Richardson, MD8, Paula Rodriguez-Otero, MD, PhD9*, Ashraf Z. Badros, MD10, Edward Stadtmauer11*, Sara Bringhen, MD12, Erica Campagnaro, MD13*, David S. Siegel, MD14, Barbara Gamberi15*, Mercedes Gironella Mesa16*, Pieter Sonneveld, MD, PhD17, Tuong Vi Nguyen18*, Antonia Di Micco19*, April Sorrell18*, Min Chen18*, Michael Amatangelo18*, Elisabeth Kueenburg19* and Sagar Lonial, MD20
1Department of Hematology, Cancer Center Amsterdam, Amsterdam UMC, VU University Medical Center, Amsterdam, Netherlands
2NIHR UCLH Clinical Research Facility, University College London Hospitals NHS Foundation Trust, London, United Kingdom
3Mayo Clinic, Scottsdale, AZ
4Department of Hematology, University Medical Center Utrecht, Utrecht University, Utrecht, Netherlands
5The Mount Sinai Hospital, New York, NY
6Institut Josep Carreras and Institut Català d’Oncologia, Hospital Germans Trias i Pujol, Badalona, Spain
7Karmanos Cancer Institute, Detroit, MI
8Dana-Farber Cancer Institute, Boston, MA
9Clínica Universidad de Navarra, CIMA, IDISNA, CIBERONC, Pamplona, Spain
10The University of Maryland Marlene and Stewart Greenebaum Comprehensive Cancer Center, University of Maryland Medical Center, Baltimore, MD
11University of Pennsylvania, Philadelphia, PA
12Myeloma Unit, Division of Hematology, University of Torino, Azienda-Ospedaliero Universitaria Città della Salute e della Scienza di Torino, Torino, Italy
13Michigan Medicine Hematology Clinic, University of Michigan Rogel Cancer Center, Ann Arbor, MI
14Division of Multiple Myeloma, John Theurer Cancer Center, Hackensack University Medical Center, Hackensack, NJ
15Hematology Unit, AUSL-IRCSS, Reggio Emilia, Italy
16University Hospital Vall d’Hebron, Barcelona, Spain
17Erasmus Medical Center Rotterdam, Rotterdam, Netherlands
18Bristol Myers Squibb, Princeton, NJ
19Celgene International Sàrl, a Bristol-Myers Squibb Company, Boudry, Switzerland
20Winship Cancer Institute of Emory University, Atlanta, GA
*signifies non-member of ASH
Disclosures: Van De Donk: Bristol Myers Squibb: Membership on an entity’s Board of Directors or advisory committees, Research Funding; Novartis: Membership on an entity’s Board of Directors or advisory committees; Janssen: Membership on an entity’s Board of Directors or advisory committees, Research Funding; Bayer: Membership on an entity’s Board of Directors or advisory committees; Takeda: Membership on an entity’s Board of Directors or advisory committees; Ferrer: Membership on an entity’s Board of Directors or advisory committees; Amgen: Membership on an entity’s Board of Directors or advisory committees, Research Funding. Popat: Takeda: Consultancy, Honoraria, Other: Travel support, Research Funding; Bristol Myers Squibb: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; Janssen: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company); AbbVie: Consultancy, Honoraria; GSK: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company). Larsen: Janssen Oncology: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Takeda: Honoraria, Membership on an entity’s Board of Directors or advisory committees. Minnema: Servier: Consultancy; Kite, a Gilead Company: Speakers Bureau; Celgene: Other: travel support, Research Funding; Amgen: Consultancy. Jagannath: BMS, Janssen, Karyopharm, Legend Biotech, Sanofi, Takeda: Consultancy. Oriol: Sanofi: Membership on an entity’s Board of Directors or advisory committees; GlaxoSmithKline: Membership on an entity’s Board of Directors or advisory committees; Janssen: Consultancy; Bristol-Myers Squibb: Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; Amgen: Consultancy, Speakers Bureau. Zonder: Prothena: Consultancy; Intellia: Membership on an entity’s Board of Directors or advisory committees, Other: Personal fees; Celgene: Research Funding; BMS: Consultancy, Research Funding; Amgen: Membership on an entity’s Board of Directors or advisory committees, Other: Personal fees; Oncopeptide: Membership on an entity’s Board of Directors or advisory committees, Other: Personal fees; Alnylam: Membership on an entity’s Board of Directors or advisory committees, Other: Personal fees; Janssen: Consultancy, Other: Personal fees; Caelum: Consultancy; Takeda: Membership on an entity’s Board of Directors or advisory committees, Other: Personal fees. Richardson: Celgene/BMS, Oncopeptides, Takeda, Karyopharm: Research Funding. Rodriguez-Otero: Sanofi: Consultancy, Honoraria; Medscape: Membership on an entity’s Board of Directors or advisory committees; Celgene/Bristol-Myers Squibb: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company); GlaxoSmithKline: Consultancy, Current Employment, Current equity holder in publicly-traded company, Honoraria; Abbvie: Consultancy, Honoraria; Kite: Consultancy, Honoraria; Janssen: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company); Amgen: Consultancy, Honoraria; Oncopeptides: Consultancy, Honoraria. Badros: Amgen: Consultancy; University of Maryland: Current Employment. Stadtmauer: Sanofi: Consultancy; AbbVie: Research Funding; Takeda: Consultancy; Bristol Myers Squibb: Consultancy, Research Funding. Bringhen: Bristol-Myers Squibb: Honoraria; Amgen: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Janssen: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Karyopharm: Membership on an entity’s Board of Directors or advisory committees; Takeda: Consultancy; Celgene: Honoraria, Membership on an entity’s Board of Directors or advisory committees. Siegel: Takeda: Consultancy, Honoraria, Speakers Bureau; BMS: Consultancy, Honoraria, Speakers Bureau; Amgen: Consultancy, Honoraria, Speakers Bureau; Merck: Consultancy, Honoraria, Speakers Bureau; Janssen: Consultancy, Honoraria, Speakers Bureau; Celulatiry: Consultancy; Karyopharma: Consultancy, Honoraria. Gamberi: Janssen: Consultancy, Honoraria; GSK: Consultancy; Amgen: Consultancy, Honoraria; Sanofi: Consultancy, Honoraria; Bristol Myers Squibb: Consultancy, Honoraria. Gironella Mesa: Janssen: Consultancy, Honoraria; Bristol Myers Squibb: Honoraria, Speakers Bureau. Sonneveld: Celgene: Honoraria, Research Funding; Karyopharm: Consultancy, Honoraria, Research Funding; Skyline Dx: Honoraria, Research Funding; Takeda: Consultancy, Honoraria, Research Funding; Bristol-Myers Squibb: Consultancy, Honoraria, Research Funding; Sanofi: Consultancy; Amgen: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Research Funding. Nguyen: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Di Micco: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Sorrell: Children’s Oncology Group: Other: Non-member; Previous Study Chair AAML08B1; Bristol Myers Squibb: Current Employment. Chen: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Amatangelo: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Kueenburg: Celgene International Sàrl, a Bristol Myers Squibb Company, Boudry, Switzerland: Current Employment. Lonial: Onyx: Honoraria; Novartis: Consultancy, Honoraria, Other: Personal fees; Genentech: Consultancy; Amgen: Consultancy, Honoraria, Other: Personal fees; Merck: Consultancy, Honoraria, Other: Personal fees; Abbvie: Consultancy; Millennium: Consultancy, Honoraria; Karyopharm: Consultancy; Sanofi: Consultancy; JUNO Therapeutics: Consultancy; TG Therapeutics: Membership on an entity’s Board of Directors or advisory committees; Takeda: Consultancy, Other: Personal fees, Research Funding; Janssen: Consultancy, Honoraria, Other: Personal fees, Research Funding; BMS: Consultancy, Honoraria, Other: Personal fees, Research Funding; GSK: Consultancy, Honoraria, Other: Personal fees.
Introduction: IBER is an oral, potent novel cereblon E3 ligase modulator (CELMoD) agent with marked synergistic tumoricidal and immune-stimulatory effects in combination with BORT or DARA in preclinical models. CC-220-MM-001 is a phase 1/2 study evaluating dose escalations of IBER with different treatment combinations in independent cohorts, in patients (pts) with RRMM (NCT02773030); the IBER + DEX cohort showed a favorable safety profile with promising efficacy and a selected IBER RP2D of 1.6 mg 21/28 days (D). Here, we present results from the IBER + DARA + DEX (IberDd) and IBER + BORT + DEX (IberVd) cohorts.
Methods: Eligible pts had received ≥ 2 prior regimens in the IberDd cohort, and ≥ 1 prior regimen in the IberVd cohort, containing at least lenalidomide or pomalidomide, and a proteasome inhibitor (PI), and had experienced disease progression on or within 60 days of last MM therapy. Escalating doses of IBER were given orally, in the IberDd cohort on D1–21, with DARA (16 mg/kg) on D1, D8, D15, and D22 (cycles [C]1–2), D1 and D15 (C3–6), and D1 (C ≥ 7), of each 28-day cycle; in the IberVd cohort, on D1–14, with BORT (1.3 mg/m2) on D1, D4, D8, and D11 (C1–8), and on D1 and D8 (C ≥ 9), of each 21-day cycle. In both cohorts DEX was given weekly. Primary objectives were to evaluate MTD, RP2D, and safety separately for each cohort; a key secondary objective was preliminary assessment of efficacy. Immune profiling was evaluated by flow cytometry from pt peripheral blood at C1D1, C2D15, C4D1, and C4D15.
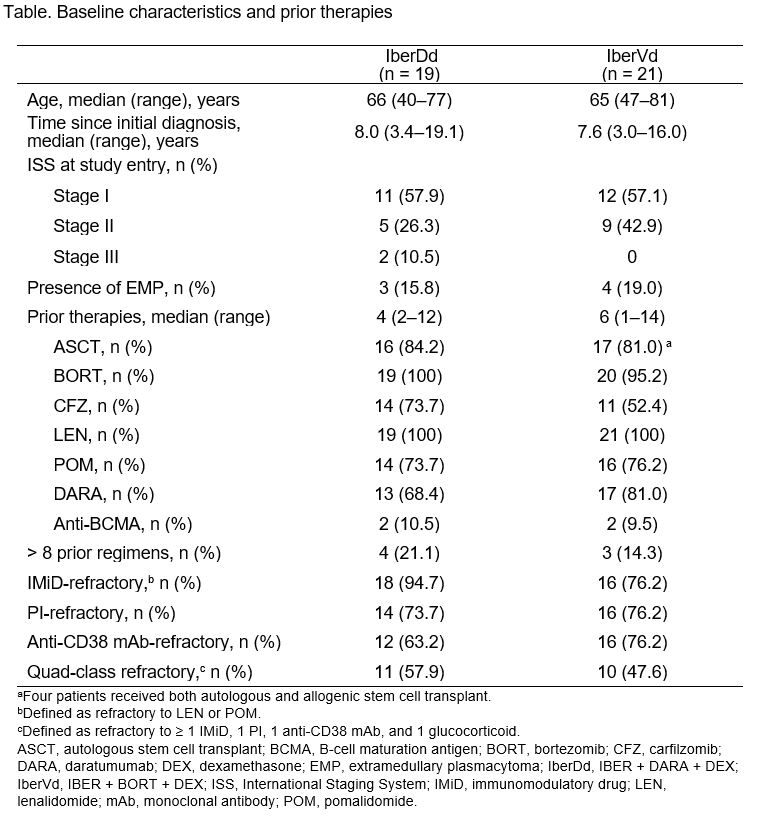
Results: As of June 18, 2020, 19 pts had received IberDd and 21 pts IberVd. Baseline characteristics for the 2 independent cohorts are shown in the table. All pts were refractory to their last prior regimen, and exposure to prior regimens was heterogeneous. IBER doses ranged from 1.0 to 1.6 mg; the MTD/RP2D has not been reached in either cohort. Median follow-up was 5 (0–14) and 3 (0–11) months, 10 (53%) and 13 (62%) pts continue on treatment, median cycles received was 5 (1–14) and 4.5 (1–17), with IberDd and IberVd, respectively.
Grade (Gr) 3–4 treatment-emergent adverse events (TEAEs) were reported in 14 (78%) pts with IberDd, and in 13 (65%) pts with IberVd. Most frequent Gr 3–4 TEAEs of interest included neutropenia (50%), leukopenia (22%), and anemia (22%) with IberDd; and neutropenia (20%) and thrombocytopenia (20%) with IberVd. In both cohorts, neutropenia was managed with G-CSF. One pt (IberDd; 1.2 mg dose) had Gr 4 neutropenic sepsis. Occurrence of Gr 3–4 non-hematological TEAEs was low in both cohorts. One pt had Gr 2 infusion-related reaction with IberDd, and 3 pts had Gr 1–2 peripheral neuropathy with IberVd. Six (33%) and 4 (20%) pts had IBER dose reductions with IberDd and IberVd, respectively.
In the IberDd cohort, with 12/19 (63%) DARA-refractory pts and 11 (58%) quad-class-refractory pts, the overall response rate (ORR) was 35% across all dosing groups (2 very good partial responses [VGPRs], 4 partial responses [PRs]); the clinical benefit rate (CBR) was 47% and disease control rate (DCR) was 88%. In the IberVd cohort, with 16/21 (76%) PI-refractory pts, 9 (43%) BORT-refractory pts, and 10 (48%) quad-class refractory pts, ORR was 50% (1 complete response, 3 VGPRs, 6 PRs); CBR was 65% and DCR was 85%. Responses with IberDd and IberVd were observed irrespective of DARA- and BORT-refractoriness, respectively. Median time to response was 4.1 (4.1–12.0) and 4.9 (3.0–13.1) weeks, in the IberDd and IberVd cohorts, respectively; median duration of response was not reached in both cohorts.
Immune profiling showed dose-dependent decreases in B cells and increases in activated and differentiated T cells, in both cohorts. Except for reductions in CD38+ T cells in pts receiving IberDd, these observations were similar in pts treated with IBER + DEX.
Conclusions: IberDd and IberVd showed a favorable tolerability profile in heavily pretreated RRMM pts, with promising clinical activity, even among pts refractory to the last prior regimen and previously exposed to IMiD agents, PIs, and CD38 antibodies. Immune-profiling data confirm that IBER + DEX was pharmacodynamically active in triplet combination and not augmented by the addition of DARA or BORT. The study is ongoing with continued enrollment at the 1.6 mg dose level for both cohorts. Updated results, including the MTD/RP2D, will be presented at the meeting. These results support the further development of IBER-based regimens in MM; phase 3 trials are planned to further evaluate these combinations.
692 – CC-486 Prolongs Survival for Patients with Acute Myeloid Leukemia (AML) in Remission after Intensive Chemotherapy (IC) Independent of the Presence of Measurable Residual Disease (MRD) at Study Entry: Results from the QUAZAR AML-001 Maintenance Trial
Author(s): Gail J. Roboz, MD1, Farhad Ravandi, MBBS2, Andrew H Wei, MBBS, PhD3,4, Hervé Dombret, MD5,6, Hartmut Döhner7, Felicitas Thol8, Maria Teresa Voso, MD9, Andre C. Schuh10, Kimmo Porkka11,12, Ignazia La Torre13*, Barry Skikne, MD14,15*, Keshava Kumar, PhD15*, Qian Dong, DrPH15*, C.L. Beach, PharmD15*, Alberto Risueño16*, Daniel Lopes de Menezes, PhD15* and Gert Ossenkoppele, MD, PhD17*
1Weill Medical College of Cornell University New York-Presbyterian Hospital, New York, NY
2Department of Leukemia, The University of Texas MD Anderson Cancer Center, Houston, TX
3The Alfred Hospital, Melbourne, Australia
4Australian Centre for Blood Diseases, Monash University, Melbourne, Australia
5Hôpital Saint-Louis, Assistance Publique – Hôpitaux de Paris (AP-HP), Paris, France
6Institut de Recherche Saint Louis, Université de Paris, Paris, France
7Ulm University Hospital, Ulm, Germany
8Medizinische Hochschule Hannover, Hannover, Germany
9University of Rome Tor Vergata, Rome, Italy
10Princess Margaret Cancer Centre, Toronto, ON, Canada
11iCAN Digital Precision Cancer Medicine Flagship, University of Helsinki, Helsinki, Finland
12Hematology Research Unit Helsinki, Helsinki University Hospital Comprehensive Cancer Center, Helsinki, Finland
13Celgene, a Bristol-Myers Squibb Company, Boudry, Switzerland
14Kansas University Medical Center, Kansas City, KS
15Bristol Myers Squibb, Princeton, NJ
16BMS Center for Innovation and Translational Research Europe (CITRE), a Bristol Myers Squibb Company, Seville, Spain
17Amsterdam UMC, Location VU University Medical Center, Amsterdam, Netherlands
*signifies non-member of ASH
Disclosures: Roboz: Abbvie: Consultancy; Array BioPharma: Consultancy; Bayer: Consultancy; Celltrion: Consultancy; Jazz: Consultancy; Eisai: Consultancy; Sandoz: Consultancy; Actinium: Consultancy; Argenx: Consultancy; Astellas: Consultancy; Daiichi Sankyo: Consultancy; AstraZeneca: Consultancy; Orsenix: Consultancy; Otsuka: Consultancy; Agios: Consultancy; Amphivena: Consultancy; Astex: Consultancy; Celgene: Consultancy; Janssen: Consultancy; Novartis: Consultancy; Pfizer: Consultancy; GlaxoSmithKline: Consultancy; Bristol Myers Squibb: Consultancy; Mesoblast: Consultancy; MEI Pharma: Consultancy; Amgen: Consultancy; Trovagene: Consultancy; Cellectis: Research Funding; Jasper Therapeutics: Consultancy; Epizyme: Consultancy; Helsinn: Consultancy; Takeda: Consultancy; Roche/Genentech: Consultancy. Ravandi: Abbvie: Consultancy, Honoraria, Research Funding; Xencor: Consultancy, Honoraria, Research Funding; AstraZeneca: Consultancy, Honoraria; Macrogenics: Research Funding; Jazz Pharmaceuticals: Consultancy, Honoraria, Research Funding; Astellas: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria; BMS: Consultancy, Honoraria, Research Funding; Amgen: Consultancy, Honoraria, Research Funding; Orsenix: Consultancy, Honoraria, Research Funding. Wei: Pfizer: Honoraria; Bristol Myers Squibb: Honoraria, Research Funding, Speakers Bureau; Janssen: Honoraria; Walter and Eliza Hall Institute of Medical Research: Patents & Royalties: AW is eligible for royalty payments related to venetoclax; Roche: Honoraria; Amgen: Honoraria, Research Funding; Novartis: Honoraria, Research Funding, Speakers Bureau; Abbvie: Honoraria, Research Funding, Speakers Bureau; Servier: Consultancy, Honoraria, Research Funding; Macrogenics: Honoraria; Astra Zeneca: Honoraria, Research Funding. Dombret: Menarini: Consultancy; Janssen: Consultancy; Cellectis: Consultancy; Shire-Baxalta: Consultancy; Immunogen: Consultancy; Otsuka: Consultancy; Abbvie: Consultancy; Astellas: Consultancy; Daiichi Sankyo: Consultancy; Servier: Consultancy, Research Funding; Sunesis: Consultancy; Amgen: Consultancy, Research Funding; Jazz Pharma: Consultancy, Research Funding; Celgene: Consultancy; Nova: Consultancy, Research Funding; Incyte: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding. Döhner: Astex: Consultancy, Honoraria; Astellas: Consultancy, Honoraria, Research Funding; AROG: Research Funding; Amgen: Consultancy, Honoraria, Research Funding; Agios: Consultancy, Honoraria, Research Funding; Abbvie: Consultancy, Honoraria; AstraZeneca: Consultancy, Honoraria; GEMoaB: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria; Novartis: Consultancy, Honoraria, Research Funding; Oxford Biomedicals: Consultancy, Honoraria; Sunesis: Research Funding; Pfizer: Research Funding; Roche: Consultancy, Honoraria; Jazz: Consultancy, Honoraria, Research Funding; Helsinn: Consultancy, Honoraria; Bristol Myers Squibb: Consultancy, Honoraria, Research Funding. Thol: Abbvie: Membership on an entity’s Board of Directors or advisory committees; Celgene: Membership on an entity’s Board of Directors or advisory committees; Pfizer: Membership on an entity’s Board of Directors or advisory committees; Astellas: Membership on an entity’s Board of Directors or advisory committees; Daiichi Sankyo: Membership on an entity’s Board of Directors or advisory committees. Voso: Bristol Myers Squibb: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Research Funding. Porkka: Novartis: Consultancy, Honoraria, Research Funding; BMS/Celgene: Honoraria, Research Funding. La Torre: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Skikne: Bristol Myers Squibb: Current Employment. Kumar: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Dong: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Beach: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Risueño: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company, Patents & Royalties: Named in BMS (before Celgene) patent filings related to predictive patient response biomarkers in hematological malignancies. Lopes de Menezes: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Ossenkoppele: Novartis: Consultancy, Research Funding; Genentech: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; Roche: Consultancy; J&J: Consultancy, Research Funding; Agios: Consultancy; Jazz: Consultancy; Astellas: Consultancy; Daiichi Sayko: Consultancy; Amgen: Consultancy.
BACKGROUND: In newly diagnosed AML, high remission rates are typically achieved with IC, but the response is often transient, and detectable residual disease in the bone marrow post-chemotherapy is predictive of early relapse. Emerging data show that the identification of ≥ 0.1% MRD by multiparameter flow cytometry (MFC) in patients with AML in remission after IC is an important prognostic marker that may help guide treatment (Tx) decisions. CC-486 is an oral hypomethylating agent that allows for extended dosing schedules to prolong drug exposure over the Tx cycle. In the QUAZAR AML-001 Maintenance Trial, Tx with CC-486 300 mg QD for 14 days/28-day Tx cycle was associated with significantly improved overall (OS) and relapse-free survival (RFS) vs. placebo (PBO) in patients (pts) with AML in first remission after induction chemotherapy ± consolidation. Samples for MFC were obtained prior to randomization and serially throughout the study to assess the impact of MRD on OS and RFS, and to evaluate rates of conversion from MRD positivity (+) to negativity (–) in the CC-486 and PBO arms.
METHODS: Eligible pts aged ≥ 55 years with AML were randomized 1:1 to CC-486 300 mg or PBO within 4 months of achieving first complete remission (CR) or CR with incomplete blood count recovery (CRi). MFC assessments of bone marrow aspirates were performed centrally at screening; at cycles 3, 6, 9, 12, 15, 18, 21, 24, 30, and 36; and as clinically indicated. Samples were analyzed with a panel of 22 cell surface markers using an MRD+ cutoff of ≥ 0.1% (per ELN MRD guidelines). For pts MRD+ at baseline (BL; ie, at randomization), an MRD response was defined as achievement of MRD– for ≥ 2 consecutive assessments. MRD– duration was calculated from the time of randomization (for pts MRD– at BL) or from the first of ≥ 2 consecutive MRD– tests (for pts MRD+ at BL), until the last MRD– assessment (for pts who became MRD+) or Tx discontinuation. OS, RFS, and MRD– durations were estimated using Kaplan-Meier methods. Multivariate (MV) Cox regression analyses were performed to evaluate the association of BL MRD status (MRD+ vs. MRD–) and randomized Tx arm (CC-486 vs. PBO) with OS and RFS.
RESULTS: The MRD-evaluable cohort comprised 463/472 randomized pts (98.1%; CC-486, n=236; PBO, n=227) who had samples available for evaluation at BL and at ≥ 1 post-BL visit. At BL, 43% of pts (n=103) in the CC-486 arm and 50% (n=116) in the PBO arm were MRD+. Overall, BL characteristics were similar between MRD+ and MRD– pts: median ages were 69 (range 55–84) and 68 (55–86) years, respectively; 84% and 88% had intermediate-risk cytogenetics at diagnosis; 52% and 46% of pts had an ECOG PS of 0; and 79% and 82% received ≥ 1 cycle of consolidation after induction.
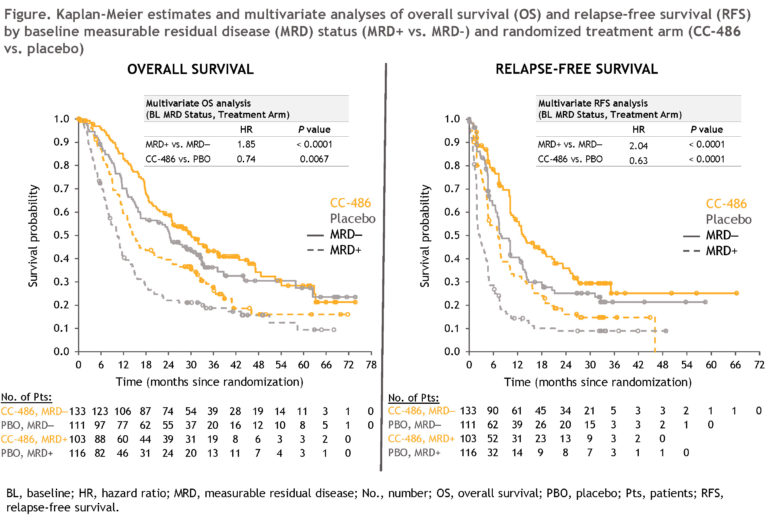
CC-486 Tx resulted in improved OS from time of randomization compared with PBO in pts who were either MRD+ (median 14.6 vs. 10.4 mo, respectively; HR 0.69 [95%CI 0.51, 0.93]) or MRD– (median 30.1 vs. 24.3 mo; HR 0.81 [0.59, 1.12]) at BL. Median RFS was also extended with CC-486 vs. PBO for both MRD+ (7.1 vs. 2.7 mo, respectively; HR 0.58 [95%CI 0.43, 0.78]) and MRD– pts (13.4 vs. 7.8 mo; HR 0.71 [0.52, 0.98]). In MV analyses, BL MRD status (MRD+ vs. MRD–) was significantly associated with OS (HR 1.85; P < 0.0001) and RFS (HR 2.04; P < 0.0001), and CC-486 showed a significant Tx benefit vs. PBO on both OS (HR 0.74; P = 0.0067) and RFS (HR 0.63; P < 0.0001) independent of MRD status at BL (Figure).
The median duration of MRD negativity was extended with CC-486 vs. PBO: 11.0 vs. 5.0 mo, respectively (HR 0.62 [95%CI 0.48, 0.78]). Tx with CC-486 also resulted in a higher rate of MRD response (MRD+ to MRD–) vs. PBO: 37% vs. 19%, respectively. Among MRD responders, 9/38 patients (24%) in the CC-486 arm achieved MRD negativity > 6 mo after randomization, compared with only 1/22 patients (5%) in the PBO arm.
CONCLUSIONS: The QUAZAR AML-001 Maintenance Trial was the first prospective, randomized trial to include long-term longitudinal assessment of MRD in older patients with AML in remission. In both treatment arms, MRD+ status (≥ 0.1%) after induction ± consolidation was associated with significantly shorter OS and RFS compared with MRD– status. Approximately one-fourth of MRD responders treated with CC-486 achieved MRD negativity > 6 mo after study entry, suggesting that CC-486 could induce MRD negativity after prolonged MRD+ status. Maintenance Tx with CC-486 substantially improved OS and RFS independent of MRD status at BL.
The Word to HTML converter helps you convert documents like Excel, Google docs, PDF files and any other visual document to HTML.
111 – Escalated Dosing Schedules of CC-486 Are Effective and Well Tolerated for Patients Experiencing First Acute Myeloid Leukemia (AML) Relapse: Results from the Phase III QUAZAR AML-001 Maintenance Trial
Author(s): Hartmut Döhner1, Andrew H Wei, MBBS, PhD2,3, Pau Montesinos, MD, PhD4,5*, Hervé Dombret, MD6,7, Farhad Ravandi, MBBS8, Hamid Sayar, MD, MSc9, Kimmo Porkka10,11, Irwindeep Sandhu, MD12*, Francesco Passamonti13*, Fabrizio Pane, MD14, Tadeusz Robak, MD PhD15, José F. Falantes16*, Andre C. Schuh17, Gert Ossenkoppele, MD, PhD18*, Ignazia La Torre19*, Barry Skikne, MD20,21*, Keshava Kumar, PhD21*, Qian Dong, DrPH21*, C.L. Beach, PharmD21* and Gail J. Roboz, MD22
1Ulm University Hospital, Ulm, Germany
2The Alfred Hospital, Melbourne, Australia
3Australian Centre for Blood Diseases, Monash University, Melbourne, Australia
4CIBERONC, Instituto de Salud Carlos III, Madrid, Spain
5Hematology Department, Hospital Universitario La Fe de Valencia,, Valencia, Spain, Spain
6Institut de Recherche Saint Louis, Université de Paris, Paris, France
7Hôpital Saint-Louis, Assistance Publique – Hôpitaux de Paris (AP-HP), Paris, France
8Department of Leukemia, University of Texas- MD Anderson Cancer Center, Houston, TX
9Indiana University Cancer Center, Indianapolis, IN
10iCAN Digital Precision Cancer Medicine Flagship, University of Helsinki, Helsinki, Finland
11Hematology Research Unit Helsinki, Helsinki University Hospital Comprehensive Cancer Center, Helsinki, Finland
12University of Alberta Hospital, Edmonton, Canada
13Department of Medicine and Surgery, University of Insubria, Varese, Italy
14Azienda Ospedaliera Universitaria Federico II, Naples, Italy
15Department of Hematology, Medical University of Lodz, Copernicus Memorial Hospital, Lodz, Poland
16Hospital Virgen del Rocío. Hematología, Seville, Spain
17Princess Margaret Cancer Centre, Toronto, ON, Canada
18Amsterdam UMC, Location VU University Medical Center, Amsterdam, Netherlands
19Celgene, a Bristol-Myers Squibb Company, Boudry, Switzerland
20Kansas University Medical Center, Kansas City, KS
21Bristol Myers Squibb, Princeton, NJ
22Weill Medical College of Cornell University New York-Presbyterian Hospital, New York, NY
*signifies non-member of ASH
Disclosures: Döhner: Helsinn: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Oxford Biomedicals: Consultancy, Honoraria; Bristol Myers Squibb: Consultancy, Honoraria, Research Funding; Astex: Consultancy, Honoraria; Astellas: Consultancy, Honoraria, Research Funding; AROG: Research Funding; Amgen: Consultancy, Honoraria, Research Funding; Agios: Consultancy, Honoraria, Research Funding; Abbvie: Consultancy, Honoraria; Jazz: Consultancy, Honoraria, Research Funding; Sunesis: Research Funding; AstraZeneca: Consultancy, Honoraria; GEMoaB: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Pfizer: Research Funding; Roche: Consultancy, Honoraria. Wei: Servier: Consultancy, Honoraria, Research Funding; Pfizer: Honoraria; Abbvie: Honoraria, Research Funding, Speakers Bureau; Macrogenics: Honoraria; Janssen: Honoraria; Novartis: Honoraria, Research Funding, Speakers Bureau; Amgen: Honoraria, Research Funding; Roche: Honoraria; Walter and Eliza Hall Institute of Medical Research: Patents & Royalties: AW is eligible for royalty payments related to venetoclax; Astra Zeneca: Honoraria, Research Funding; Bristol Myers Squibb: Honoraria, Research Funding, Speakers Bureau. Dombret: Immunogen: Consultancy; Otsuka: Consultancy; Abbvie: Consultancy; Cellectis: Consultancy; Shire-Baxalta: Consultancy; Janssen: Consultancy; Menarini: Consultancy; Astellas: Consultancy; Daiichi Sankyo: Consultancy; Servier: Consultancy, Research Funding; Sunesis: Consultancy; Amgen: Consultancy, Research Funding; Jazz Pharma: Consultancy, Research Funding; Celgene: Consultancy; Nova: Consultancy, Research Funding; Incyte: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding. Ravandi: Jazz Pharmaceuticals: Consultancy, Honoraria, Research Funding; BMS: Consultancy, Honoraria, Research Funding; Amgen: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria; Xencor: Consultancy, Honoraria, Research Funding; Orsenix: Consultancy, Honoraria, Research Funding; Macrogenics: Research Funding; Abbvie: Consultancy, Honoraria, Research Funding; Astellas: Consultancy, Honoraria, Research Funding; AstraZeneca: Consultancy, Honoraria. Sayar: Bristol Myers Squibb: Membership on an entity’s Board of Directors or advisory committees. Porkka: BMS/Celgene: Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding. Sandhu: Bristol Myers Squibb: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Janssen: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Amgen: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Takeda: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Pfizer: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Sanofi/Bioverativ: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Gilead: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees. Passamonti: Novartis: Speakers Bureau; Roche: Other: Support of parent study and funding of editorial support; BMS: Speakers Bureau. Pane: Amgen: Consultancy, Other: Travel Expenses, Speakers Bureau; AbbVie: Consultancy, Other: Travel Expenses, Speakers Bureau; Daiichi Sankyo: Consultancy, Other: Travel Expenses; Jazz Pharmaceuticals: Consultancy, Other: travel expenses, Speakers Bureau; Novartis pharma SAS: Consultancy, Other: Travel Expenses, Research Funding, Speakers Bureau; Janssen: Other: Travel Expenses; Bristol Myers Squibb: Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; Celgene: Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau. Robak: GSK: Research Funding; Sandoz: Consultancy, Honoraria; UTX-TGR: Research Funding; Momenta: Consultancy; Takeda: Consultancy; Acerta: Research Funding; BioGene: Honoraria, Research Funding; Octapharma: Honoraria; AstraZeneca: Honoraria, Research Funding; Medical University of Lodz: Current Employment; AbbVie: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Research Funding; Pharmacyclics LLC, an AbbVie Company: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Morphosys: Research Funding; Roche: Consultancy, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Research Funding; Bristol Meyers Squibb: Research Funding; Pfizer: Research Funding; Janssen: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Research Funding; UCB: Honoraria, Research Funding. Falantes: Bristol Myers Squibb: Membership on an entity’s Board of Directors or advisory committees; Celgene: Membership on an entity’s Board of Directors or advisory committees; Novartis: Membership on an entity’s Board of Directors or advisory committees. Ossenkoppele: Genentech: Consultancy, Research Funding; Novartis: Consultancy, Research Funding; Amgen: Consultancy; Daiichi Sayko: Consultancy; Astellas: Consultancy; Jazz: Consultancy; Celgene: Consultancy, Research Funding; Roche: Consultancy; J&J: Consultancy, Research Funding; Agios: Consultancy. La Torre: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Skikne: Bristol Myers Squibb: Current Employment. Kumar: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Dong: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Beach: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Roboz: Otsuka: Consultancy; Takeda: Consultancy; Trovagene: Consultancy; Cellectis: Research Funding; Jasper Therapeutics: Consultancy; Epizyme: Consultancy; Helsinn: Consultancy; MEI Pharma: Consultancy; Amgen: Consultancy; GlaxoSmithKline: Consultancy; Bristol Myers Squibb: Consultancy; Mesoblast: Consultancy; Agios: Consultancy; Orsenix: Consultancy; Sandoz: Consultancy; Actinium: Consultancy; Argenx: Consultancy; Astellas: Consultancy; Daiichi Sankyo: Consultancy; AstraZeneca: Consultancy; Amphivena: Consultancy; Celgene: Consultancy; Astex: Consultancy; Novartis: Consultancy; Janssen: Consultancy; Abbvie: Consultancy; Pfizer: Consultancy; Bayer: Consultancy; Array BioPharma: Consultancy; Eisai: Consultancy; Celltrion: Consultancy; Jazz: Consultancy; Roche/Genentech: Consultancy.
INTRODUCTION: Standard intensive induction chemotherapy (IC) for AML leads to complete remission (CR) in 60%–80% of patients aged ≤ 60 years and in 40%–60% of patients aged > 60 years. However, about two-thirds of patients relapse after frontline therapy, and most relapses occur within the first 18 months (Yilmaz, Blood Cancer J, 2019).
Effective post-induction AML maintenance treatment should decrease the risk of relapse by suppressing growth of residual leukemic cells. CC-486 is an oral hypomethylating agent that allows for extended dosing schedules (> 7 days per 28-day treatment cycle) to sustain therapeutic activity. In the phase III international, randomized, double-blind QUAZAR AML-001 trial (NCT01757535), CC-486 significantly prolonged overall survival (OS) and relapse-free survival (RFS) vs. placebo in patients with AML in first remission following IC, who were not candidates for hematopoietic stem cell transplant (HSCT) (Wei, ASH 2019, LBA-3). Patients initially received CC-486 or placebo for 14 days per 28-day cycle, but patients identified as having early AML relapse with 5–15% blasts in peripheral blood or bone marrow could receive an escalated 21-day/cycle dosing schedule at investigators’ discretion.
OBJECTIVE: Evaluate clinical outcomes in patients in QUAZAR AML-001 who relapsed with 5–15% blasts on-study who then received escalated 21-day dosing of study drug.
METHODS: Eligible patients were aged ≥ 55 years, with intermediate- or poor-risk cytogenetics and Eastern Cooperative Oncology Group performance status (ECOG PS) scores ≤ 3, and had achieved a first CR or CR with incomplete blood count recovery (CRi) after IC ± consolidation. Within 4 months of achieving CR/CRi, patients were randomized 1:1 to receive CC-486 300 mg or placebo once-daily on days 1–14 of repeated 28-day treatment cycles. CR/CRi status was assessed centrally every 3 cycles; patients who exhibited signs of relapse in hematology parameters at routine clinic visits (conducted every 2 weeks) could have an unscheduled bone marrow test to confirm AML relapse. Patients who developed 5%–15% blasts in blood or bone marrow could receive study drug for 21 days/cycle at the investigator’s discretion. Treatment could continue until >15% blasts, unacceptable toxicity, or HSCT.
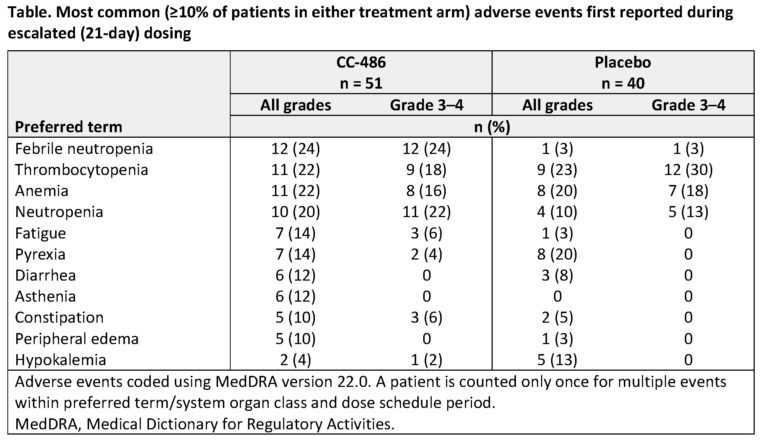
RESULTS: In all, 472 patients were randomized to CC-486 (N=238) or placebo (N=234). During the course of the study, 91 patients (CC-486, n=51 [21%]; placebo, n=40 [17%]) were identified as having early AML relapse with 5–15% blasts and were assigned to receive a 21-day/cycle dosing schedule. Median time to dose escalation of CC-486 was 9.2 months (range 1.0–52.7) and of placebo was 6.0 months (0.5–19.3). Median number of 21-day dosing cycles was 2.0 in both the CC-486 (range 1–45) and placebo (1–16) arms, but proportionally more patients in the CC-486 arm received > 3 escalated dosing cycles (CC-486, 43%; placebo, 18%). Among 78 evaluable patients with ≥ 5% blasts in the most recent bone marrow on or before day 1 of 21-day dosing, 23% (10/43) of patients in the CC-486 arm and 11% (4/35) of patients in the placebo arm regained CR/CRi (< 5% blasts in bone marrow; central review) while receiving an escalated dosing regimen. Among all patients who received escalated dosing schedules, median OS from the time of randomization was 22.8 months in the CC-486 arm vs. 14.6 months in the placebo arm (hazard ratio [HR] 0.66 [95% CI 0.42, 1.0]; P = 0.073), and 1-year survival rates were 80.4% vs. 59.5%, respectively (+20.9% [2.1, 39.7]).
The most common adverse events first reported during 21-day dosing were febrile neutropenia (CC-486, 24%; placebo, 3%), thrombocytopenia (22% and 23%), anemia (22% and 20%), and neutropenia (20% and 10%) (Table). A similar proportion of patients in each arm (CC-486, 31%; placebo, 35%) first experienced a grade 3 or grade 4 adverse event while receiving escalated dosing. CC-486 dose-escalation did not lead to detrimental effects on patient-reported quality of life measures (as assessed by the FACIT-Fatigue and EQ-5D-3L instruments) vs. placebo.
CONCLUSIONS: An escalated 21-day CC-486 dosing regimen was well tolerated and resulted in restoration of remission in approximately one-fourth of patients. Hematologic adverse events first reported during escalated dosing in both treatment arms may be due in part to disease relapse. A 21-day CC-486 dosing schedule could be considered for patients who experience AML relapse with ≤ 15% blasts.
118 – Safety and Preliminary Efficacy in Patients with Relapsed/Refractory Mantle Cell Lymphoma Receiving Lisocabtagene Maraleucel in Transcend NHL 001
Author(s): Maria Lia Palomba, MD1, Leo I. Gordon, MD2, Tanya Siddiqi, MD3*, Jeremy S. Abramson, MD4*, Manali Kamdar, MD5*, Matthew A. Lunning, DO, FACP6, David G. Maloney, MD, PhD7, Charalambos Andreadis, MD, MSCE8*, Jon E. Arnason, MD9, Nilanjan Ghosh, MD, PhD10, Amitkumar Mehta, MD11, Scott R. Solomon, MD12, Thalia Farazi, MD, PhD13*, Jacob Garcia, MD13*, Christine Dehner, BSc13*, Ken Ogasawara, PhD, MPH14*, Jie Gao, PhD14* and Michael Wang, MD15
1Memorial Sloan Kettering Cancer Center, New York, NY
2Northwestern University, Feinberg School of Medicine, Robert H. Lurie Comprehensive Cancer Center, Chicago, IL
3City of Hope National Medical Center, Duarte, CA
4Massachusetts General Hospital Cancer Center, Boston, MA
5University of Colorado Cancer Center, Aurora, CO
6University of Nebraska Medical Center, Omaha, NE
7Fred Hutchinson Cancer Research Center, Seattle, WA
8Helen Diller Family Comprehensive Cancer Center, University of California, San Francisco, San Francisco, CA
9Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA
10Levine Cancer Institute, Atrium Health, Charlotte, NC
11University of Alabama at Birmingham, Birmingham, AL
12Immunotherapy Program, Northside Hospital Cancer Institute, Atlanta, GA
13Juno Therapeutics, a Bristol-Myers Squibb Company, Seattle, WA
14Bristol-Myers Squibb Company, Princeton, NJ
15Department of Lymphoma and Myeloma, U.T. M.D. Anderson Cancer Center, Houston, TX
*signifies non-member of ASH
Disclosures: Palomba: Pharmacyclics: Honoraria; Juno: Honoraria; Celgene: Honoraria; Merck: Honoraria; Novartis: Honoraria; Regeneron: Research Funding; Juno: Research Funding; Genentech: Research Funding. Gordon: Zylem Biosciences: Patents & Royalties: Patents, No Royalties. Siddiqi: Juno: Membership on an entity’s Board of Directors or advisory committees; Kite: Membership on an entity’s Board of Directors or advisory committees; Celgene: Membership on an entity’s Board of Directors or advisory committees; BeiGene: Other: DMC member; Juno Therapeutics, Pharmacyclics LLC, an AbbVie Company, AstraZeneca, Celgene, Kite Pharma, and BeiGene: Consultancy; Pharmacyclics LLC, an AbbVie Company, Seattle Genetics, Janssen, and AstraZeneca: Speakers Bureau; Pharmacyclics LLC, an AbbVie Company, Juno Therapeutics, KITE Pharma, AstraZeneca, TG Therapeutics, Celgene, Oncternal, and BeiGene: Research Funding; AstraZeneca: Other: Travel/accommodations/expenses; Astrazenca: Membership on an entity’s Board of Directors or advisory committees; PCYC: Membership on an entity’s Board of Directors or advisory committees. Abramson: Celgene: Honoraria, Other: Scientific Advisory Board; Juno Therapeutics: Other: Scientific Advisory Board; AbbVie: Other: Scientific Advisory Board; EMD Serono: Other: Scientific Advisory Board; Genentech/Roche: Other: Scientific Advisory Board; Janssen: Other: Scientific Advisory Board; Karyopharm: Other: Scientific Advisory Board; Gilead: Other: Scientific Advisory Board; Verastem: Other: Scientific Advisory Board; Bayer: Other: Scientific Advisory Board; Merck: Other; KIte Pharma: Other; Novartis: Other; Amgen: Other; Seattle Genetics: Other; Allogene: Other; Morphosys: Other; C4 Therapeutics: Other; BeiGene: Other; AstraZeneca: Honoraria; Incyte: Honoraria. Kamdar: Seattle Genetics: Speakers Bureau; Karyopharm: Consultancy; BMS: Consultancy; Abbvie: Consultancy; AstraZeneca: Consultancy; Pharmacyclics: Consultancy. Lunning: Acrotech: Consultancy; ADC Therapeutics: Consultancy; Bristol Meyers Squibb: Consultancy, Honoraria, Research Funding; Curis: Research Funding; Gilead: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Karyopharm: Consultancy, Honoraria; Kite: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; TG Therapeutics: Research Funding; Verastem: Consultancy, Honoraria; AstraZeneca: Consultancy, Honoraria; Legend: Consultancy; Beigene: Consultancy, Honoraria; Aeratech: Consultancy, Honoraria. Maloney: Kite, a Gilead Company: Consultancy, Honoraria, Research Funding; MorphoSys: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Pharmacyclics: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Gilead Sciences: Consultancy, Honoraria; A2 Biotherapeutics: Consultancy, Current equity holder in publicly-traded company, Honoraria; Juno Therapeutics: Consultancy, Honoraria, Patents & Royalties: Patents are pending, but not issued, licensed, no royalties, no licensees., Research Funding; Bioline Rx: Consultancy, Honoraria; Genentech: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Research Funding. Andreadis: Genentech: Other: Spouse Employee (salary and stock); Novartis: Research Funding; Celgene/Juno: Research Funding; Amgen: Research Funding; Merck: Research Funding; Gilead/Kite: Other: Advisor; Jazz Pharmaceuticals: Other: Advisor; Astellas: Other: Advisor; Seattle Genetics: Other: Advisor; Karyopharm: Other: Advisor; Incyte: Other. Arnason: Regeneron: Consultancy; Juno: Consultancy. Ghosh: Forty Seven Inc: Consultancy, Other: Research Bureau, Research Funding; Genmab: Consultancy, Speakers Bureau; AbbVie: Speakers Bureau; Karyopharm: Consultancy; Juno/Celgene/Bristol-Myers Squibb: Consultancy, Research Funding; Kite/Gilead: Consultancy, Speakers Bureau; AstraZeneca: Speakers Bureau; Roche/Genentech: Research Funding; SGN: Consultancy, Research Funding, Speakers Bureau; TG Therapeutics: Consultancy, Research Funding; Celgene/Bristol-Myers Squibb: Speakers Bureau; Janssen: Consultancy, Research Funding, Speakers Bureau; Pharmacyclics LLC, an AbbVie Company: Consultancy, Research Funding, Speakers Bureau. Mehta: Innate Pharmaceuticals: Research Funding; Kite/Gilead: Research Funding; Merck: Research Funding; Gelgene/BMS: Research Funding; Oncotartis: Research Funding; TG Therapeutics: Consultancy, Membership on an entity’s Board of Directors or advisory committees, Research Funding, Speakers Bureau; Seattle Genetics: Consultancy, Membership on an entity’s Board of Directors or advisory committees, Research Funding, Speakers Bureau; Juno Parmaceuticals/BMS: Research Funding; fortyseven Inc/Gilead: Research Funding; Takeda: Research Funding; Roche-Genentech: Research Funding; Incyte: Consultancy, Membership on an entity’s Board of Directors or advisory committees, Research Funding, Speakers Bureau; Affimed: Research Funding. Farazi: Juno Therapeutics, a Bristol-Myers Squibb Company: Current Employment; Bristol-Myers Squibb: Current equity holder in publicly-traded company. Garcia: Juno Therapeutics, a Bristol-Myers Squibb Company: Current Employment; Bristol-Myers Squibb Company: Current equity holder in publicly-traded company. Dehner: Juno Therapeutics, a Bristol-Myers Squibb Company: Current Employment; Bristol-Myers Squibb: Current equity holder in publicly-traded company. Ogasawara: Bristol-Myers Squibb: Current Employment; Bristol-Myers Squibb: Current equity holder in publicly-traded company. Gao: Bristol-Myers Squibb: Current equity holder in publicly-traded company; Bristol-Myers Squibb: Current Employment. Wang: Juno: Consultancy, Research Funding; Acerta Pharma: Research Funding; Loxo Oncology: Consultancy, Research Funding; AstraZeneca: Consultancy, Honoraria, Other: Travel, accommodation, expenses, Research Funding; InnoCare: Consultancy; Kite Pharma: Consultancy, Other: Travel, accommodation, expenses, Research Funding; Pharmacyclics: Consultancy, Honoraria, Other: Travel, accommodation, expenses, Research Funding; Janssen: Consultancy, Honoraria, Other: Travel, accommodation, expenses, Research Funding; MoreHealth: Consultancy; Lu Daopei Medical Group: Honoraria; Beijing Medical Award Foundation: Honoraria; OncLive: Honoraria; Molecular Templates: Research Funding; Verastem: Research Funding; Dava Oncology: Honoraria; Guidepoint Global: Consultancy; Pulse Biosciences: Consultancy; Celgene: Consultancy, Other: Travel, accommodation, expenses, Research Funding; Oncternal: Consultancy, Research Funding; Nobel Insights: Consultancy; OMI: Honoraria, Other: Travel, accommodation, expenses; Targeted Oncology: Honoraria; BioInvent: Research Funding; VelosBio: Research Funding.
Background: Mantle cell lymphoma (MCL) is an aggressive subtype of B-cell non-Hodgkin lymphoma (NHL). Most patients with MCL relapse after first-line immunochemotherapy, with poor responses to salvage therapy. Chimeric antigen receptor (CAR) T cell therapy has shown clinical efficacy in patients with relapsed/refractory (R/R) NHL. We report the results of the dose-finding and dose-expansion parts of the ongoing phase 1 TRANSCEND NHL 001 study (NCT02631044) in patients with R/R MCL (MCL cohort) who received lisocabtagene maraleucel (liso-cel), an investigational, CD19-directed, defined composition, 4-1BB CAR T cell product administered at equal target doses of CD8+ and CD4+ CAR+ T cells.
Methods: Eligible patients had confirmed MCL (cyclin D1 expression, t[11;14]) with R/R disease after ≥1 prior line of therapy. After lymphodepleting chemotherapy, patients received liso-cel infusion at 1 of 2 dose levels (DLs): DL1 (50 × 106 CAR+ T cells) or DL2 (100 × 106 CAR+ T cells). Bridging therapy was allowed between leukapheresis and initiation of lymphodepleting chemotherapy. Primary endpoints were safety and objective response rate (ORR). Secondary endpoints included complete response (CR) rate, duration of response, progression-free survival, overall survival, and pharmacokinetics (PK).
Results: At data cutoff, 41 patients had undergone leukapheresis and 32 had received liso-cel (DL1, n = 6; DL2, n = 26). Among the 32 patients who received liso-cel, the median (range) age was 67 (36‒80) years and 27 patients (84%) were male. Twelve patients (37.5%) had blastoid morphology, 23 (72%) had documented Ki67 ≥30%, 7 (22%) had a TP53 mutation, and 11 (34%) had a complex karyotype. Patients had a median (range) sum of the product of perpendicular diameters before lymphodepleting chemotherapy of 28.7 (0–209.6) cm2 and median lactate dehydrogenase of 251.5 (117–811) U/L. Patients had received a median (range) of 3 (1–7) prior systemic therapies, and most (72%) were refractory to their last prior therapy. Of 28 patients (87.5%) who had received a prior Bruton tyrosine kinase inhibitor, 11 (34%) were refractory to the therapy. Seventeen patients (53%) received bridging therapy.
Eighteen patients (56%) had serious treatment-emergent adverse events (TEAEs), and 27 (84%) had grade ≥3 TEAEs, primarily neutropenia (41%), anemia (34%), and thrombocytopenia (31%). Grade ≥3 thrombocytopenia was more frequent at DL2 (n = 9/26 [35%]) than at DL1 (n = 1/6 [17%]). Prolonged grade ≥3 cytopenias (present at study Day 29) occurred in 11 patients (34%). Sixteen patients (50%; DL1, n = 2/6 [33%]; DL2, n = 14/26 [54%]) had cytokine release syndrome (CRS), including 1 grade 4 event at DL2. There were no grade 3 or 5 CRS events. Median (range) time to CRS onset and resolution was 6 (2‒10) days and 4 (2‒9) days, respectively. Nine patients (28%) had neurological events (NEs), all at DL2, including 3 grade 3 NEs. No grade 4 or 5 NEs were reported. Median (range) time to NE onset and resolution was 8 (2‒25) days and 3 (1‒51) days, respectively. Ten patients (31%) received tocilizumab and/or corticosteroids for treatment of CRS and/or NEs. Grade 5 TEAEs occurred in 2 patients (at DL2): one patient with high tumor burden had tumor lysis syndrome and 1 patient had cryptococcal meningoencephalitis. DL2 was selected for dose expansion.
Of 32 patients, 27 responded to liso-cel (ORR, 84%: DL1, n = 4/6 [67%]; DL2, n = 23/26 [88%]), and 19 (59%) achieved a CR (DL1, n = 2/6 [33%]; DL2, n = 17/26 [65%]). Among the 12 patients with blastoid morphology, 9 patients had a response (ORR, 75%), including 7 (58%) who achieved a CR. Overall, the median (range) time to first CR was 1 (1–6) month. At data cutoff, 20 (74%) of 27 responders were censored with an ongoing response or had completed the study. Median (range) follow-up duration was 10.9 (1.2–24.8) months for DL1 and 3.1 (0.4–23.0) months for DL2. Preliminary PK analysis indicated that median maximum expansion was higher among patients at DL2 than at DL1.
Conclusions: In this phase 1 study of patients with R/R MCL, treatment with liso-cel was associated with a low incidence of grade ≥3 CRS and NEs, late onset of CRS/NEs, and promising clinical activity. Dose confirmation is ongoing at DL2 in the MCL cohort.
544 – Transcend CLL 004: Phase 1 Cohort of Lisocabtagene Maraleucel (liso-cel) in Combination with Ibrutinib for Patients with Relapsed/Refractory (R/R) Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (CLL/SLL)
Author(s): William G. Wierda, MD, PhD1, Kathleen A. Dorritie, MD2, Javier Munoz, MD3, Deborah M. Stephens, DO4, Scott R. Solomon, MD5, Heidi H. Gillenwater, MD6*, Lucy Gong, PharmD6*, Lin Yang, PhD6*, Ken Ogasawara, PhD, MPH7*, Jerill Thorpe, BS6* and Tanya Siddiqi, MD8*
1The University of Texas MD Anderson Cancer Center, Houston, TX
2UPMC Hillman Cancer Center, Pittsburgh, PA
3Banner MD Anderson Cancer Center, Gilbert, AZ
4Huntsman Cancer Institute, University of Utah, Salt Lake City, UT
5Immunotherapy Program, Northside Hospital Cancer Institute, Atlanta, GA
6Juno Therapeutics, a Bristol-Myers Squibb Company, Seattle, WA
7Bristol-Myers Squibb Company, Princeton, NJ
8City of Hope National Medical Center, Duarte, CA
*signifies non-member of ASH
Disclosures: Dorritie: Juno Therapeutics: Research Funding; Kite-Gilead: Research Funding. Munoz: Portola: Research Funding; Incyte: Research Funding; Acrotech/Aurobindo: Speakers Bureau; Alexion: Consultancy; Seattle Genetics: Consultancy, Honoraria, Research Funding, Speakers Bureau; Fosunkite: Consultancy; Pharmacyclics: Consultancy, Research Funding, Speakers Bureau; Innovent: Consultancy; Genentech/Roche: Research Funding, Speakers Bureau; Pfizer: Consultancy; Kite, a Gilead Company: Consultancy, Research Funding, Speakers Bureau; Juno/Celgene/BMS: Consultancy, Research Funding, Speakers Bureau; Janssen: Consultancy, Research Funding, Speakers Bureau; Bayer: Consultancy, Research Funding, Speakers Bureau; Beigene: Consultancy, Speakers Bureau; Millenium: Research Funding; Verastem: Speakers Bureau; Merck: Research Funding; AbbVie: Consultancy, Speakers Bureau; AstraZeneca: Speakers Bureau; Kyowa: Consultancy, Honoraria, Speakers Bureau. Stephens: Innate: Consultancy; Verastem: Research Funding; Beigene: Consultancy; Karyopharm: Consultancy, Research Funding; Acerta: Research Funding; Gilead: Research Funding; Juno: Research Funding; MingSight: Research Funding; Arqule: Research Funding; Janssen: Consultancy; Pharmacyclics: Consultancy. Gillenwater: Juno Therapeutics, a Bristol-Myers Squibb Company: Current Employment; Bristol-Myers Squibb: Current equity holder in publicly-traded company. Gong: Juno Therapeutics, a Bristol-Myers Squibb Company: Current Employment; Bristol-Myers Squibb: Current equity holder in publicly-traded company. Yang: Juno Therapeutics, a Bristol-Myers Squibb Company: Current Employment; Bristol-Myers Squibb: Current equity holder in publicly-traded company. Ogasawara: Bristol-Myers Squibb: Current Employment; Bristol-Myers Squibb: Current equity holder in publicly-traded company. Thorpe: Bristol-Myers Squibb: Current equity holder in publicly-traded company; Juno Therapeutics, a Bristol-Myers Squibb Company: Current Employment. Siddiqi: Astrazenca: Membership on an entity’s Board of Directors or advisory committees; PCYC: Membership on an entity’s Board of Directors or advisory committees; Juno: Membership on an entity’s Board of Directors or advisory committees; Kite: Membership on an entity’s Board of Directors or advisory committees; Celgene: Membership on an entity’s Board of Directors or advisory committees; BeiGene: Other: DMC member; Juno Therapeutics, Pharmacyclics LLC, an AbbVie Company, AstraZeneca, Celgene, Kite Pharma, and BeiGene: Consultancy; Pharmacyclics LLC, an AbbVie Company, Juno Therapeutics, KITE Pharma, AstraZeneca, TG Therapeutics, Celgene, Oncternal, and BeiGene: Research Funding; Pharmacyclics LLC, an AbbVie Company, Seattle Genetics, Janssen, and AstraZeneca: Speakers Bureau; AstraZeneca: Other: Travel/accommodations/expenses.
Background: In CLL/SLL, ibrutinib treatment before leukapheresis improved in vivo and ex vivo expansion of the CD19-directed chimeric antigen receptor (CAR) T cell therapy tisagenlecleucel, and concurrent ibrutinib therapy improved engraftment and therapeutic efficacy of anti-CD19 CAR T cells in human xenograft mouse models (Fraietta et al. Blood. 2016;127:1117–27). Recent studies in patients with R/R CLL suggest that CD19-directed CAR T cell therapy combined with ibrutinib improves response rates with CTL119 and JCAR014 (Gill et al. Blood. 2018;132:298; Gauthier et al. Blood. 2020;135:1650–60). Liso-cel is an investigational, CD19-directed, defined composition, 4-1BB CAR T cell product administered at equal doses of CD8+ and CD4+ CAR+ T cells. We report initial safety and preliminary efficacy from the phase 1 liso-cel and ibrutinib combination cohort of the ongoing phase 1/2 TRANSCEND CLL 004 study (NCT03331198) in patients with R/R CLL/SLL.
Methods: Eligible patients with CLL/SLL met ≥1 of the following: 1) received ibrutinib and progressed at time of study enrollment; 2) had high-risk features and received ibrutinib for ≥6 months (mo) with less than a complete response (CR); 3) had a Bruton tyrosine kinase (BTK) or PLCγ2 gene mutation, with or without progression on ibrutinib; 4) had received prior ibrutinib with no contraindication to reinitiating ibrutinib. Baseline disease assessments included bone marrow (BM) biopsy, complete blood count, lymphocyte enumeration, and CT scan. At enrollment, patients started or continued ibrutinib. Patients continued ibrutinib through leukapheresis and for ≥90 days after liso-cel infusion. Patients received liso-cel infusion at 50 × 106 (dose level [DL]1) or 100 × 106 (DL2) CAR+ T cells after 3 days of lymphodepletion with fludarabine/cyclophosphamide. Primary endpoints were safety and to determine the recommended dose (RD) of liso-cel in combination with ibrutinib for R/R CLL/SLL; overall response (OR) rate (CR + CR with incomplete blood count recovery [CRi] + partial response) and pharmacokinetics (PK) were exploratory endpoints. The RD was selected based on the modified toxicity probability interval algorithm.
Results: At data cutoff, 19 patients received liso-cel (DL1, n=4; DL2, n=15) with ibrutinib. Median age was 60 (range, 50‒77) years, and 18 patients (95%) had high-risk cytogenetics (del[17p], n=8; TP53 mutation, n=6; unmutated IGHV, n=16). Patients had a median of 4 (range, 2‒11) prior therapies. All patients were R/R to prior ibrutinib; 14 patients (74%) had BTK inhibitor as last prior therapy and 10 (53%) had prior venetoclax. No dose-limiting toxicities were observed at either DL. The most common grade ≥3 treatment-emergent adverse events (TEAEs) were neutropenia/neutrophil count decrease (n=17; 89%), anemia (n=9; 47%), and febrile neutropenia (n=5; 26%; Table). Six patients had infections at DL2: grade 3 and grade 2 lung infection (n=1 each) and grade 2 coccidioidomycosis, scabies, skin, and gum infections (n=1 each). Ibrutinib-related AEs included diarrhea (n=7), hypertension (n=4), atrial fibrillation (n=1), and rash (n=1). No grade 5 TEAEs occurred. Fourteen patients (74%) had cytokine release syndrome (CRS; 1 grade 3) and 6 (32%) had neurological events (NEs; 3 grade ≥3). Seven patients (37%) required tocilizumab and/or corticosteroids to manage CRS and/or NEs. Preliminary PK data showed a median time to peak liso-cel expansion of 11 days across DLs (DL1, 12 days; DL2, 11 days). Of 19 patients with ≥1-mo follow-up, 18 (95%) had an OR (DL2, 100%; DL1, 75%) and 9 (47%) had a CR/CRi. One patient (5%) had stable disease. All ORs were achieved by Day 30 postinfusion, and 15 (83%) of 18 patients maintained their response at 3-mo follow-up. Of 19 patients evaluable for minimal residual disease (MRD), 17 (89%) achieved undetectable MRD in blood via flow cytometry and 15 (79%) in BM by next-generation sequencing (both sensitivity of ≤10–4).
Conclusions: Preliminary data show that liso-cel in combination with ibrutinib is associated with manageable safety, including a low incidence of grade 3 CRS and grade ≥3 NEs, and promising efficacy in heavily pretreated patients with R/R CLL/SLL. No clear difference in safety was observed across DLs, and DL2 was selected as the RD for liso-cel in combination with ibrutinib in patients with R/R CLL/SLL. Updated results from the full combination cohort and additional PK/pharmacodynamic data will be reported.
You can find the best online HTML, CSS and JavaScript tools in one place. Online editors, code optimizers and more.
2116 – Matching-Adjusted Indirect Comparison (MAIC) of Lisocabtagene Maraleucel (liso-cel) Vs Axicabtagene Ciloleucel (axi-cel) and Tisagenlecleucel in Relapsed/Refractory (R/R) Large B-Cell Lymphoma (LBCL)
Author(s): David G. Maloney, MD, PhD1, John Kuruvilla, MD2, Christopher P Fox, MD, PhD3*, Guillaume Cartron, MD, PhD4*, Daniel Li, PhD5*, Jens Hasskarl, MD6*, Ashley Bonner, PhD7*, Yixie Zhang, PhD7* and Fei Fei Liu, BSc, MBA8*
1Fred Hutchinson Cancer Research Center, Seattle, WA
2Princess Margaret Cancer Centre, Toronto, ON, Canada
3Nottingham University Hospitals NHS Trust, Nottingham, United Kingdom
4Centre Hospitalier Universitaire de Montpellier, Montpellier, France
5Juno Therapeutics, a Bristol-Meyers Squibb Company, Seattle, WA
6Celgene, a Bristol-Myers Squibb Company, Boudry, Switzerland
7Eversana, Burlington, ON, Canada
8Bristol Myers Squibb, Princeton, NJ
*signifies non-member of ASH
Disclosures: Maloney: A2 Biotherapeutics: Consultancy, Current equity holder in publicly-traded company, Honoraria; Bioline Rx: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Genentech: Consultancy, Honoraria; MorphoSys: Consultancy, Honoraria; Pharmacyclics: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Gilead Sciences: Consultancy, Honoraria; Kite, a Gilead Company: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Juno Therapeutics: Consultancy, Honoraria, Patents & Royalties: Patents are pending, but not issued, licensed, no royalties, no licensees., Research Funding. Kuruvilla: Bristol-Myers Squibb Company: Consultancy; AbbVie: Consultancy; Antengene: Honoraria; TG Therapeutics: Honoraria; Novartis: Honoraria; Pfizer: Honoraria; Seattle Genetics: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; Celgene Corporation: Honoraria; Amgen: Honoraria; Roche: Consultancy, Honoraria, Research Funding; Karyopharm: Consultancy, Honoraria; Merck: Consultancy, Honoraria; Janssen: Honoraria, Research Funding; AstraZeneca Pharmaceuticals LP: Honoraria, Research Funding. Fox: AstraZeneca: Research Funding; Celgene: Research Funding; Sunesis: Research Funding; AbbVie: Honoraria, Research Funding; Roche: Honoraria, Research Funding; Takeda: Honoraria, Research Funding; Gilead: Honoraria, Research Funding; Adienne: Honoraria, Research Funding; Atarabio: Research Funding. Cartron: F. Hoffmann-La Roche: Consultancy, Honoraria; Abbvie: Honoraria; Sanofi: Honoraria; Celgene: Consultancy, Honoraria; Gilead: Honoraria; Jansen: Honoraria. Li: Juno Therapeutics, a Bristol-Myers Squibb Company: Current Employment; Bristol-Myers Squibb: Current equity holder in publicly-traded company. Hasskarl: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Bonner: Eversana: Current Employment. Zhang: Eversana: Current Employment. Liu: Bristol-Myers Squibb: Current Employment, Current equity holder in publicly-traded company.
Introduction: Chimeric antigen receptor T cell therapies have shown encouraging, durable responses in patients with R/R LBCL, yet no head-to-head clinical trials comparing options exist to date. We conducted 2 separate pair-wise MAICs to compare treatment effects of liso-cel vs both axi-cel and tisagenlecleucel.
Methods: MAICs were used to estimate population-adjusted relative treatment effects associated with liso-cel (TRANSCEND NHL 001 [TRANSCEND]; NCT02631044; N = 256) vs axi-cel (ZUMA-1; NCT02348216; N = 101) and vs tisagenlecleucel (JULIET; NCT02445248; N = 111). Outcomes of interest included efficacy (overall and complete response rates [ORR/CRR], overall survival [OS], and progression-free survival [PFS]) and safety (cytokine release syndrome [CRS] by Lee criteria, neurological events [NEs], aphasia, encephalopathy, infections, hypogammaglobulinemia, and prolonged cytopenia).
Individual patient data (IPD) from TRANSCEND were adjusted to match the marginal distribution (eg, mean, variance) of clinical factors among patients from ZUMA-1 and JULIET. Patients from TRANSCEND were removed from the IPD set if they did not satisfy eligibility criteria specified in the comparator trial for each MAIC. IPD for patients who remained in the TRANSCEND data set were weighted using a method-of-moments propensity score model. Baseline characteristic and outcome definitions were aligned with those in ZUMA-1 or JULIET. Clinically relevant prognostic factors (identified from literature, TRANSCEND data, and 5 independent clinical experts) were adjusted collectively in a stepwise fashion by ranked order. Key matched and adjusted variables in 1 or both comparisons included: disease histology, Eastern Cooperative Oncology Group performance status (ECOG PS), central nervous system (CNS) involvement, prior allogeneic/autologous hematopoietic stem cell transplant (HSCT), tumor burden, International Prognostic Index score, response to last therapy, bulky disease, and age. Efficacy outcomes in patients without bridging therapy were evaluated; however, ZUMA-1 and TRANSCEND treatment protocols differed in bridging therapy use (not allowed in ZUMA-1) and time to product availability (median, 17 vs 24 days, respectively).
Results: After aligning definitions of baseline characteristics among trials, substantial differences were noted for ECOG PS of 2, tumor burden, active CNS involvement, number of prior lines of therapy, prior allogeneic HSCT, and history of hematologic comorbidities between studies. Overall, TRANSCEND included a larger sample size and broader patient population vs comparator trials, allowing for successful MAIC adjustments.
When comparing TRANSCEND to ZUMA-1, MAIC-weighted efficacy outcomes were comparable between trials: odds ratios (ORs [95% CI]) for ORR and CRR with liso-cel vs axi-cel were 0.85 (0.48–1.52) and 0.78 (0.47–1.27), respectively; hazard ratios (HRs [95% CI]) for OS and PFS were 1.15 (0.80–1.65) and 1.30 (0.96–1.77), respectively (Figure). When limited to patients without bridging therapy, differences between trials remained statistically insignificant. MAIC-weighted safety outcomes showed a favorable safety profile for liso-cel, with a statistically significant lower odds of CRS, NEs (including aphasia and encephalopathy), and infections vs axi-cel. ORs (95% CI) for all-grade and grade ≥3 CRS with liso-cel vs axi-cel were 0.06 (0.03–0.13) and 0.16 (0.06–0.47), respectively; ORs for all-grade and grade ≥3 NEs were 0.21 (0.13–0.35) and 0.31 (0.18–0.54), respectively.
When comparing TRANSCEND to JULIET, liso-cel showed a statistically significant higher ORR/CRR and longer OS/PFS than tisagenlecleucel. ORs (95% CI) for ORR and CRR achieved with liso-cel vs tisagenlecleucel were 2.78 (1.63–4.74) and 2.01 (1.22–3.30), respectively; HRs (95% CI) for OS and PFS were 0.67 (0.47–0.95) and 0.65 (0.47–0.91), respectively. Adjusted safety outcomes showed generally comparable profiles with lower ORs (95% CI) for all-grade and grade ≥3 CRS with liso-cel vs tisagenlecleucel: 0.53 (0.32–0.89) and 0.10 (0.03–0.31), respectively.
Conclusions: MAIC-weighted outcomes suggest that liso-cel may provide a more well-balanced overall efficacy and safety profile for the treatment of R/R LBCL, with better efficacy compared with tisagenlecleucel and better safety compared with axi-cel.
554 – CRISPR-Cas9 Knock out of CD5 Enhances the Anti-Tumor Activity of Chimeric Antigen Receptor T Cells
Author(s): Inkook Chun, MS1,2*, Ki Hyun Kim, PhD1*, Yi-Hao Chiang, MD1,3*, Wei Xie, MD1*, Yong Gu Gu Lee, PhD1,2, Raymone Pajarillo, MS1,2*, Antonia Rotolo, MD, PhD1*, Olga Shestova, PhD1,2*, Seok Jae Hong, MS1*, Mohamed Abdel-Mohsen, PhD4*, Maria Wysocka, PhD5*, Hatcher J. Ballard, BS2*, David M. Barrett6*, Avery D. Posey, PhD1, Daniel Powell Jr., PhD1*, Saar I. Gill, MD, PhD1,7, Stephen J. Schuster, MD2, Stefan K. Barta, MD, MRCP, MS1,2, Alain H Rook, MD5*, Carl H June, MD1 and Marco Ruella, MD1,7
1Center for Cellular Immunotherapies, University of Pennsylvania, Philadelphia, PA
2Division of Hematology-Oncology, Hospital of the University of Pennsylvania, Philadelphia, PA
3Division of Hematology-Oncology, MacKay Memorial Hospital, Taipei, Taiwan
4Vaccine and Immunotherapy Center, The Wistar Institute, Philadelphia, PA
5Division of Dermatology, Hospital of the University of Pennsylvania, Philadelphia, PA
6Division of Oncology, Children’s Hospital of Philadelphia, Philadelphia, PA
7Division of Hematology-Oncology, University of Pennsylvania, Philadelphia, PA
*signifies non-member of ASH
Disclosures: Schuster: Novartis, Genentech, Inc./ F. Hoffmann-La Roche: Research Funding; AlloGene, AstraZeneca, BeiGene, Genentech, Inc./ F. Hoffmann-La Roche, Juno/Celgene, Loxo Oncology, Nordic Nanovector, Novartis, Tessa Therapeutics: Consultancy, Honoraria. Barta: Pfizer: Honoraria; Acrotech: Honoraria; Janssen: Honoraria; Seattle Genetics: Honoraria, Research Funding; Atara: Honoraria; Monsanto: Consultancy. June: Tmunity Therapeutics: Current equity holder in private company, Membership on an entity’s Board of Directors or advisory committees, Patents & Royalties, Research Funding; Ziopharm Oncology: Current equity holder in private company, Membership on an entity’s Board of Directors or advisory committees; Novartis: Patents & Royalties, Research Funding; Bluesphere Bio: Membership on an entity’s Board of Directors or advisory committees; Cabaletta Bio: Current equity holder in private company, Membership on an entity’s Board of Directors or advisory committees; Carisma Therapeutics: Membership on an entity’s Board of Directors or advisory committees; Cellares: Membership on an entity’s Board of Directors or advisory committees; Celldex: Consultancy, Membership on an entity’s Board of Directors or advisory committees; DeCART Therapeutics: Membership on an entity’s Board of Directors or advisory committees; Immune Design: Membership on an entity’s Board of Directors or advisory committees; Kiadis Pharma: Current equity holder in private company. Ruella: Abclon, BMS, NanoString: Consultancy; UPenn/Novartis: Patents & Royalties; Abclon: Consultancy, Research Funding.
Chimeric Antigen Receptor T cells (CART) have led to unprecedented clinical responses in relapsed or refractory (r/r) B-cell acute lymphoblastic leukemia (B-ALL), non-Hodgkin lymphomas (NHL), and multiple myeloma. However, despite these exciting results, most patients treated with CART therapy either do not respond or eventually relapse. Moreover, CART therapy has not yet been proven effective in several hematological malignancies, such as T cell lymphoma and leukemia (T-NHL/T-ALL) and acute myeloid leukemia (AML). Thus, there is a need to enhance currently available CART products and also to develop next-generation CART therapies to successfully treat additional neoplasms like T-NHL /T-ALL and AML.
To this goal, we studied the cysteine-rich scavenger receptor CD5, an attractive target for CART immunotherapy because of its dual role in malignant cells and normal T cells. In malignant cells, CD5 is expressed by ~90% of TCL cells, by ~15-20% of AML cells, and also by most cases of chronic lymphocytic leukemia and mantle cell lymphoma (MCL). Of note, promising results using a CD28-based anti-CD5 CART against T-NHL and T-ALL were reported at this meeting in 2019 (LaQuisa C. Hill #199). In T cells, CD5 is highly expressed and inhibits T cell receptor (TCR)-mediated activation through several mediators including SHP-1, CBL, CBL-B, and GRB2. Therefore, we hypothesized that the genetic deletion of CD5 in engineered T cells could potentially enhance their effector functions.
First, we designed and screened six 4-1BB-costimulated anti-CD5 lentiviral CAR constructs designed to have high, medium, and low affinity for CD5. We then selected the lead CAR5 construct (high affinity, heavy to light light chain orientation) based on its superior anti-tumor function in vivo in NOD-SCID IL2Rgnull (NSG) mice engrafted with T-cell leukemia (Jurkat). Then, to further improve CART5 activity, we optimized a CD5 short-guide RNA and deleted CD5 in CART5 cells using CRISPR-Cas9. CD5 gene deletion was reproducibly efficient (95-100% by flow cytometry and TIDE) during manufacturing (6 donors). Interestingly, the growth rate of wild type (WT) CART5 was comparable to CD5 KO CART5 and the expression of CD5 in WT CART5 was reduced. However, at the end of manufacturing, CD5 KO CART5 had increased central memory T cells (33.0% vs. 18.4%) and reduced expression of activation/exhaustion markers (PD-1 4.4% vs. 14.8%, LAG3 13.1% vs. 55.9%) compared to WT CART5, potentially indicating that CD5 KO reduces CART5-CART5 fratricide during manufacturing.
We then compared wild type (WT) CART5 to CD5 KO CART5 in vitro using several T-NHL/T-ALL, MCL, and AML models, including primary samples (Sezary cells, primary MCL cells, and CD5+ AML cells). Both WT and CD5 KO CART5 were highly effective in killing CD5+ malignant cells, but CD5 KO CART5 showed enhanced proliferation upon activation. In two xenograft models of T-cell leukemia (primary T-ALL and Jurkat), CD5 KO CART5 showed dramatically increased tumor control compared to WT (Fig.1A, median overall survival for WT= 62 days vs. CD5 KO=not reached, p = 0.006, Mantel-Cox). This enhanced anti-tumor effect was associated with increased expansion of CD5 KO CART5 in the peripheral blood (PB) compared to WT CART5.
To test the hypothesis that deletion of CD5 could increase the anti-tumor effect of CART targeting antigens other than CD5, we knocked out CD5 in anti-CD19 CART cells and tested their function in a CD19+ B-ALL xenograft model (NALM6). Remarkably, CD5 KO CART19 displayed significantly enhanced anti-leukemia activity and PB expansion compared to WT (Fig.1B,C, p<0.05, p = 0.001, Mantel-Cox).
Finally, we aimed to define the mechanisms by which CD5 KO enhances CART anti-tumor efficacy. We analyzed the phosphorylation of multiple targets in T cells after 15 minutes of CAR stimulation. Remarkably, CD5 KO CART5 cells had higher (>2fold) phosphorylation of several signaling proteins, including key regulators of T cell activation, migration, and survival compared to WT CART5 (Fig. 1D). To confirm that the CD5 pathway was indeed the mediator of this effect, we knocked out SHP-1 in CART19 cells using CRISPR/Cas9 and observed increased leukemia killing.
In conclusion, we demonstrate that CRISPR-Cas9 KO of CD5 enhances the anti-tumor activity of CAR T cells by enhancement of CAR-mediated activation and proliferation. These findings support the development of CD5 KO CART products in early-phase clinical trials.
555 – The Enhanced Functionality of Low-Affinity CD19 CAR T Cells Is Associated with Activation Priming and Polyfunctional Cytokine Phenotype
Author(s): Ilaria M. Michelozzi, PhD1*, Eduardo Gomez-Castaneda, PhD2*, Ruben V.C. Pohle2*, Ferran Cardoso Rodriguez3*, Jahangir Sufi3*, Pau Puigdevall, PhD4*, Meera Subramaniyam2*, Si Wei Wu2*, Aleks Guvenel, PhD2*, Sara Ghorashian, FRCPath, PhD5,6, Martin Pule, MD PhD7*, Christopher J. Tape, PhD3*, Sergi Castellano, PhD4*, Persis J Amrolia, FRCP, FRCPath, PhD2,8* and Alice Giustacchini, PhD1*
1Molecular and Cellular Immunology Section, UCL Great Ormond Street Institute of Child Health, London, ENG, United Kingdom
2Molecular and Cellular Immunology Section, UCL Great Ormond Street Institute of Child Health, London, United Kingdom
3Cell Communication Lab, Department of Oncology, University College London Cancer Institute, London, United Kingdom
4Genetics and Genomic Medicine Programme, Great Ormond Street Institute of Child Health, University College London, London, United Kingdom
5Department of Haematology, Great Ormond Street Hospital, London, United Kingdom
6Developmental Biology and Cancer, UCL Great Ormond Street Institute of Child Health, London, ENG, United Kingdom
7UCL Cancer Institute, London, United Kingdom
8Bone Marrow Transplantation Department, Great Ormond Street hospital NHS Trust, London, United Kingdom
*signifies non-member of ASH
Disclosures: Ghorashian: Amgen: Honoraria; UCLB: Patents & Royalties; Novartis: Honoraria. Pule: Autolus: Current Employment, Other: owns stock in and receives royalties, Patents & Royalties; UCLB: Patents & Royalties; Mana Therapeutics: Other: entitled to share of revenue from patents filed by UCL.
We have recently described a low-affinity second-generation anti-CD19 Chimeric Antigen Receptor (CAR) (CAT), characterized by faster antigen dissociation rate which showed enhanced expansion, cytotoxicity and anti-tumour efficacy compared with the high affinity (FMC63 based) CAR used in Tisagenlecleucel in pre-clinical models. Furthermore, CAT CAR T cells showed an excellent toxicity profile, enhanced in vivo expansion and long-term persistence in a Phase I clinical study (Ghorashian et al Nature Med 2019). However the molecular mechanisms behind the improved properties of CAT CAR T cells remain unknown. Herein, we performed a systematic in vitro characterization of the transcriptomic (bulk RNA-seq) and protein (CyTOF) changes occurring in CAR T cells expressing a low-affinity (CAT) vs high affinity (FMC63) anti-CD19 CARs following stimulation with CD19 expressing targets.
Untransduced (UT) controls and T cells lentivirally transduced to express CAT or FMC63 CD19 CARs were compared both at baseline and following stimulation with CD19+ Acute Lymphoblastic Leukaemia cell line NALM6. In Principal Component Analysis for both RNA-seq and protein results, we found that the major variance across conditions was explained by CD19-mediated CAR T activation. Strikingly, unstimulated CAT CAR T cells showed an intermediate degree of activation between UT T cells and antigen stimulated CAR T cells. Indeed, when comparing RNA-seq results of unstimulated CAT vs FMC63, we found enhanced expression (FDR <0.1) of genes involved in cytotoxicity (GNLY, GZMK) and T cell activation (HLA-DRA and HLA-DPA1) (Figure 1a), confirmed at protein level by CyTOF. This “activation priming” observed in CAT CAR T cells was associated with and may be driven by residual CD19-expressing B-cells present in the manufacture product, preferentially inducing a T Central Memory (TCM) phenotype in CAT vs FMC63, in both CD4 and CD8 T cells. Such priming is likely to be instrumental to CAT CAR T cells more potent cytotoxic response upon NALM6 stimulation, when they displayed further increase in the expression of immune stimulatory cytokines (IFNG, CSF2), chemokines (CCL3L1, CCL4, CXCL8) and IFNg responsive genes (CIITA) by RNA-seq, as well as augmented T cell activation (CD25, NFAT1) and proliferation (pRB) markers by CyTOF.
To identify the mechanisms underlying the stronger basal activation of CAT CAR T cells, we analysed cytokine expression at the single cell level by mass cytometry. Interestingly, rather than an increment in the expression of individual cytokines, we found that the distinctive feature of CAT CAR T cells was a shift toward a cytokine polyfunctional phenotype, with a marked increase in the proportion of cells co-expressing 3 or more cytokines (17.50% CAT vs 7.33% FMC63) (Figure 1b). Of note, cytokine polyfunctionality (expression of more than 1 cytokine/cell) in pre-infusion CAR T cell products has been associated to improved clinical efficacy. The functional phenotype observed in CAT CAR T cells was linked to the preferential activation of the p38 MAPK phospo-signalling, which is activated downstream of TCR CD3ζ chain (present in the CARs) but is also central to cytokine-dependent T cell activation in memory T cells.
Interestingly, cytokine polyfunctional CAT CAR T cells were enriched in the CD3+CD19+ trogocytic (trog+) population, found at higher proportion in CAT vs FMC63 at 24h post antigen stimulation. Although trogocytosis has been associated to CAR T cell fratricide killing, trog+ CAT CAR T cells displayed higher levels of proliferation (pRB), activation (CD25, NFAT1) and cytotoxic (Granzyme B, Perforin B) markers, pointing at a stimulatory role of trogocytosis over fratricide killing, potentially due to the low-affinity CAR T cells distinctive property of better discriminating between low (trog+ CAR T cells) and high (tumour cells) target expression levels.
In conclusion, we described the molecular mechanisms underlying the low affinity CAT CAR T cells functional phenotype. Our results show that the potent and long-term anti-tumour responses observed with CAT may be sustained by the establishment of CAR T cells self-reinforcing circuits activated through polyfunctional cytokine crosstalk. This work may inform the future design of versatile CAR T cells, capable of balancing safety, efficacy and long-term persistence.
556 – CD58 Aberrations Limit Durable Responses to CD19 CAR in Large B Cell Lymphoma Patients Treated with Axicabtagene Ciloleucel but Can be Overcome through Novel CAR Engineering
Author(s): Robbie G. Majzner, MD1, Matthew J. Frank, MD, PhD2, Christopher Mount, MD, PHD3*, Aidan Tousley3*, David M. Kurtz, MD, PhD4, Brian Sworder, MD, PhD5, Katherine A Murphy6*, Antigoni Manousopoulou7*, Kennedy Kohler3*, Maria Caterina Rotiroti, PhD3*, Jay Y. Spiegel, MD, FRCPC2, Yasodha Natkunam, MD, PhD8, Sheren F. Younes, MD, PhD9*, Elena Sotillo, PhD1*, Vandon Duong3*, Charles Macaulay, MSc, BA10*, Zinaida Good, PhD11, Peng Xu, MD3*, Louai Labanieh1*, Leo D Wang, MD, PhD12, Ash A. Alizadeh, MD, PhD13, Michelle Monje, MD, PHD3*, David B. Miklos, MD, PhD2 and Crystal L. Mackall, MD11
1Department of Pediatrics, Stanford University School of Medicine, Palo Alto, CA
2Division of Blood and Marrow Transplantation, Stanford University, Stanford, CA
3Stanford University School of Medicine, Palo Alto, CA
4Stanford University, San Francisco, CA
5Department of Medicine, Divisions of Hematology & Oncology, Stanford University, Stanford, CA
6Stanford University, Palo Alto, CA
7City of Hope, Duarte, CA
8Department of Pathology, Stanford University Medical Center, Stanford, CA
9Department of Pathology, Stanford University School of Medicine, Stanford, CA
10Department of Medicine, Divisions of Hematology & Oncology, Stanford University Medical Center, Palo Alto, CA
11Center for Cancer Cell Therapy, Stanford University School of Medicine, Stanford, CA
12City of Hope National Medical Center, Beckman Research Institute, Duarte, CA
13Stanford University, Stanford, CA
*signifies non-member of ASH
Disclosures: Majzner: Xyphos Biopharma: Consultancy; Zai Lab: Consultancy; Lyell Immunopharma: Consultancy; GammaDelta Therapeutics: Membership on an entity’s Board of Directors or advisory committees; Aprotum Group: Consultancy; Illumina Radiopharmaceuticals: Consultancy. Kurtz: Foresight Diagnostics: Consultancy, Membership on an entity’s Board of Directors or advisory committees; Genentech: Consultancy; Roche: Consultancy. Sotillo: Lyell Immunopharma: Consultancy, Other: Consultancy. Alizadeh: Roche: Consultancy; Gilead: Consultancy; Celgene: Consultancy; Chugai: Consultancy; Pharmacyclics: Consultancy; Genentech: Consultancy; Janssen: Consultancy; Pfizer: Research Funding. Miklos: Miltenyi Biotec: Research Funding; Janssen: Consultancy, Other: Travel support; Pharmacyclics: Consultancy, Other: Travel support, Patents & Royalties, Research Funding; Novartis: Consultancy, Other: Travel support, Research Funding; Allogene Therapeutics Inc.: Research Funding; Juno-Celgene-Bristol-Myers Squibb: Consultancy, Other: Travel support, Research Funding; Kite-Gilead: Consultancy, Membership on an entity’s Board of Directors or advisory committees, Other: Travel support, Research Funding; Adaptive Biotech: Consultancy, Other: Travel support, Research Funding. Mackall: BMS: Consultancy; Allogene: Current equity holder in publicly-traded company; Apricity Health: Consultancy, Current equity holder in private company; Nektar Therapeutics: Consultancy; NeoImmune Tech: Consultancy; Lyell Immunopharma: Consultancy, Current equity holder in private company.
CD19 CAR T cells have revolutionized the treatment of relapsed and refractory (R/R) large B cell lymphomas (LBCL), mediating durable complete responses in approximately 40-50% of patients. Besides a loss or decrease in CD19 expression, no studies have identified tumor specific factors driving inherent or acquired resistance to CAR T cells in LBCL. Mutations in and loss of expression of LFA-3 (CD58) have been described in approximately 20% of cases of LBCL. As the ligand for CD2 on T cells, CD58 provides costimulation to T cells and CD58 loss or mutation has been linked to immune resistance in LBCL.
We evaluated CD58 status in fifty-one R/R LBCL patients treated at Stanford with commercial axicabtagene ciloleucel (axi-cel) through immunohistochemistry (IHC) on tumor biopsy samples and/or deep sequencing of circulating tumor DNA by CAPP-Seq. We identified 12/51 (24%) patients with a CD58 aberration (lack of expression by IHC or mutation by CAPP-Seq). Progression-free survival (PFS) was significantly decreased in patients with a CD58 aberration (median PFS for CD58 aberration 3 months vs. not reached for CD58 intact, p<0.0001). In fact, only 1/12 patients with a CD58 alteration achieved a durable, complete response to axi-cel, while the remaining 11 patients progressed, most commonly after a period of initial response. Partial responses were more common among patients with CD58 aberrations (58% for CD58 aberration vs 10% for CD58 intact, p<0.001), and complete responses were less common (25% for CD58 aberration vs 82% for CD58 intact, p<0.0001).
To probe the biology of CAR T cell responses towards tumors lacking functional CD58, we generated a CD58 knockout Nalm6 model. CD19.CD28.ζ, CD19.4-1BB.ζ, and CD22.4-1BB.ζ CAR T cells demonstrated significantly reduced cytokine production and cytolytic activity in response to CD58 KO vs wildtype (WT) tumor cells. Additionally, while mice inoculated with WT Nalm6 and treated with any of the three CARs demonstrate complete responses and prolonged leukemia-free survival, mice inoculated with CD58KO Nalm6 demonstrated only partial responses, eventual tumor progression, and death from leukemia.
CD2, the T cell ligand for CD58, plays both an adhesive role and a costimulatory role in T cells. CD2 knockout resulted in significantly reduced cytokine production after CAR stimulation. Re-expression of only the CD2 extracellular domain did not rescue CAR function, indicating that CD2 signaling is essential for full CAR activation. Additionally, when we stimulated CD19 CAR T cells with anti-idiotype antibody (CAR stimulation), soluble CD58 (CD2 stimulation), or both, we observed significantly enhanced phosphorylation of both CD3ζ and ERK by western blot in CAR T cells stimulated through both the CAR and CD2. Phosphorylation analysis by mass spectrometry revealed that CD2 stimulation enhances phosphorylation of proximal signaling molecules in the TCR pathway (LCK, LAT, CD3ε among others) and also mediators of actin-cytoskeletal rearrangement in CAR T cells, consistent with effects in natural T cell responses.
To overcome CD58 loss in LBCL, we generated second- and third-generation CAR T cell constructs integrating CD2 costimulatory domains within the CAR molecule. While these cis constructs demonstrated increased potency against CD58KO cells in vitro, they were unable to ultimately overcome CD58 loss in vivo. However, when CARs were co-expressed with an additional CD2 receptor in trans, they mediated significant anti-tumor activity in vivo, overcoming CD58 knockout in tumor cells.
In conclusion, we have identified that CD58 status is an important biomarker for durable response to CAR T cells in LBCL. We modeled the biologic basis for this finding and generated CAR T cells capable of overcoming CD58 loss in B cell malignancies. CD58 mutations have been reported in many cancers, including multiple myeloma and colon cancer, and are likely to play a role in immune evasion for CAR T cells as they are developed for additional histologies. These data provide rationale for investigating CD58 status for patients receiving CAR based therapeutics and devising next generation CARs capable of overcoming this newly discovered mechanism of resistance.
557 – Loss of TET2 Uncouples Proliferative and Effector Functions in CAR T Cells
Author(s): Nayan Jain1*, Zeguo Zhao, PhD2*, Archana S. Iyer, PhD2*, Michael Lopez2*, Judith Feucht, MD3*, Richard P. Koche, PhD4*, Yingqian Zhan4* and Michel Sadelain, MD, PhD2
1Louis V. Gerstner Jr. Graduate School of Biomedical Sciences, Memorial Sloan Kettering Cancer Center, New York City, NY
2Center for Cell Engineering and Immunology Program, Memorial Sloan Kettering Cancer Center, New York City, NY
3Memorial Sloan Kettering Cancer Center, New York, NY, Germany
4Center for Epigenetics Research, Memorial Sloan Kettering Cancer Center, New York City, NY
*signifies non-member of ASH
Disclosures: Feucht: Fate Therapeutics: Patents & Royalties; TAKEDA Pharmaceuticals: Patents & Royalties; Atara Biotherapeutics: Patents & Royalties. Sadelain: Atara: Patents & Royalties, Research Funding; Mnemo: Patents & Royalties; Minerva: Other: Biotechnologies , Patents & Royalties; Fate Therapeutics: Patents & Royalties, Research Funding; Takeda: Patents & Royalties, Research Funding.
Chimeric antigen receptor (CAR) T cells have opened a new paradigm for the treatment of leukemia and lymphoma. Their production, however, is laborious, requiring tens of millions of CAR T cells per infusion. This constraint could be significantly alleviated if safe and more efficacious T cells could be generated. In a patient with chronic lymphocytic leukemia, treated with anti-CD19 CAR T cells, a recent report described the emergence of a single T cell clone that at its expansion peak accounted for 94% of circulating CAR T cells, coinciding with the development of cytokine release syndrome and tumor regression (Fraietta et. al. Nature 2018). Insertional mutagenesis in this T cell had disrupted an allele of TET2, an epigenetic regulator mediating the oxidation of 5-methylcytosine. The other allele appeared to bear an inherited hypomorphic variant, resulting in the near complete loss of TET2 function in this clone. To understand the mechanisms accounting for this chance clinical finding, we investigated the effect of TET2 loss in human T cells engineered to express different chimeric receptors.
Using CRISPR/Cas9, we edited TET2 in T cells engineered to express a CD19-specific second-generation CAR encompassing the costimulatory domain of either CD28 or 4-1BB (Rv-1928z and Rv-19BBz). TET2 disruption enhanced the in vivo anti-tumor activity of Rv-19BBz but not Rv-1928z CAR T cells tested under stress test conditions using limiting CAR T cell doses (as previously described in a human B cell acute lymphoblastic leukemia (B-ALL) NALM6 model, Zhao et. al. Cancer Cell 2015). Since Rv-1928z induces potent effector differentiation but limited persistence compared to Rv-19BBz, we hypothesized that loss of TET2 could amplify the expansion and persistence of 4-1BB-costimulated T cells but not override the differentiation program imparted by Rv-1928z.
To test this hypothesis, we utilized two orthogonal approaches known to limit exhaustion and increase persistence of CD28-costimulated CAR T cells, Rv-1928z co-expressed with 4-1BB ligand (Rv-1928z-41BBL) and 1928z driven by the TRAC promoter (TRAC-1928z). Disruption of TET2 enhanced the anti-tumor efficacy of both these CAR T cells and promoted acquisition of a central memory phenotype. However, over time (50-200 days), TET2-edited TRAC-1928z and Rv-1928z-41BBL attained a hyper-proliferative phenotype ultimately requiring euthanasia due to splenomegaly and extensive CAR T cell accumulation in various organs. Post-mortem analysis found no evidence of NALM6 in these mice. This was in contrast to stress test studies with Rv-1928z and Rv-19BBz where most mice succumbed to NALM6 progression. These observations established an essential role for CAR signaling in determining the phenotypic outcome of TET2 loss in T cells.
To examine the long-term effects of TET2 disruption in the context of all 4 receptors, we treated human B-ALL bearing mice with curative doses of all 4 CAR T cells and followed them for up to 200 days. We found that all 4 CAR expressing TET2-edited T cells could eventually attain a hyper-proliferative phenotype, but with varying frequency depending on the CAR design (Rv-1928z-41BBL and TRAC-1928z > Rv-19BBz > Rv-1928z). To assess their effector function, NALM6-bearing mice were infused with adoptively transferred hyper-proliferative TET2-edited CAR T cells. Strikingly, these T cells were unable to elicit any tumor control, despite their maintaining a central memory phenotype as assessed by flow cytometry. This loss of effector function was observed for all 4 CAR T cell types, suggesting a discrepancy between function and flow cytometric phenotype.
Transcriptional, methylation and genome accessibility studies revealed a unique T cell state wherein the proliferative program is uncoupled from effector response. We identified a unique transcriptional and epigenetic signature that is manifested in a loss of effector function while maintaining robust proliferation. This state stands in contrast to the classically described T cell exhaustion state where loss of effector function is preceded by loss of proliferative ability. TET2 disruption thus promotes a CAR T cell proliferative program that depends on the CAR design but does not in itself enhance anti-tumor activity.
558 – Dual Inhibition of PI3KDelta/Gamma during Manufacturing Reprograms Metabolism of CAR T Cells to Enhance Expansion and Cytotoxicity Against CLL
Author(s): Christopher Ronald Funk1, Shuhua Wang, MD1*, Alex Waller, BS2*, Claudia Edgar, BS1*, Aditi Sharma, PhD1*, Kevin Chen, BS1*, Vikas Gupta, MD, PhD3*, Shanmuganathan Chandrakasan, MD4*, Jaquelyn Zoine5,6*, Andrew Fedanov, BS5,6*, Sunil S. Raikar, MD7, Jean L. Koff, MD1, Christopher Flowers, MD, MS8, Silvia Coma, PhD9*, Jonathan A. Pachter, PhD9*, Sruthi Ravindranathan, PhD1*, H. Trent Spencer, Ph.D.5,10*, Mala Shanmugam, PhD1 and Edmund K. Waller, PhD, MD11
1Department of Hematology and Medical Oncology, Emory University School of Medicine, Atlanta, GA
2Department of Lymphoma/Myeloma, Division of Cancer Medicine, The University of Texas MD Anderson Cancer Center, Houston, TX
3Emory University, Atlanta, GA
4Division of Bone Marrow Transplant, Department of Pediatrics, Emory University School of Medicine, Atlanta, GA
5Aflac Cancer and Blood Disorders Center, Children’s Healthcare of Atlanta, Atlanta, GA
6Department of Pediatrics, Emory University School of Medicine, Atlanta
7Aflac Cancer and Blood Disorders Center, Emory University/Children’s Healthcare of Atlanta, Atlanta, GA
8Department of Lymphoma and Myeloma, The University of Texas MD Anderson Cancer Center, Houston, TX
9Verastem Oncology, Inc., Needham, MA
10Department of Pediatrics, Emory University School of Medicine, Atlanta, GA
11Department of Hematology and Medical Oncology, Winship Cancer Institute, Emory University, Atlanta, GA
*signifies non-member of ASH
Disclosures: Funk: Verastem Oncology: Research Funding. Flowers: BeiGene: Consultancy; Bayer: Consultancy; Denovo Biopharma: Consultancy; V Foundation: Research Funding; National Cancer Institute: Research Funding; Eastern Cooperative Oncology Group: Research Funding; Burroughs Wellcome Fund: Research Funding; AbbVie: Consultancy, Research Funding; Leukemia and Lymphoma Society: Membership on an entity’s Board of Directors or advisory committees; Cancer Prevention and Research Institute of Texas: Research Funding; TG Therapeutics: Research Funding; Millennium/Takeda: Consultancy, Research Funding; Spectrum: Consultancy; Acerta: Research Funding; Pharmacyclics/Janssen: Consultancy; Karyopharm: Consultancy; OptumRx: Consultancy; Genentech, Inc./F. Hoffmann-La Roche Ltd: Consultancy, Research Funding; Kite: Research Funding; Celgene: Consultancy, Research Funding; Gilead: Consultancy, Research Funding. Coma: Verastem Oncology, Inc: Current Employment, Current equity holder in publicly-traded company. Pachter: Verastem Oncology, Inc: Current Employment, Current equity holder in publicly-traded company. Waller: Verastem Oncology, Inc: Consultancy, Research Funding.
Chronic lymphocytic leukemia (CLL) creates an immune-suppressive microenvironment that induces terminal T-cell differentiation and T-cell exhaustion, thereby limiting the efficacy of chimeric antigen receptor (CAR) T cells. Indeed, analysis of CLL patients has shown that a high frequency of senescent T-cells (dashed line, Figure 1A) prior to CAR T-cell manufacturing was associated with disease progression and failure to benefit from CAR T (Fraietta et al. 2018). In a cohort of early stage untreated CLL patients, we show that 55% of patients had a predominant senescent phenotype (Figure 1A). This striking observation highlights the need for strategies to enrich non-senescent T cells for CAR T therapy in CLL patients. To address this need, we studied the effect of adding phosphoinositide 3-kinase inhibitors (PI3Ki) during ex vivo CAR T cell-manufacturing on T cell phenotype, metabolism, in vivo expansion, persistence and anti-CLL cytotoxicity in NOG mice bearing the human OSU-CLL compared with conventional CART.
To investigate the relative influence of PI3Kd-selective versus dual PI3K-d/g inhibition, we first cultured T cells from CLL patients activated with anti-CD3/CD28 beads across logarithmic dose scales of duvelisib or idelalisib. With dual-PI3K-d/g inhibition, CLL donor T cell expansion was 150% of conventionally expanded CART (Figure 1B) with dose-dependent decreases in expression of exhaustion markers TIM-3 and LAG-3 (Figure 1C). PI3K-d/g blockade increased frequencies of CD8+ CAR T cells, thus normalizing the ratio of CD4:CD8 CAR T cells (Figure 1D). To further characterize the effects of dual PI3K-d/g inhibition on T-cell phenotype, mass cytometry time-of-flight (CyTOF) analyzed the phenotype of CAR T cells cultured with or without duvelisib. Unsupervised clustering algorithms showed duvelisib increased frequencies and numbers of T-stem cell memory (Tscm), naïve, and central memory CD8+ CAR T cells (data not shown). Finally, unbiased clustering algorithms identified increases in frequencies of CD27+CD45RO- CD8+ CAR T cells which was confirmed in additional patients using flow cytometry (Figure 1E). In summary, duvelisib enriched populations of less differentiated, more potent CD8+ CAR T cells during manufacturing.
To assess whether these phenotypic changes conferred functional benefit, the cytotoxicity of CAR T cells cultured with or without duvelisib was compared using the OSU-CLL cell line. Duvelisib-cultured CAR T cells (Duv-CAR T cells) had greater cytotoxicity against OSU-CLL cell line (Figure 1F). Subsequently, gene expression profiling of Duv-CAR T cells showed decreased glycolysis pathway scores, down-regulated glucose transporters, and up-regulated glutamine transporters relative to control CAR T cells, leading us to explore T cell metabolism. Elevated protein expression of PGC1-a, a critical regulator of mitochondrial biogenesis and autophagy, was associated with increased staining with NAO, a measure of mitochondrial mass in Duv-CAR T cells (data not shown). To directly measure mitochondrial content of cells, transmission electron microscopy of control CAR T and Duv-CAR T cells generated from CLL patients was performed after 14 days of culture with representative images shown in Figure 1H. A 1.45-fold increase in mitochondrial cross-sectional area was observed (representative images in Figure 1G), which imputes a 175% increase in mitochondrial volume for duvelisib cultured CAR T cells relative to control CAR T cells (p=0.0013; Figure 1G).
Following transfer to NOG mice engrafted with a human CLL cell line, Duv-CAR T cells demonstrated greater in vivo expansion (Figure 1H), faster elimination of CLL (data not shown), and improved mouse survival (Figure 1I) in a model of high disease burden OSU-CLL. In summary, dual PI3K-d/g inhibition during CLL patient-derived CAR T-cell manufacturing increased yields of Tscm, naïve, and central memory CD8+ Duv-CAR T cells with greater mitochondrial mass and enhanced efficacy in eliminating CLL in a mouse model.
ASH Disclaimer
© 2020 American Society of Hematology. All rights reserved. ASH® and the ASH logo are registered trademarks of the American Society of Hematology. Used with permission.
This content is provided for informational purposes and personal use only, and is not intended to provide medical advice, diagnosis, or treatment or for commercial use. The ideas and opinions expressed herein do not necessarily reflect those of the American Society of Hematology (ASH). The mention of any product, service or therapy in this collection of materials should not be construed as an endorsement of the products mentioned.
Site provided by Springer Healthcare. Part of the Springer Nature Group.